-
Name
KETENE DIETHYL ACETAL
- EINECS
- CAS No. 2678-54-8
- Article Data28
- CAS DataBase
- Density 0.859 g/cm3
- Solubility
- Melting Point
- Formula C6H12O2
- Boiling Point 105.645 °C at 760 mmHg
- Molecular Weight 116.16
- Flash Point 7.004 °C
- Transport Information UN 1993 3/PG 3
- Appearance
- Safety 23
- Risk Codes 10
-
Molecular Structure
- Hazard Symbols
- Synonyms Ketene,diethyl acetal (6CI,7CI,8CI);1,1-Diethoxyethylene;Diethylketene acetal;
- PSA 18.46000
- LogP 1.53060
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 130 - 140℃; under 100 Torr; | 92% |
| With potassium tert-butylate for 0.1 - 0.183333h; Cooling with ice; | 72% |
| With potassium tert-butylate at 20℃; Reflux; Inert atmosphere; | 69.9% |

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium tert-butylate In tetrahydrofuran at 0℃; for 2h; Elimination; | 70% |
-
-
855175-05-2
β-(1,1-diethoxy-ethoxy)-chalcone

-
A
-
2678-54-8
ketene diethyl acetal

-
B
-
120-46-7
1,3-diphenylpropanedione

| Conditions | Yield |
|---|---|
| at 140℃; under 38 Torr; |

| Conditions | Yield |
|---|---|
| With aluminium tert-butyl oleate at 180℃; |
-
-
865-47-4
potassium tert-butylate

-
-
51806-20-3
1,1-diethoxy-2-iodoethane

-
-
2678-54-8
ketene diethyl acetal
-
-
51806-20-3
1,1-diethoxy-2-iodoethane

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| With potassium tert-butylate | |
| With potassium tert-butylate |

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| With sodium methylate for 0.666667h; Ambient temperature; Yield given; |

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| With sodium; benzene |

-
-
75-85-4
tert-Amyl alcohol

-
-
1634-04-4
tert-butyl methyl ether

-
-
107-21-1
ethylene glycol

-
-
621-62-5
chloroacetaldehyde diethyl acetal

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| With potassium hydroxide In Dimethyl ether |

-
-
75-85-4
tert-Amyl alcohol

-
-
107-21-1
ethylene glycol

-
-
621-62-5
chloroacetaldehyde diethyl acetal

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| With potassium hydroxide In Dimethyl ether; tert-butyl methyl ether |

-
-
127826-29-3
N-propylsulfonylchloralimine

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| In dichloromethane Ambient temperature; | 100% |
-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| dirhodium tetraacetate In dichloromethane at 20℃; for 2h; | 100% |
-
-
2678-54-8
ketene diethyl acetal

-
-
436146-73-5
(2-ethynylcyclohex-1-en-1-yl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| chromium(0) hexacarbonyl In tetrahydrofuran at 20℃; for 2h; | 99% |
| With pentacarbonyl(tetrahydrofuran)chromium(0) In tetrahydrofuran at 20℃; for 2h; | 99% |
-
-
2678-54-8
ketene diethyl acetal

-
-
74357-22-5
methyl 6-ethoxynicotinate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 60℃; for 0.5h; Diels-Alder reaction; Inert atmosphere; regioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With (2,4,5-trifluorophenyl)boronic acid In tert-butyl methyl ether at 50℃; for 14h; | 98% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
2678-54-8
ketene diethyl acetal

-
-
97585-04-1, 20511-20-0, 24393-49-5
ethyl (E)-3-(4-methylphenyl)acrylate

| Conditions | Yield |
|---|---|
| With (2,4,5-trifluorophenyl)boronic acid In tert-butyl methyl ether at 50℃; for 14h; | 97% |
-
-
2678-54-8
ketene diethyl acetal

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
24393-53-1, 136265-11-7, 15795-20-7
ethyl (E)-3-(4-bromophenyl)-2-propenoate

| Conditions | Yield |
|---|---|
| With (2,4,5-trifluorophenyl)boronic acid In tert-butyl methyl ether at 50℃; for 14h; | 97% |
-
-
70591-46-7
(S)-5-methoxycarbonylmethyl-6,6-dimethyl-2-cyclohexen-1-one

-
-
2678-54-8
ketene diethyl acetal

-
-
70591-47-8, 82509-28-2, 82509-31-7
4-carbomethoxymethyl-7,7-diethoxy-3,3-dimethylbicyclo<4.2.0>octan-2-one

| Conditions | Yield |
|---|---|
| for 3h; Ambient temperature; Irradiation; | 96% |
-
-
2678-54-8
ketene diethyl acetal

-
-
684-16-2
Hexafluoroacetone

-
-
63504-98-3
4,4-diethoxy-1,1,1-trifluoro-2-trifluoromethyl-but-3-en-2-ol

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 95% |
-
-
2678-54-8
ketene diethyl acetal

-
-
213320-13-9
6-[(benzyloxycarbonyl)amino]-3-(methylthio)-1,2,4,5-tetrazine

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 100℃; for 2h; Diels-Alder reaction; | 95% |

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene at 140℃; for 24h; Diels-Alder reaction; regioselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| In toluene at 150℃; for 17h; Diels-Alder Cycloaddition; Inert atmosphere; regioselective reaction; | 95% |
-
-
79-52-7
1,1,3-trichloro-1,3,3-trifluoroacetone

-
-
2678-54-8
ketene diethyl acetal

-
-
138351-94-7
1,1-Diethoxy-4,4-difluoro-4-chloro-3-fluorodichloromethyl-1-buten-3-ol

| Conditions | Yield |
|---|---|
| for 18h; Ambient temperature; | 94% |
-
-
2678-54-8
ketene diethyl acetal

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
24393-56-4
ethyl (E)-3-(4-methoxyphenyl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With (2,4,5-trifluorophenyl)boronic acid In tert-butyl methyl ether at 50℃; for 14h; | 94% |
-
-
66-77-3
1-naphthaldehyde

-
-
2678-54-8
ketene diethyl acetal

-
-
98978-43-9
(E)-ethyl 3-(naphthalen-1-yl)acrylate

| Conditions | Yield |
|---|---|
| With (2,4,5-trifluorophenyl)boronic acid In tert-butyl methyl ether at 50℃; for 14h; | 93% |
-
-
126685-93-6
1-(2-diazo-1,3-dioxobutyl)-2-pyrrolidinone

-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| dirhodium tetraacetate In benzene for 2h; Heating; | 92% |
-
-
58303-62-1
5-(4-chlorophenyl)-2,3-dihydrofuran-2,3-dione

-
-
2678-54-8
ketene diethyl acetal

-
-
159107-87-6
[5-(4-Chloro-phenyl)-2-hydroxy-3-oxo-2,3-dihydro-furan-2-yl]-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 40℃; for 1h; | 91% |
-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| In toluene for 1.5h; Heating; | 91% |
-
-
2678-54-8
ketene diethyl acetal

-
-
84011-69-8
5-<<<(1,1-dimethylethyl)dimethylsilyl>oxy>methyl>-2-cyclohexen-1-one

-
-
84011-67-6
4-<<<(1,1-dimethylethyl)dimethylsilyl>oxy>methyl>-7,7-diethoxybicyclo<4.2.0>octane-2-one

| Conditions | Yield |
|---|---|
| In benzene for 3.5h; Irradiation; | 90% |
-
-
2678-54-8
ketene diethyl acetal

-
-
623-91-6
diethyl Fumarate

-
-
127696-10-0
trans-3,3-diethoxy-1,2-cyclobutanedicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| With diisobutylaluminum chloride; N-ethyl-N,N-diisopropylamine In toluene at -40℃; | 90% |
| In tert-butyl alcohol at 84℃; for 72h; | 54% |
| In tert-butyl alcohol at 84℃; for 72h; | 53% |
| Stage #1: ketene diethyl acetal With diethylaluminium chloride In toluene at -45℃; for 0.166667h; Inert atmosphere; Stage #2: With N-ethyl-N,N-diisopropylamine In toluene at -45℃; for 0.166667h; Inert atmosphere; Stage #3: diethyl Fumarate In toluene at -45℃; for 3h; Inert atmosphere; | 42.3% |
-
-
2678-54-8
ketene diethyl acetal

-
-
55991-70-3
5-(4-bromo-phenyl)-furan-2,3-dione

-
-
159107-86-5
[5-(4-Bromo-phenyl)-2-hydroxy-3-oxo-2,3-dihydro-furan-2-yl]-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 40℃; for 1h; | 90% |
-
-
37643-54-2
N-[(4-methylbenzene-1-sulfonyl)(phenyl)methyl]formamide

-
-
2678-54-8
ketene diethyl acetal

-
-
850011-53-9
3-formylamino-3-phenyl-propionic acid ethyl ester

| Conditions | Yield |
|---|---|
| With caesium carbonate In dichloromethane for 24h; Heating; | 90% |
| With caesium carbonate In dichloromethane for 24h; Product distribution / selectivity; Heating / reflux; | 90% |
| With triethylamine In dichloromethane for 24h; Product distribution / selectivity; Heating / reflux; | 80% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane for 24h; Product distribution / selectivity; Heating / reflux; | 48% |
-
-
141-97-9
ethyl acetoacetate

-
-
2678-54-8
ketene diethyl acetal

-
-
182916-87-6
Ethyl 2-acetyl-3-ethoxybut-2-enoate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol at 22 - 90℃; for 8.5h; | 89% |
-
-
904731-83-5
5-chloro-2,2-dimethyl-1,3-dioxin-4-one

-
-
2678-54-8
ketene diethyl acetal

-
-
904731-84-6
5-chloro-2,2-diethoxy-2,3-dihydropyran-4-one

| Conditions | Yield |
|---|---|
| In toluene for 0.666667h; Product distribution / selectivity; Heating / reflux; | 89% |
-
-
2678-54-8
ketene diethyl acetal

-
-
2043-61-0
cyclohexanecarbaldehyde

-
-
17343-88-3
(E)-3-cyclohexyl-acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With (2,4,5-trifluorophenyl)boronic acid In tert-butyl methyl ether at 50℃; for 14h; | 89% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In cyclohexane at 20℃; for 0.0833333h; Inert atmosphere; | 89% |
-
-
3888-00-4
nitropentafluoroacetone

-
-
2678-54-8
ketene diethyl acetal

-
-
40657-28-1
4,4-diethoxy-1,1-difluoro-1-nitro-2-trifluoromethyl-but-3-en-2-ol

| Conditions | Yield |
|---|---|
| at -30°C, then at 20°C 2 h; | 88% |
| at -30°C, then at 20°C 2 h; | 88% |
-
-
2678-54-8
ketene diethyl acetal

| Conditions | Yield |
|---|---|
| In acetonitrile at 20 - 50℃; for 19h; Cycloaddition; | 87% |
-
-
401-78-5
3-bromo-1-trifluoromethylbenzene

-
-
2678-54-8
ketene diethyl acetal

-
-
1017235-56-1
1,1-diethoxy-6-(trifluoromethyl)benzocyclobutene

| Conditions | Yield |
|---|---|
| With sodium amide In tetrahydrofuran for 22h; Heating; | 87% |
Ethene, 1,1-diethoxy- Specification
The Ethene, 1,1-diethoxy-, with the CAS registry number 2678-54-8, is also known as Ketene, diethyl acetal. This chemical's molecular formula is C6H12O2 and molecular weight is 116.16. What's more, both its IUPAC name and systematic name are the same which is called 1,1-Diethoxyethene. When you are dealing with this chemical, you should be very careful. This chemical is flammable. Therefore, you can not breathe the gas/fumes/vapour/spray.
Physical properties about Ethene, 1,1-diethoxy- are: (1)ACD/LogP: 1.195; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.20; (4)ACD/LogD (pH 7.4): 1.20; (5)ACD/BCF (pH 5.5): 4.77; (6)ACD/BCF (pH 7.4): 4.77; (7)ACD/KOC (pH 5.5): 106.42; (8)ACD/KOC (pH 7.4): 106.42; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 18.46 Å2; (13)Index of Refraction: 1.402; (14)Molar Refractivity: 32.905 cm3; (15)Molar Volume: 135.196 cm3; (16)Polarizability: 13.044×10-24cm3; (17)Surface Tension: 22.788 dyne/cm; (18)Density: 0.859 g/cm3; (19)Flash Point: 7.004 °C; (20)Enthalpy of Vaporization: 33.033 kJ/mol; (21)Boiling Point: 105.645 °C at 760 mmHg; (22)Vapour Pressure: 34.079 mmHg at 25 °C.
Preparation of Ethene, 1,1-diethoxy-: this chemical can be prepared by 2-bromo-1,1-diethoxy-ethane. This reaction needs reagent t-BuOK at temperature of 120-130 °C.

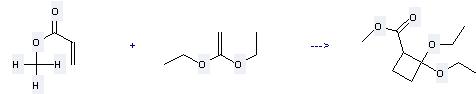
Uses of Ethene, 1,1-diethoxy-: it is used to produce other chemicals. For example, it can react with acrylic acid methyl ester to get 2.2-Diethoxy-cyclobutancarbonsaeuremethylester. The reaction occurs with solvent acetonitrile and other condition of heating for 8 days. The yield is 17 %.
You can still convert the following datas into molecular structure:
(1) SMILES: CCOC(=C)OCC
(2) InChI: InChI=1S/C6H12O2/c1-4-7-6(3)8-5-2/h3-5H2,1-2H3
(3) InChIKey: VTGIVYVOVVQLRL-UHFFFAOYSA-N
Related Products
- Ethene Homopolymer Chlorinated Chlorosulfonated
- Ethene, 1,1-diethoxy-
- Ethene, homopolymer, oxidized
- Ethene, polymer with carbon monoxide
- Ethene,1-(2-bromo-1,1,2,2-tetrafluoroethoxy)-1,2,2-trifluoro-
- Ethene,1,1,2-trifluoro-2-iodo-
- Ethene,1,1-dibromo-2-fluoro-
- Ethene,1-bromo-2-fluoro- (9CI)
- Ethene,1-chloro-1,2-difluoro- (9CI)
- Ethene,1-chloro-2-fluoro- (9CI)
- 267875-39-8
- 267875-45-6
- 267875-65-0
- 267875-68-3
- 267876-26-6
- 26787-78-0
- 267880-81-9
- 267880-87-5
- 267881-10-7
- 2679-14-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View