-
Name
Ethyl isobutyrate
- EINECS 202-595-4
- CAS No. 97-62-1
- Article Data88
- CAS DataBase
- Density 0.883 g/cm3
- Solubility Not miscible or difficult to mix in water. Soluble in alcohol.
- Melting Point -88 °C
- Formula C6H12O2
- Boiling Point 112.6 °C at 760 mmHg
- Molecular Weight 116.16
- Flash Point 19.4 °C
- Transport Information UN 2385 3/PG 2
- Appearance clear colourless liquid
- Safety 16-26-36-36/37/39
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Isobutyricacid, ethyl ester (6CI,8CI);2-Methylpropanoic acid ethyl ester;Ethyl 2,2-dimethylacetate;Ethyl 2-methylpropionate;Ethylisobutyrate;NSC 97194;
- PSA 26.30000
- LogP 1.20550
Synthetic route

| Conditions | Yield |
|---|---|
| at 120℃; for 24h; | 98% |

| Conditions | Yield |
|---|---|
| With water; lithium diisopropyl amide In tetrahydrofuran 1) -78 deg C, 30 min 2) 30 min; | 96% |
| With Graphite; acetophenone; potassium bromide In water at 25℃; Reagent/catalyst; Electrolysis; | |
| With trans-Mo2(μ-N,N′-bis(4-methoxyphenyl)formamidinato-κ2N:N′)2(OOCCH3)2; 1-methyl-3,6-bis(trimethylsilyl)-1,4-cyclohexadiene; tetrabutyl-ammonium chloride In acetonitrile at 80℃; for 24h; | 73 %Spectr. |
| With di-tert-butoxydiazene; tri-n-butyl-tin hydride In cyclohexane at 42℃; Kinetics; Inert atmosphere; Darkness; |

| Conditions | Yield |
|---|---|
| With S,S-bis<4,6-dimethyl-2-pyrimidinyl> dithiocarbonate; dmap In dichloromethane for 1.5h; Ambient temperature; | 91% |
| With toluene-4-sulfonic acid In cyclohexane Reflux; | 68.9% |
| With Rhizomucor miehei lipase In n-heptane at 40℃; for 24h; Enzymatic reaction; | 65.7% |

| Conditions | Yield |
|---|---|
| In benzene Product distribution; Irradiation; | A 90% B 90% |

-
-
64-17-5
ethanol

-
-
2446-83-5
di-isopropyl azodicarboxylate

-
-
565-80-0
2,4-dimethylpentan-3-one

-
B
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Stage #1: di-isopropyl azodicarboxylate; 2,4-dimethylpentan-3-one With cerium(IV) trifluoromethanesulfonate; trimethylsilyl cyanide; 9,10-diphenylanthracene; tetrabutyl-ammonium chloride; titanium tetrachloride In acetonitrile at 0 - 50℃; for 24h; Inert atmosphere; Irradiation; Glovebox; Stage #2: ethanol With triethylamine In acetonitrile at 20℃; for 2h; Inert atmosphere; Glovebox; | A 87% B 90 %Chromat. |

-
-
822-83-3
2-isopropyl-1,3-dioxolane

-
A
-
6942-58-1
2-hydroxyethyl isobutyrate

-
B
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With Cumene hydroperoxide In chlorobenzene at 130℃; for 1h; Product distribution; Rate constant; | A 70% B n/a |
| With Cumene hydroperoxide at 110℃; |
-
-
474094-06-9
ethyl 2-methyl-2-methyl tellurium propionate

-
A
-
20334-43-4
dimethyl ditelluride

-
B
-
79-31-2
isobutyric Acid

-
C
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran; water at 70℃; for 3h; Inert atmosphere; | A 52% B 14% C 22% |

-
-
600-00-0
ethyl 2-bromoisobutyrate

-
-
10170-69-1
dimanganese decacarbonyl

-
A
-
33367-54-3
diethyl 2,2,3,3-tetramethylsuccinate

-
C
-
106471-98-1
diethyl 2,2-dimethyl-4-methylenepenta-1,5-dioate

-
D
-
97-63-2
methyl methacrylate

-
E
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Irradiation (UV/VIS); (N2 or Ar); 250-W sun lamp, 3 h; colorless solution is filtered, (1)H-NMR; | A 45% B 13% C 5% D 12% E 17% |
| In chloroform-d1 Irradiation (UV/VIS); (N2 or Ar); 250-W sun lamp, 3 h; colorless solution is filtered, (1)H-NMR; | A 36% B 9% C 4% D 5% E 13% |


| Conditions | Yield |
|---|---|
| In benzene Product distribution; Irradiation; | A 41% B 38% |

-
-
600-00-0
ethyl 2-bromoisobutyrate

-
A
-
33367-54-3
diethyl 2,2,3,3-tetramethylsuccinate

-
B
-
106471-98-1
diethyl 2,2-dimethyl-4-methylenepenta-1,5-dioate

-
C
-
97-63-2
methyl methacrylate

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With {bis(triphenylphosphine)nitrogen}{manganese(carbonyl)5} In tetrahydrofuran for 5h; | A 36% B 3% C 6% D 35% |

-
-
600-00-0
ethyl 2-bromoisobutyrate

-
-
52542-59-3, 192520-17-5
bis(triphenylphosphineiminium) pentacarbonylmanganate(1-)

-
A
-
33367-54-3
diethyl 2,2,3,3-tetramethylsuccinate

-
B
-
106471-98-1
diethyl 2,2-dimethyl-4-methylenepenta-1,5-dioate

-
C
-
97-63-2
methyl methacrylate

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (N2 or Ar); solution is allowed to stand for 5 h; determination by NMR and MS fragmentation; | A 36% B 3% C 6% D 35% |

-
-
600-00-0
ethyl 2-bromoisobutyrate

-
-
13859-41-1
sodium pentacarbonyl manganate

-
A
-
33367-54-3
diethyl 2,2,3,3-tetramethylsuccinate

-
B
-
106471-98-1
diethyl 2,2-dimethyl-4-methylenepenta-1,5-dioate

-
C
-
97-63-2
methyl methacrylate

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (N2 or Ar); solution is allowed to stand for 5 h; determination by NMR and MS fragmentation; | A 14% B 1% C 16% D 36% |

-
-
64-17-5
ethanol

-
-
151699-17-1
3-isobutanoyl-thiazolidine-2-thione

-
A
-
134469-06-0
thiazolidine-2-thione

-
B
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Irradiation; Yields of byproduct given; | A n/a B 17% |

-
-
600-00-0
ethyl 2-bromoisobutyrate

-
A
-
33367-54-3
diethyl 2,2,3,3-tetramethylsuccinate

-
B
-
106471-98-1
diethyl 2,2-dimethyl-4-methylenepenta-1,5-dioate

-
C
-
127599-82-0
diethyl 3-carbethoxy-2,2,3,5,5-pentamethylhexa-1,6-dioate

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With dimanganese decacarbonyl In tetrahydrofuran for 0.5h; Irradiation; or CDCl3; Further byproducts given; | A 45 % Chromat. B 5 % Chromat. C 13% D 17 % Chromat. |

-
-
600-00-0
ethyl 2-bromoisobutyrate

-
-
119-61-9
benzophenone

-
A
-
597-04-6
ethyl 2,2-dimethyl-3-oxobutanoate

-
B
-
33367-54-3
diethyl 2,2,3,3-tetramethylsuccinate

-
C
-
59697-75-5
Ethyl 3-hydroxy-2,2-dimethyl-3,3-diphenylpropanoate

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With tetrabutylammonium tetrafluoroborate; zinc dibromide; zinc In acetonitrile Ambient temperature; | A n/a B n/a C 10% D n/a |

-
-
597-04-6
ethyl 2,2-dimethyl-3-oxobutanoate

-
-
60-29-7
diethyl ether

-
-
141-52-6
sodium ethanolate

-
A
-
26823-10-9
2,2-dimethyl-3,5-dioxo-hexanoic acid ethyl ester

-
B
-
97-62-1
Ethyl isobutyrate

-
-
60-29-7
diethyl ether

-
-
52178-65-1, 133270-73-2, 35022-34-5
ethyl 3-hydroxy-2,2-dimethyl-3-phenylpropanoate

-
-
141-52-6
sodium ethanolate

-
A
-
65-85-0
benzoic acid

-
B
-
97-62-1
Ethyl isobutyrate

-
C
-
100-51-6
benzyl alcohol

-
-
64-17-5
ethanol

-
-
63289-89-4
5-methyl-hexane-2,4-dione 2-enol tautomer

-
A
-
67-64-1
acetone

-
B
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Geschwindigkeit; |


| Conditions | Yield |
|---|---|
| With aluminum oxide at 325℃; |
-
-
54285-47-1
ethyl 2-ethyl-4-methyl-3-oxopentanoate

-
-
93-89-0
benzoic acid ethyl ester

-
A
-
24346-56-3
2-benzoyl-butyric acid ethyl ester

-
B
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| at 145℃; Na-Verbindung; |

-
-
857793-16-9
2-isobutyryl-4-methyl-glutaric acid diethyl ester

-
-
141-52-6
sodium ethanolate

-
A
-
18545-83-0
diethyl 2-methylglutarate

-
B
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| at 115 - 120℃; |

-
-
555-75-9
aluminum ethoxide

-
-
100-52-7
benzaldehyde

-
-
78-84-2
isobutyraldehyde

-
A
-
103-28-6
benzyl 2-methylpropanoate

-
B
-
120-50-3
isobutyl benzoate

-
C
-
120-51-4
benzoic acid benzyl ester

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Produkt 5:Isobutylisobutyrat; |


| Conditions | Yield |
|---|---|
| With sodium; methyl iodide |
-
-
2234-82-4
1-propylmagnesium chloride

-
-
1619-62-1
2,2-dimethylmalonic acid diethyl ester

-
A
-
7379-12-6
n-propyl isopropyl ketone

-
B
-
123-19-3
4-heptanone

-
C
-
589-55-9
heptan-4-ol

-
D
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Produkt 5: Tripropylcarbinol; Produkt 6: 5.5-Dimethyl-nonandiol-(4.6); |


-
-
1619-62-1
2,2-dimethylmalonic acid diethyl ester

-
A
-
19424-43-2
2,3,3,4-tetramethylpentane-2,4-diol

-
B
-
62817-86-1
3-hydroxy-2,2,3-trimethyl-butyric acid ethyl ester

-
C
-
67-64-1
acetone

-
D
-
97-62-1
Ethyl isobutyrate

-
-
10557-57-0
propylmagnesium iodide

-
-
1619-62-1
2,2-dimethylmalonic acid diethyl ester

-
A
-
123-19-3
4-heptanone

-
B
-
97-62-1
Ethyl isobutyrate

-
-
1619-62-1
2,2-dimethylmalonic acid diethyl ester

-
A
-
119-61-9
benzophenone

-
B
-
92-52-4
biphenyl

-
C
-
97-62-1
Ethyl isobutyrate


| Conditions | Yield |
|---|---|
| With diethyl ether; sodium | |
| With hydrogen; platinum at 200℃; |
-
-
159751-61-8
((S)-3-Iodo-2-methyl-propoxymethoxymethyl)-benzene

-
-
97-62-1
Ethyl isobutyrate

-
-
159751-62-9
(R)-5-Benzyloxymethoxy-2,2,4-trimethyl-pentanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran | 100% |
-
-
591-31-1
3-methoxy-benzaldehyde

-
-
97-62-1
Ethyl isobutyrate

-
-
602304-70-1, 293769-53-6
ethyl 3-hydroxy-3-(3-methoxyphenyl)-2,2-dimethylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.5h; Stage #2: 3-methoxy-benzaldehyde In tetrahydrofuran at -78℃; for 1.5h; | 100% |
-
-
777075-01-1
2-bromomethyl-1,3,4-trimethoxynaphthalene

-
-
97-62-1
Ethyl isobutyrate

-
-
777075-05-5
ethyl 2,2-dimethyl-3-(1,3,4-trimethoxynaphthalen-2-yl)propanoate

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide | 100% |
| Stage #1: Ethyl isobutyrate With n-butyllithium; lithium diisopropyl amide In tetrahydrofuran; hexane at -80℃; Inert atmosphere; Stage #2: 3-bromomethyl-1,2,4-trimethoxynaphthalene In tetrahydrofuran; hexane at -80 - 20℃; Inert atmosphere; | 5.5 g |
-
-
459-46-1
4-Fluorobenzyl bromide

-
-
97-62-1
Ethyl isobutyrate

-
-
676621-95-7
3-(4-Fluorophenyl)-2,2-dimethyl-propionic Acid Ethyl Ester

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 1h; Stage #2: 4-Fluorobenzyl bromide In tetrahydrofuran at -78 - 20℃; for 18h; | 100% |
| Stage #1: Ethyl isobutyrate With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 1h; Stage #2: 4-Fluorobenzyl bromide In tetrahydrofuran for 18.0833h; | 99% |
-
-
104-53-0
3-phenyl-propionaldehyde

-
-
97-62-1
Ethyl isobutyrate

-
-
133270-74-3, 133270-75-4, 134924-29-1
ethyl 3-hydroxy-2,2-dimethyl-5-phenylpentanoate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 3-phenyl-propionaldehyde In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: bromoundecane In tetrahydrofuran; hexane at 20℃; | 100% |
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: bromoundecane In tetrahydrofuran; hexane at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.166667h; Stage #2: 1-Bromo-2-butyne In tetrahydrofuran; hexane at -78℃; for 0.75h; | 99% |
| With 1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone; lithium diisopropyl amide In tetrahydrofuran at -10℃; | 95% |
| With lithium diisopropyl amide | 76% |
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.75h; Inert atmosphere; Stage #2: 1-Bromo-2-butyne In tetrahydrofuran; hexane at -78 - 20℃; for 0.4h; Inert atmosphere; | 61% |

-
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 2h; | 99% |
-
-
97-62-1
Ethyl isobutyrate

-
-
371154-03-9
1-iodo-2-(triethylsilyloxy)ethane

-
-
886220-98-0
2,2-dimethyl-4-triethylsilanyloxy-butyric acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.333333h; Stage #2: 1-iodo-2-(triethylsilyloxy)ethane In tetrahydrofuran; hexane cooling; | 99% |
| With lithium diisopropyl amide In tetrahydrofuran at -78 - 20℃; |
-
-
621-23-8
1,2,3-trimethoxybenzene

-
-
97-62-1
Ethyl isobutyrate

-
-
916916-55-7
1-(2-methylpropyl)-2,4,6-trimethoxybenzene

| Conditions | Yield |
|---|---|
| With dimethylethylsilane; (μ3,η2,η3,η5-acenaphthylene)Ru3(CO)7 at 20℃; for 5h; Inert atmosphere; Neat (no solvent); | 99% |
-
-
140-29-4
phenylacetonitrile

-
-
97-62-1
Ethyl isobutyrate

-
-
32039-89-7
4-methyl-3-oxo-2-phenyl-pentanenitrile

| Conditions | Yield |
|---|---|
| With potassium 2-methylbutan-2-olate In tetrahydrofuran at 60℃; for 1h; | 98.4% |
| Stage #1: Ethyl isobutyrate With potassium tert-butylate In tetrahydrofuran; water Stage #2: phenylacetonitrile In tetrahydrofuran; water at 20℃; for 0.5h; | 75% |
| Stage #1: phenylacetonitrile With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; Stage #2: Ethyl isobutyrate In tetrahydrofuran; mineral oil at 60℃; for 2h; Stage #3: With hydrogenchloride In tetrahydrofuran; water; mineral oil | 19% |
| With diethyl ether; ethanol; potassium ethoxide | |
| With lithium hexamethyldisilazane In tetrahydrofuran at 0 - 29℃; for 1h; Inert atmosphere; Schlenk technique; |
-
-
100-52-7
benzaldehyde

-
-
97-62-1
Ethyl isobutyrate

-
-
52178-65-1, 133270-73-2, 35022-34-5
ethyl 3-hydroxy-2,2-dimethyl-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: benzaldehyde In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; | 98% |
| With n-butyllithium; diisopropylamine THF, -78 deg C; 1.) 50 min, 2.) 5 min; Yield given. Multistep reaction; |
-
-
25597-16-4
ethyl 4,4,4-trifluorocrotonate

-
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; hexane 1.) -78 deg C, 30 min, 2.) -78 deg C, 1.0 h; | 98% |
-
-
49669-22-9
6,6'-Dibromo-2,2'-bipyridine

-
-
97-62-1
Ethyl isobutyrate

-
-
455317-76-7
6,6'-bis(1-ethoxycarbonyl-1-methylethyl)-[2,2']bipyridinyl

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.666667h; Stage #2: 6,6'-Dibromo-2,2'-bipyridine In tetrahydrofuran; hexane at -78℃; for 0.166667h; Irradiation; | 98% |
-
-
623-00-7
4-bromobenzenecarbonitrile

-
-
97-62-1
Ethyl isobutyrate

-
-
1273587-21-5
ethyl 2-(4-cyanophenyl)-2-methylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With zinc chloride-2,2,6,6-tetramethylpiperidin-1-ide lithium chloride complex In tetrahydrofuran at 25℃; Inert atmosphere; Stage #2: 4-bromobenzenecarbonitrile With palladium diacetate In tetrahydrofuran at 50℃; for 9h; Inert atmosphere; | 98% |
-
-
51608-60-7
N-(benzylidene)-p-methylbenzenesulfonamide

-
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: N-(benzylidene)-p-methylbenzenesulfonamide In tetrahydrofuran; hexane for 2h; | 98% |
-
-
41423-29-4
4-iodo-1-butynyltrimethylsilane

-
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78 - 0℃; for 0.5h; Inert atmosphere; Stage #2: 4-iodo-1-butynyltrimethylsilane In tetrahydrofuran; hexane at 20℃; Inert atmosphere; | 98% |
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78 - 0℃; for 1.16667h; Corey-Fuchs Alkyne Synthesis; Inert atmosphere; Stage #2: 4-iodo-1-butynyltrimethylsilane In tetrahydrofuran; hexane at -78 - 20℃; for 2h; Inert atmosphere; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran at -20℃; for 0.916667h; Inert atmosphere; Stage #2: 2-bromo-3-(bromomethyl)benzonitrile In tetrahydrofuran at -20℃; for 0.75h; | 97.5% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.833333h; Stage #2: 2-methylpropenal In tetrahydrofuran; hexane at -78℃; for 0.166667h; Further stages.; | 97% |
-
-
97-62-1
Ethyl isobutyrate

-
-
4392-24-9
3-bromo-1-phenyl-1-propenyl bromide

-
-
72524-20-0
(E)-ethyl 2,2-dimethyl-5-phenylpent-4-enoate

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran at -5℃; for 0.5h; Inert atmosphere; Stage #2: 3-phenylprop-2-en-1-yl bromide In tetrahydrofuran at -50 - 20℃; for 18h; Inert atmosphere; | 97% |
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -50 - -5℃; for 1h; Inert atmosphere; Stage #2: 3-phenylprop-2-en-1-yl bromide In tetrahydrofuran; hexane at -50 - 20℃; for 10h; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With iron(III)-acetylacetonate In n-heptane at 105℃; for 11h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: N-(4-methoxybenzylidene)-p-toluenesulfonamide In tetrahydrofuran; hexane for 2h; | 97% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
97-62-1
Ethyl isobutyrate

-
-
31469-16-6
1-ethoxy-1-((trimethylsilyl)oxy)-2-methylpropene

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.333333h; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at 20℃; | 96% |
| With lithium diisopropyl amide In tetrahydrofuran | 95% |
| Stage #1: Ethyl isobutyrate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: chloro-trimethyl-silane With 1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone In tetrahydrofuran; hexane at -78 - 20℃; for 12.5h; Inert atmosphere; | 83% |
-
-
80542-40-1
N-cinnamylidene-p-anisidine

-
-
97-62-1
Ethyl isobutyrate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran for 12h; Ambient temperature; | 96% |
-
-
146726-49-0
(4S,5S)-4,5-dimethoxymethyl-2-(N-p-methoxyphenyl)iminomethyl-2-methyl-1,3-dioxolane

-
-
97-62-1
Ethyl isobutyrate

-
-
139308-01-3
(4S)-4-<(4S,5S)-4,5-dimethoxymethyl-2-methyl-1,3-dioxolan-2-yl>-1-p-methoxyphenyl-3,3-dimethyl-2-azetidinone

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide Mechanism; Product distribution; multistep reaction; other metal ester enolates; enantioselectivity; | 96% |
| With lithium diisopropyl amide 1.) DME, -78 deg C, 15 min, 2.) DME, r.t, 12 h; Yield given. Multistep reaction; |
-
-
102429-07-2
2-bromo-2',4'-difluoroacetophenone

-
-
97-62-1
Ethyl isobutyrate

-
-
145793-68-6
3-(2,4-difluorophenyl)-3,4-epoxy-2,2-dimethylbutanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With n-butyllithium; ammonium chloride; sodium hydrogencarbonate; lithium diisopropyl amide In tetrahydrofuran; hexane; water | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: o-chloro-N-tosylbenzaldimine In tetrahydrofuran; hexane for 2h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: Ethyl isobutyrate With lithium diisopropyl amide In tetrahydrofuran; n-heptane; ethylbenzene at -78℃; Stage #2: (Z,Z,Z)-1-Bromooctadeca-9,12,15-triene In tetrahydrofuran; n-heptane; ethylbenzene at -78 - 20℃; Inert atmosphere; | 96% |
Ethyl isobutyrate Chemical Properties
Empirical Formula: C6H12O2
Molecular Weight: 116.1583
EINECS: 202-595-4
Index of Refraction: 1.395
Density: 0.883 g/cm3
Flash Point: 19.4 °C
Enthalpy of Vaporization: 33.67 kJ/mol
Boiling Point: 112.6 °C at 760 mmHg
Vapour Pressure: 21.6 mmHg at 25 °C
Melting point: -88 °C
Storage temp: Flammables area
Water solubility: Insoluble
Appearance: Colorless volatile liquid
Solubility: Slightly soluble in water, energy and most organic solvents miscible
Product categories of 6-Nitroisatoic anhydride (CAS NO.4693-02-1): Organics;Carbonyl Compounds;Esters;Alphabetical Listings;Flavors and Fragrances
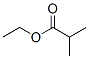
Structure of Ethyl isobutyrate (CAS NO.97-62-1):
Ethyl isobutyrate Uses
Ethyl isobutyrate (CAS NO.97-62-1) has been used as a food flavor ingredient and also be used as incense raw material in cigarettes, daily chemical products or other products. It is also an excellent organic solvent.
Ethyl isobutyrate Production
Ethyl isobutyrate (CAS NO.97-62-1) is derived by the different butyric acid and Ethanol in the azeotropic distillation under the condition of heat after esterification.
Ethyl isobutyrate Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MOD | FCTXAV Food and Cosmetics Toxicology. 16 (1978),741. | ||
| 2. | ipr-mus LD50:800 mg/kg | FCTXAV Food and Cosmetics Toxicology. 16 (1978),741. |
Ethyl isobutyrate Consensus Reports
Reported in EPA TSCA Inventory.
Ethyl isobutyrate Safety Profile
Moderately toxic by intraperitoneal route. A skin irritant. Flammable liquid. A very dangerous fire hazard when exposed to heat or flame; can react vigorously with oxidizing materials. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. See also ESTERS.
Hazard Codes:  F
F Xi
Xi
Risk Statements: 11-36/37/38
R11:Highly flammable.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 16-26-36-36/37/39
S16:Keep away from sources of ignition.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
Ethyl isobutyrate Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
Ethyl isobutyrate Specification
Ethyl isobutyrate ,its cas register number is 97-62-1. It also can be called Ethyl-2-methylpropanoat ; Propanoic acid, 2-methyl-, ethyl ester and 2-Méthylpropanoate d'éthyle . It is hazardous,so the first aid measures and others should be known. Such as: When on the skin: first,should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly,Get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While ,it's Inhaled: Remove from exposure and move to fresh air immediately.Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product : Wash mouth out with water,and get medical aid immediately.
In addition, Ethyl isobutyrate (CAS NO.97-62-1) can be stable under normal temperature and pressure conditions.It is not compatible with strong oxidizing agents, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: carbon dioxide, carbon monoxide.
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- ethyl (1R,2R)-1-phenyl-2-(trideuteriomethylamino)cyclohex-3-ene-1-carboxylate,hydrochloride
- Ethyl (1S,2R)-2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate hydrochloride
- Ethyl (2,4,6-trimethylbenzoyl) phenylphosphinate
- Ethyl (2-amino-4-hydroxy-6-methyl-5-pyrimidinyl)acetate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- ETHYL (2E,4Z)-DECADIENOATE
- Ethyl (2-hydroxyethyl)dimethyl-ammonium benzilate chloride
- Ethyl (2-mercaptoethyl) carbamate S-ester with O,O-dimethyl phosphorodithioate
- 97628-91-6
- 97-63-2
- 97632-67-2
- 97633-99-3
- 97635-27-3
- 97635-28-4
- 97635-35-3
- 97635-38-6
- 97635-41-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View