-
Name
1-Dodecanal
- EINECS 203-983-6
- CAS No. 112-54-9
- Article Data205
- CAS DataBase
- Density 0.823 g/cm3
-
Solubility
Insoluble in water
-
Hazard Symbols
 Xi,
Xi,  N
N
- Synonyms Lauraldehyde(8CI);1-Dodecanal;Dodecanaldehyde;Lauric aldehyde;Laurinaldehyde;Lauryl aldehyde;NSC 46128;NSC 52196;NSC 55212;n-Dodecanal;n-Dodecyl aldehyde;n-Lauraldehyde;
- PSA 17.07000
- LogP 4.10620
Synthetic route

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical at 20℃; for 0.5h; electrolysis; | 100% |
| Stage #1: 1-dodecyl alcohol With oxalyl dichloride; dimethyl sulfoxide In dichloromethane at -78℃; for 0.25h; Oxidation; Stage #2: With triethylamine at -78 - 20℃; for 0.166667h; | 100% |
| With iodosylbenzene; Cl-CH2-PS supported 5-amino-1,10-phenanthroline-Ru In acetonitrile at 60℃; for 6h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-undecanyl-1,3-dioxolane With 2,4,6-trimethyl-pyridine; 2-methyl-2-octyl-[1,3]dioxolane; triethylsilyl trifluoromethyl sulfonate In dichloromethane at 0℃; for 1h; Stage #2: With water In dichloromethane for 0.1h; | 100% |
| Stage #1: 2-undecanyl-1,3-dioxolane With triethylsilyl trifluoromethyl sulfonate; tris-(o-tolyl)phosphine In dichloromethane at -5℃; for 0.5h; Schlenk technique; Inert atmosphere; Stage #2: With 2,6-di-tert-butyl-pyridine; water In dichloromethane at 20℃; for 1.5h; Schlenk technique; | 95% |
| With 2,6-dimethylpyridine; triethylsilyl trifluoromethyl sulfonate In dichloromethane at 0℃; for 1h; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl laurate With morpholine; diisobutylaluminium hydride In tetrahydrofuran; hexane at 0℃; for 3.16667h; Inert atmosphere; Stage #2: With lithium diisobutylmethoxy aluminum hydride In tetrahydrofuran; hexane at 0℃; for 0.166667h; Inert atmosphere; | 99% |
| With chromite at 375℃; under 735.5 Torr; Hydrogenation; |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; sodium hydrogencarbonate; potassium bromide; [4-(TEMPO-4-yloxymethyl)-1H-[1,2,3]triazol-1-ylmethyl]-PS In dichloromethane at 0℃; for 0.5h; | A n/a B 99% |
| With C30H24N2O7W; dihydrogen peroxide In water; acetonitrile for 22h; Reflux; | A 1% B 82% |
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; oxone; potassium bromide; methyltrioxorhenium(VII) In acetonitrile at 0℃; for 4h; | A 6% B 53% |
-
-
131291-89-9
1-dodecene sulfate

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane 1.) -78 deg C, 2 h, 2.) -78 deg C, 1 h; r.t., 14 h; | 99% |

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With chloral hydrate; Py(HF)x In acetonitrile at 20℃; for 5h; Product distribution; | 97% |
| With 6C72H112O8*8H2O In chloroform-d1; water for 1h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; formic acid In diethyl ether; chloroform for 0.416667h; Ambient temperature; | 96% |
| With polymer(resin Amberlyst A-26)-supported tetracarbonylhydridoferrate anion In tetrahydrofuran for 2h; Heating; | 94% |
| With pumice stone; platinum at 200 - 205℃; under 50 Torr; Hydrogenation; | |
| With piperidine; diethyldihydroaluminate 1.) THF, RT, 30 min, 2.) THF, toluene, RT, 1 h; Multistep reaction; |
-
-
14620-52-1
1,1-dimethoxy-dodecane

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| Stage #1: 1,1-dimethoxy-dodecane With triethylsilyl trifluoromethyl sulfonate; tris-(o-tolyl)phosphine In dichloromethane at -5℃; for 0.5h; Inert atmosphere; Stage #2: With water In dichloromethane at 20℃; for 1h; Inert atmosphere; | 95% |
| With copper(II) sulfate; sodium iodide In acetone at 56℃; for 0.333333h; | 93% |
| With chloral hydrate; Py(HF)x In acetonitrile at 20℃; for 1h; Product distribution; | 90% |

| Conditions | Yield |
|---|---|
| With chloro(cyclopentadienyl)[bis(diphenylphosphino)methane]ruthenium; water In isopropyl alcohol at 100℃; for 12h; | 94% |
| With dihydrogen peroxide; sodium acetate; benzo[1,3,2]dioxaborole 1.) C6H6, 80 deg C, 12 h, 2.) C6H6; Yield given. Multistep reaction; | |
| With N,N-dimethyl acetamide; benzo[1,3,2]dioxaborole 1.) CH2Cl2, room temperature, 1 h; other reagent: catecholborane; Yield given. Multistep reaction; |

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With silica gel | 93% |
-
-
874963-98-1
1-(1,2,3,4,5-pentamethyl-2,4-cyclopentadienyl)-1-dodecanol

-
A
-
112-54-9
Dodecanal

-
B
-
4045-44-7
1,2,3,4,5-pentamethylcyclopentadiene

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In toluene at 110℃; for 12h; | A 92% B n/a |

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate; 1,1,3,3-Tetramethyldisiloxane In methyl cyclohexane at 20℃; for 48h; Inert atmosphere; | 90% |
-
-
56438-07-4
1,1-dodecanediol diacetate

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| zeolite HSZ-360 for 0.333333h; Irradiation; | 88% |

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With aluminium trichloride; 1-benzyl-1-aza-4-azoniabicyclo<2.2.2>octane periodate at 20℃; for 0.583333h; | 88% |
| With cetyl(trimethyl)-ammonium tribromide In dichloromethane at 0 - 5℃; for 0.5h; | 85% |
| Stage #1: 1,1-bis-ethylsulfanyl-dodecane With acetyl chloride; sodium nitrite In dichloromethane at 0 - 5℃; for 0.25h; Stage #2: With water In dichloromethane at 0 - 20℃; for 0.916667h; | 82% |
| With dihydrogen peroxide; vanadia; ammonium bromide In dichloromethane; water at 0 - 5℃; for 1.75h; | 80% |
| With ammonium heptamolybdate; dihydrogen peroxide; ammonium bromide; perchloric acid In dichloromethane; water at 0 - 5℃; for 0.5h; | 60% |
-
-
77953-91-4
1,1-bis(phenyltelluro)dodecane

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With iodine; sodium iodide | 86% |

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water for 2.5h; | 86% |
| In tetrahydrofuran; water for 2.5h; Product distribution; Mechanism; acid-catalyzed solvolysis of polyenol ethers under var. conditions; | 86% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water for 4h; | A 36% B 85% |
-
-
13372-76-4
n-dodecanaldoxime

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; acrylic acid methyl ester In dichloromethane at 20℃; for 18h; Oxidation; | 85% |
| With manganese(IV) oxide In hexane for 0.25h; | 78% |
-
-
139021-95-7
2-Undecyl-[1,3]dithiane

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; 1-n-butyl-3-methylimidazolim bromide at 60 - 70℃; for 0.366667h; | 84% |
| With antimonypentachloride In dichloromethane at 0℃; for 0.166667h; | 81% |
| With water; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile for 1h; Ambient temperature; | 71% |
| With oxygen; 2,4,6-tris(p-chlorophenyl)pyrylium perchlorate In dichloromethane for 5h; Irradiation; | 14% |

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| Stage #1: 2-Undecyl-1,3-oxathiolane With acetyl chloride; sodium nitrite In dichloromethane at 0 - 5℃; for 0.25h; Stage #2: With water In dichloromethane at 0 - 20℃; for 0.166667h; | 84% |
| With perchloric acid; dihydrogen peroxide; ammonium bromide; molybdic acid In dichloromethane at 0 - 5℃; for 1.5h; | 76% |
| With ammonium bromide; dihydrogen peroxide; vanadia In dichloromethane at 0 - 5℃; for 1.5h; | 76% |

| Conditions | Yield |
|---|---|
| With trimethylamine-N-oxide In dimethyl sulfoxide for 5h; Ambient temperature; other halides; | 83% |
| With trimethylamine-N-oxide In dimethyl sulfoxide for 5h; Ambient temperature; | 83% |
| With sodium hydrogencarbonate; dimethyl sulfoxide; sodium iodide at 115℃; for 2h; | 76 % Chromat. |
| With 4-methylmorpholine N-oxide; potassium iodide In neat (no solvent) at 80℃; for 1h; Sonication; Green chemistry; |

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate; ammonium iron(III) sulfate; acetic acid for 3h; Heating; | A 16 mg B 82% |

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate; ammonium iron(III) sulfate for 3h; Heating; | A 16 mg B 82% |


| Conditions | Yield |
|---|---|
| With sodium tris(diethylamino)aluminum hydride In tetrahydrofuran; dodecane at 0℃; for 3h; | 82% |
| Multi-step reaction with 2 steps 1: chlorobis(cyclooctene)-iridium(I) dimer / dichloromethane / 2 h / 23 °C / Inert atmosphere 2: hydrogenchloride / water; tetrahydrofuran / 1 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In tetrahydrofuran; N,N-dimethyl-formamide at -70℃; for 0.0333333h; | A 81% B n/a |
| With pyridine; sodium tetrahydroborate In tetrahydrofuran; N,N-dimethyl-formamide at 0℃; for 0.0166667h; Product distribution; |
-
-
103383-70-6
2-undecyl-1,3-dithiolane

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With nitric acid; arsenic(III) trioxide In dichloromethane at 0 - 5℃; | 81% |
| With water; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile for 1h; Ambient temperature; | 21% |

| Conditions | Yield |
|---|---|
| manganese(II) iodide In tetrahydrofuran at 70℃; for 2h; | A 80% B 3% C n/a |


-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium carbonate In various solvent(s) | 78% |

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water for 3h; | 75% |
-
-
1236031-82-5
n-dodecanal diisopropyl acetal

-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| Stage #1: n-dodecanal diisopropyl acetal With triethylsilyl trifluoromethyl sulfonate; tris-(o-tolyl)phosphine In dichloromethane at -5℃; for 4h; Stage #2: With water In dichloromethane at 20℃; for 0.5h; | 72% |
| Multi-step reaction with 2 steps 1: dichloromethane / 0.5 h / -5 °C / Schlenk technique; Inert atmosphere 2: water / dichloromethane / 0.5 h / 20 °C / Schlenk technique View Scheme |
-
-
112-54-9
Dodecanal

-
-
113999-43-2, 113999-44-3
(2R*,3S*)-N-hydroxy-3-(trimethylsilyl)-4-penten-2-amine

-
-
113999-27-2
(2R*,3S*)-(Z)-N-undecylidene-3-(trimethylsilyl)-4-penten-2-amine N-oxide

| Conditions | Yield |
|---|---|
| With calcium chloride In diethyl ether at -20℃; for 12h; | 100% |

| Conditions | Yield |
|---|---|
| ytterbium (III) tris-[tris-(nonafluorobutanesulfonyl)methide] In ethanol for 48h; Heating / reflux; | 100% |
| With hydrogenchloride In ethanol; water at -10 - 90℃; for 20.1667h; | 82% |
| With hydrogenchloride In ethanol at 20 - 90℃; for 17h; | 76% |

| Conditions | Yield |
|---|---|
| Stage #1: Methoxyallene With n-butyllithium In diethyl ether; hexane at -40℃; for 0.166667h; Inert atmosphere; Stage #2: Dodecanal In diethyl ether; hexane at -40℃; Inert atmosphere; | 100% |
| Stage #1: Methoxyallene With n-butyllithium In diethyl ether; hexane at -40℃; Stage #2: Dodecanal In diethyl ether; hexane at -78℃; Further stages.; | |
| Stage #1: Methoxyallene With n-butyllithium In diethyl ether; hexane at -40℃; for 0.5h; Inert atmosphere; Stage #2: Dodecanal In diethyl ether; hexane at -78℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; thiourea at 23℃; for 0.75h; | 99% |
| With Br(1-)*C19H14Br3O3PS*Na(1+) at 20℃; | 92% |
| With Zr-MOF-808 at 45℃; for 22h; Irradiation; | 7% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride; diphenylstibane In tetrahydrofuran for 3h; Ambient temperature; | 99% |
| Stage #1: Dodecanal With phenylsilane; potassium tert-butylate In toluene at 20℃; for 0.5h; Inert atmosphere; Stage #2: With water; sodium hydroxide In toluene at 0 - 20℃; | 99% |
| With LaNi5 hydride In tetrahydrofuran; methanol 1) 0 deg C, 4 h, 2) r.t., 14 h; | 98% |
-
-
112-54-9
Dodecanal

-
-
34939-91-8, 58648-56-9
dimethyl lithiomethylphosphonate

-
-
88708-59-2
dimethyl 2-hydroxytridecylphosphonate

| Conditions | Yield |
|---|---|
| 99% |

| Conditions | Yield |
|---|---|
| With iron oxide; oxygen; ethyl acetoacetate at 75 - 80℃; under 760.051 Torr; for 24h; Green chemistry; | 99% |
| With oxygen; copper(II) acetate monohydrate; cobalt(II) diacetate tetrahydrate In water at 40℃; under 760.051 Torr; for 3h; | 97% |
| With Iron(III) nitrate nonahydrate; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium chloride; oxygen In 1,2-dichloro-ethane at 25℃; for 12h; | 94% |
-
-
112-54-9
Dodecanal

-
-
756-79-6
dimethyl methane phosphonate

-
-
88708-59-2
dimethyl 2-hydroxytridecylphosphonate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl methane phosphonate With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.0833333h; Metallation; Stage #2: Dodecanal In tetrahydrofuran; hexane at -78℃; for 0.0833333h; Addition; | 99% |
-
-
112-54-9
Dodecanal

-
-
16079-51-9
sodium glutamate

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 99% |
-
-
112-54-9
Dodecanal

| Conditions | Yield |
|---|---|
| With Iron(III) nitrate nonahydrate; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium chloride; oxygen; 18O-labeled water In 1,2-dichloro-ethane at 25℃; for 12h; | 99% |
| With Iron(III) nitrate nonahydrate; oxygen; 18O-labeled water In acetonitrile at 25℃; under 760.051 Torr; for 12h; Schlenk technique; | 89% |
-
-
112-54-9
Dodecanal

-
-
21382-82-1
(carbethoxyethylidene)triphenylphosphorane

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 18h; | 98% |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium iodide In acetonitrile for 1.46667h; Heating; | 98% |
| With hydroxylamine hydrochloride In dimethyl sulfoxide at 100℃; for 0.5h; | 96% |
| With hydroxylamine hydrochloride In o-xylene at 133℃; under 760.051 Torr; for 5h; | 89% |
-
-
112-54-9
Dodecanal

-
-
55-21-0
benzamide

-
-
135-19-3
β-naphthol

-
-
1372802-77-1
N-(1-(2-hydroxynaphthalen-1-yl)dodecyl)benzamide

| Conditions | Yield |
|---|---|
| With zirconyl triflate In neat (no solvent) at 80℃; for 0.0333333h; | 98% |
| With dodecylphosphonic acid at 90℃; for 0.5h; Neat (no solvent); | 70% |

-
-
112-54-9
Dodecanal

-
-
124156-21-4
ethyl (4-methoxyphenylimino)acetate

| Conditions | Yield |
|---|---|
| Stage #1: Dodecanal With 1-(3,5-bis(trifluoromethyl)phenyl)-3-((R)-(6-methoxyquinolin-4-yl)((1S,2R,4S,5R)-5-vinylquinuclidin-2-yl)methyl)thiourea; L-proline In neat (no solvent) at 25℃; for 0.166667h; Stage #2: ethyl (4-methoxyphenylimino)acetate In neat (no solvent) at 25℃; for 0.0833333h; Catalytic behavior; Reagent/catalyst; Solvent; Time; Concentration; stereoselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| zeolite HSZ-360 for 4h; Ambient temperature; | 97% |
| With boron trifluoride diethyl etherate at 20℃; Esterification; salt-ice cooling; | 75% |
| With boron trifluoride Addition; | 75% |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 2h; | 97% |
-
-
112-54-9
Dodecanal

-
-
1099-45-2
ethyl (triphenylphosphoranylidene)acetate

-
-
38112-60-6
(E)-ethyl tetradec-2-enoate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 15h; Wittib reaction; | 97% |
| In benzene at 0 - 20℃; for 6h; Wittig reaction; | 84% |
| In dichloromethane at 20℃; Wittig Olefination; | 45% |

| Conditions | Yield |
|---|---|
| With copper(II) acetate monohydrate In ethyl acetate at 20℃; for 12h; | 97% |
| With zinc(II) acetate dihydrate In acetonitrile at 20℃; for 18h; | 93% |
-
-
112-54-9
Dodecanal

-
-
89524-99-2
methyl 2-acetylamino-2-(dimethoxyphosphinyl)acetate

| Conditions | Yield |
|---|---|
| Stage #1: methyl 2-acetylamino-2-(dimethoxyphosphinyl)acetate With N,N,N',N'-tetramethylguanidine In tetrahydrofuran at -78℃; Horner-Wadsworth-Emmons Olefination; Stage #2: Dodecanal In tetrahydrofuran at -78 - 25℃; for 4h; Horner-Wadsworth-Emmons Olefination; | 97% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; for 1.75h; | 96.8% |
| In tetrahydrofuran; diethyl ether at -50 - 20℃; |

| Conditions | Yield |
|---|---|
| With phenyltellurotrimethylsilane; zinc(II) iodide In benzene for 3h; Ambient temperature; | 96% |

| Conditions | Yield |
|---|---|
| In diethyl ether for 1.5h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With triethylsilane; tris(pentafluorophenyl)borate In dichloromethane at 20℃; for 20h; | 96% |
| Stage #1: Dodecanal With triethylsilane; tris(pentafluorophenyl)borate In dichloromethane at 20℃; for 20h; Stage #2: In ethanol for 7h; Heating; Further stages.; | 96% |
| With hydrogen In neat (no solvent) at 180℃; under 6000.6 Torr; for 1h; Autoclave; | 71% |
| Multi-step reaction with 2 steps 1: methanol / 60 °C / Inert atmosphere 2: potassium carbonate; methanol; hydrogen; palladium 10% on activated carbon / 24 h / 65 °C / 760.05 Torr View Scheme | |
| With hydrogen In neat (no solvent) at 180℃; under 6000.6 Torr; Catalytic behavior; Autoclave; High pressure; |
Dodecyl aldehyde Chemical Properties
Product Name: Dodecyl aldehyde
Synonyms of Dodecyl aldehyde (CAS NO.112-54-9): Dodecanal ; Lauraldehyde
CAS NO: 112-54-9
Molecular Formula: C12H24O
Molecular Weight: 184.32 g/mol
Molecular Structure: 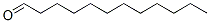
EINECS: 203-983-6
RTECS: JR1910000
Density: 0.823 g/cm3
Melting Point: 12 °C
Flash Point: 102 °C
Boiling Point: 242.2 °C at 760 mmHg
Storage temp: Refrigerator
Water Solubility: Insoluble
Appearance: colourless liquid
Stability: Stable. Combustible. Incompatible with strong oxidizing agents, strong reducing agents, strong bases
Dodecyl aldehyde Uses
Dodecyl aldehyde (CAS NO.112-54-9) is used as flavor and organic synthesis intermediate.
Dodecyl aldehyde Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 23gm/kg (23000mg/kg) | Food and Cosmetics Toxicology. Vol. 11, Pg. 483, 1973. |
Dodecyl aldehyde Consensus Reports
Reported in EPA TSCA Inventory.
Dodecyl aldehyde Safety Profile
Safety Information about Dodecyl aldehyde (CAS NO.112-54-9):
Hazard Codes:  Xi,
Xi, N
N
Risk Statements: 36/37/38-51/53
R36/37/38:Irritating to eyes, respiratory system and skin.
R51/53:Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
Safety Statements: 26-36/37-61-29
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S29: Do not empty into drains.
S36/37: Wear suitable protective clothing and gloves.
S61: Avoid release to the environment. Refer to special instructions / safety data sheets.
WGK Germany: 2
RTECS: JR1910000
F: 10-23
Hazard Note: Irritant
Mildly toxic by ingestion. A human and experimental skin irritant. Combustible liquid. When heated to decomposition it emits acrid smoke and irritating fumes. See also ALDEHYDES.
Dodecyl aldehyde Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media: Use water spray, dry chemical, carbon dioxide, or chemical foam.
Handling: Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage: Do not store in direct sunlight. Store in a tightly closed container. Store in a dry area. Keep refrigerated. (Store below 4°C/39°F.)
Related Products
- Dodecyl (9Z,12Z)-octadeca-9,12-dienoate
- Dodecyl (Z,Z)-6,6-dioctyl-4,8,11-trioxo-5,7,12-trioxa-6-stannatetracosa-2,9-dienoate
- Dodecyl 2-methylacrylate
- Dodecyl 3-amino-4-chlorobenzoate
- Dodecyl 4-chloro-3-nitrobenzoate
- Dodecyl 4-hydroxybenzoate
- Dodecyl 4-methylbenzenesulfonate
- Dodecyl acrylate
- Dodecyl aldehyde
- Dodecyl benzene sulfonic acid
- 112-55-0
- 1125592-35-9
- 112559-81-6
- 1125-60-6
- 112565-42-1
- 112566-17-3
- 1125-66-2
- 112568-12-4
- 112571-69-4
- 112-57-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View