-
Name
HEXABUTYLDITIN
- EINECS 212-383-3
- CAS No. 813-19-4
- Article Data120
- CAS DataBase
- Density 1.148 g/mL
- Solubility insoluble in water
- Melting Point
- Formula C24H54Sn2
- Boiling Point 471.8 °C at 760 mmHg
- Molecular Weight 580.113
- Flash Point 240.1 °C
- Transport Information UN 2788 6
- Appearance Clear colorless to slightly yellow liquid
- Safety 35-36/37/39-45-60-61
- Risk Codes 21-25-36/38-48/23/25-50/53
-
Molecular Structure
-
Hazard Symbols
 T,
T, N
N
- Synonyms Hexabutylditin;NSC 227367;NSC 65520;NSC 92633;Distannane,hexabutyl- (8CI,9CI);Tin, hexabutyldi- (6CI,7CI);1,1,1,2,2,2-Hexabutyldistannane;Bis(tributyltin);Hexa-n-butyldistannane;Hexa-n-butylditin;
- PSA 0.00000
- LogP 9.76300
Synthetic route
-
-
4656-04-6
bis(trimethylsilyl)mercury

-
-
1067-52-3
tributyltin methoxide

-
A
-
1825-61-2
Methoxytrimethylsilane

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In benzene 25°C; 0.1 h; | A 95% B 98% C 96% |


| Conditions | Yield |
|---|---|
| With t-C4H9OH at 110°C (200 h); | 98% |
| In not given reaction for 200 h at 110°C;; | |
| In not given reaction for 200 h at 110°C;; |
-
-
111-83-1
1-bromo-octane

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
4111-54-0
lithium diisopropyl amide

-
A
-
111-65-9
octane

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In hexane; cyclohexane to 1-bromooctane (1 mmol) and Bu3SnH (1 mmol) in hexane at 0°C added LDA (1 mmol, in cyclohexane) under Ar, stirred for 20 min, quenched; analyzed by GLPC; | A 97% B 0% |

-
-
4656-04-6
bis(trimethylsilyl)mercury

-
-
56-35-9
bis(tri-n-butyltin)oxide

-
A
-
813-19-4
bis(tri-n-butyltin)

-
B
-
17908-18-8
Bu3SnOSiMe3

| Conditions | Yield |
|---|---|
| In hexane byproducts: Hg; 2:1; 25°C; 0.2 h; | A n/a B 97% |
| In benzene byproducts: Hg; 2:1; 25°C; 0.2 h; | A n/a B 97% |
| In benzene byproducts: Hg; 2:1; 25°C; 0.2 h; | A n/a B 97% |
| In hexane byproducts: Hg; 2:1; 25°C; 0.2 h; | A n/a B 97% |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: LiCl, Ti(Sn(C4H9)3)1.7; addn. of Bu3SnLi to Ti-compound in soln. (-78°C), stirred, mixt. brought to 20°C, soln. became blue; evapn. in vac., residue washed with pentane, sublimation (10**-3 Torr, 90°C); elem. anal., chromy.; | A 90% B 0% C 30% D 96% |

-
-
1175712-18-1, 1175712-19-2
N-[(1R)-2-hydroxy-1-phenylethyl]-N-[(1S)-1-(tributylstannyl)but-3-en-1-yl]benzenesulfonamide

-
A
-
56613-80-0
(R)-Phenylglycinol

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydrogensulfate In acetonitrile Inert atmosphere; Electrolysis; | A 96% B n/a |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
4111-54-0
lithium diisopropyl amide

-
A
-
1333-74-0
hydrogen

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In diethyl ether; cyclohexane byproducts: i-Pr2NH; Bu3SnH in Et2O cooled to 0°C under Ar, LDA in cyclohexane (1.6 M) added via syringe, stirred for 60 min at room temp., hydrolysis; | A 90% B 94% |


| Conditions | Yield |
|---|---|
| ((tBuC5H4)2Y(μ-H))2 In benzene byproducts: H2; (Ar); stirring (70°C, 30 min); | 91% |
| In benzene Ar-atmosphere; stirring (70°C, 0.5 h); | 91% |
| [(κ2-P,N)-3-(N,N-dimethylamino)propyldiphenylphosphino]dimethylplatinum(II) In not given byproducts: H2; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Electrolysis; electrolysis carried out galvanostatically at 25-28°C in an atm.of dry N2, electrolyte 0.4 M LiClO4/THF; hexane added to electrolyte, ppt. filtered off, filtrate analyzed for product by GC; | 90% |
| With samarium; samarium diiodide In tetrahydrofuran chlorostannane added to soln. of SmI2 in THF (cat. SmI2/Sm-mixed system); mixt. stirred for 24 h at room temp.; mixt.poured into 1.5N hydrochloric acid; extracted (diethyl ether); washed two times with aq. NaHCO3 then aq. NaCl; dried over MgSO4; filtrated; evapd.; purified by recycling HPLC wit CHCl3; | 85% |
| With magnesium; samarium diiodide In tetrahydrofuran chlorostannane added to soln. of SmI2 in THF (cat. SmI2/Mg-mixed system); mixt. stirred for 24 h at room temp.;mixt. poured into 1.5N hydrocloric acid; extracted (diethyl ether); washed two times with aq. NaHCO3 then aq.NaCl; dried over MgSO4; filtrated; evapd.; purified by recycling HPLC with CHCl3; | 82% |

| Conditions | Yield |
|---|---|
| In xylene starting material in soln. heated at 150°C for 6h; | A n/a B 90% |
-
-
14444-77-0
diethyl phenyl orthoformate

-
-
1068-55-9
isopropylmagnesium chloride

-
-
688-73-3
tri-n-butyl-tin hydride

-
A
-
813-19-4
bis(tri-n-butyltin)

-
B
-
1461-25-2
tetra-n-butyltin(IV)

-
C
-
79411-58-8
diethoxymethyltributylstannane

| Conditions | Yield |
|---|---|
| In diethyl ether grignard reagent in Et2O adding to Sn hydride soln. in Et2O, resulting Bu3SnMgCl adding dropwise to orthoformate in Et2O, stirring for 3 h, hydrolyzing, org. phase sepg.; purified by distn. in vacuo; NMR, IR and mass spectra; | A n/a B <1 C 90% |

-
-
4226-01-1
tri-n-butyltin lithium

-
-
17022-82-1
titanium dichloride bis(tert-butoxide)

-
B
-
131551-96-7
Ti(OC(CH3)3)2(Sn(C4H9)3)2

-
D
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; hexane Sn-compound in THF added to Ti-compound (-78°C) in hexane, temp. raised to room temp., soln. became dark-brown; solvent evapd. in vac., residue dissolved in pentane, filtered, pentane evapd. under vac., distd.; elem. anal.; | A 80% B 0% C 37% D 75% E 90% |

-
-
16853-85-3
lithium aluminium tetrahydride

-
-
1461-22-9
tributyltin chloride

-
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| dichloro bis(acetonitrile) palladium(II) In diethyl ether; water treatment stannic chloride with LiAlH4 (Et2O, 20°C, 4 h), addn. of water, addn. of catalyst, treatment (15 min); | 89% |
| dichloro bis(acetonitrile) palladium(II) In diethyl ether addn. of Bu3SnCl in ether to soln. of LiAlH4 in ether (stirring), stirring (20°C, 4 h), addn. of water, addn. of catalyst to intermediate (15 min); sepn. of org. layer (decantation), washing of residue (ether), drying of extract (over Na2SO4), removal of ether (vac.), extn. (pentane), chromy. (silica thin layer), removal of pentane, distn.; | 89% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) molar ratio Cp2V/Sn-compound 1/2, sealed ampoule, 120°C, 16 h; elem. anal.; | A 89% B 10.07% C 58.5% |

-
-
4226-01-1
tri-n-butyltin lithium

-
-
5915-86-6
chloro(tristertbutoxy)titanium

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran soln. of Sn-compound added to soln. of Ti-compound at -78°C, temp. increased to 20°C, mixt. became dark-brown; solvent evapd. in vac., residue dissolved in pentane, distd.; | A 45% B 88% |


| Conditions | Yield |
|---|---|
| In benzene byproducts: (CH3)3SiOOCH3, Hg; at 25°C, 0.5 h; | 87% |
| In benzene byproducts: (CH3)3SiOOCH3, Hg; at 25°C, 0.5 h; | 87% |

| Conditions | Yield |
|---|---|
| In hexane a mixture of the Sn compd. and the V compd. in hexane was held in an evacuated ampul for 48 h at 20°C; pptn., react. soln. contained 77% of (Sn(C4H9)3)2; elem. anal.; | A 87% B n/a |
| Ar, 20°C, 48 h.; Elem. anal.; | A 87% B 77% |


| Conditions | Yield |
|---|---|
| In toluene mixt. heated in an evacuated sealed ampul at 150-180°C for 15h; | A 80% B 87% |

-
-
4656-04-6
bis(trimethylsilyl)mercury

-
-
17908-18-8
Bu3SnOSiMe3

-
A
-
107-46-0
Hexamethyldisiloxane

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In benzene 80°C; 16 h; | A 72% B 76% C 83.5% |
| In benzene 80°C; 16 h; | A 72% B 76% C 83.5% |


| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In benzene stirring (2 h); H2O addn., extg. (CH2Cl2), drying (MgSO4), filtering, concg., distg. (vac.); | 83% |
| With potassium In toluene reaction with a stoich. amt. of potassium (2 mol for 1 mol of ditin oxide); | 71% |
| With titanium In tetrahydrofuran reaction with a freshly prepared titanium slurry; | 70% |

-
-
206437-65-2
(C4H9)3SnNHC6H4SO2NHSn(C4H9)3

-
A
-
206437-66-3
N-(η(5)-cyclopentadienylvanadio)-p-(η(5)-cyclopentadienylvanadioamino)benzenesulfonamide

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In toluene byproducts: cyclopentadiene; 120 to 130°C, 24-30 h, evacuated sealed ampule; black ppt. washing (toluene, vacuum), drying; elem. anal.; | A 74% B 82% |


| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran a soln. of ditin oxide in dry THF was added dropwise to a suspn. of a magnesium powder, activated by BrCH2CH2Br or I2, in dry THF, mixt. was heated at reflux for 15 h; petroleum ether was added, filtered, solvents evapd., residue was distilled; | A 81% B 0% |
| With sodium In neat (no solvent) stoich. amt. of sodium (2 mol for 1 mol of ditin oxide), 120°C, 48 h; unreacted Bu6Sn2O was removed by filtration in pentane through a short SiO2 column, Bu4Sn was removed by distn.; | A 71% B 1-5 |
-
-
108-86-1
bromobenzene

-
-
1461-22-9
tributyltin chloride

-
A
-
813-19-4
bis(tri-n-butyltin)

-
B
-
960-16-7
tributylphenylstannane

| Conditions | Yield |
|---|---|
| With magnesium; ethylene dibromide In tetrahydrofuran at 35℃; for 1h; Barbier Coupling Reaction; Sonication; Inert atmosphere; | A n/a B 78% |

-
-
5847-51-8
tributyltin formate

-
A
-
688-73-3
tri-n-butyl-tin hydride

-
B
-
813-19-4
bis(tri-n-butyltin)

-
C
-
1461-25-2
tetra-n-butyltin(IV)

| Conditions | Yield |
|---|---|
| heating 11.5 h at 150 to 185°C; | A 6.9% B 77.5% C 5.2% |
| heating 11.5 h at 150 to 185°C; | A 6.9% B 77.5% C 5.2% |
| In neat (no solvent) byproducts: CO2; heating for 11.5 h at 150-185°C under N2;; | A 6.9% B n/a C n/a |

-
-
109-69-3
n-Butyl chloride

-
-
1461-22-9
tributyltin chloride

-
A
-
813-19-4
bis(tri-n-butyltin)

-
B
-
1461-25-2
tetra-n-butyltin(IV)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Electrolysis; electrolyte 0.4 M LiClO4 in THF; electrolysis carried out galvanostatically at 25-28°C in atm. of dry N2; hexane added to electrolyte, ppt. filtered off, filtrate analyzed for the products by GC; | A 20% B 76% |

-
-
111-66-0
oct-1-ene

-
-
688-73-3
tri-n-butyl-tin hydride

-
A
-
813-19-4
bis(tri-n-butyltin)

-
B
-
16216-27-6
n-octyltriethyltin

| Conditions | Yield |
|---|---|
| dimeric bis(tert-butylcyclopentadienyl)(μ-methyl) yttrium In benzene (Ar); stirring (room temp., 30 min); elem. anal.; | A n/a B 74% |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: (C5H5V(OOCCH3)2)2, C5H5; in evacuated ampule, at 120 ° C, 16 h, without solvent; | 72% |
| In neat (no solvent) byproducts: (C5H5V(OOCCH3)2)2, C5H5; in evacuated ampule, at 120 ° C, 16 h, without solvent; | 72% |

-
-
5035-65-4
tributylstannyl isothiocyanate

-
B
-
1461-22-9
tributyltin chloride

-
C
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In toluene molar ratio V-complex/Bu3SnNCS=1:2, evacuated sealed ampul (120°C, 2 h); collection (filtn.), washing (toluene), drying (vac.); elem. anal.; | A 71% B 65% C 11% |


-
-
153627-98-6
tributylstannyl 4-(tributylstannylamino)benzenesulfonate

-
A
-
206437-68-5
η(5)-cyclopentadienevanadio-p-(η(5)-cyclopentadienylvanadioamino)benzenesulfonate

-
B
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In toluene byproducts: cyclopentadiene; 160 to 170°C, 48 h, evacuated sealed ampule, double excess of V-compd.; black ppt. washing (hot THF), drying; elem. anal.; | A 70% B 60% |


| Conditions | Yield |
|---|---|
| In hexane; N,N-dimethyl acetamide Electrolysis; hexane layer was placed for extn. of product; concn. of hexane layer in vac., column chromy. (silica gel, hexane); | 68% |
| In N,N-dimethyl acetamide Electrolysis; | 68% |

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane for 3.5h; Heating / reflux; | 100% |

| Conditions | Yield |
|---|---|
| In not given at 60°C; | 100% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; tricyclohexylphosphine In neat (no solvent) at 110℃; under 760.051 Torr; for 24h; Sealed tube; | 99% |
| With dichloro bis(acetonitrile) palladium(II) In N,N,N,N,N,N-hexamethylphosphoric triamide at 20℃; for 2.33333h; Product distribution; other reagents, reaction time, reactants' ratio dependence; also with other R4Sn (R = Et); | 92 % Chromat. |

| Conditions | Yield |
|---|---|
| Stage #1: bis(tri-n-butyltin) With naphthalene; lithium In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran at 20℃; for 1h; Reagent/catalyst; Inert atmosphere; Schlenk technique; | 99% |
| With dichloro bis(acetonitrile) palladium(II) In N,N,N,N,N,N-hexamethylphosphoric triamide at 20℃; for 0.0833333h; Product distribution; | 98 % Chromat. |

-
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| In benzene 20°C, 1.5 h; | 99% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; tricyclohexylphosphine In neat (no solvent) at 110℃; under 760.051 Torr; for 24h; Sealed tube; | 99% |
| tetrakis(triphenylphosphine) palladium(0) In toluene byproducts: Bu3SnBr, PhPh; heating at 80°C with stirring under Ar atm., reaction completed when color of the soln. changed to black; solvent evapd., biaryl filtered off after recrystn., filtrate washed with aq. KF, organic layer extd. with benzene, dried over sodium sulfate, product chromd.; | 89% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tricyclohexylphosphine In 1,4-dioxane at 110℃; for 3h; Reagent/catalyst; Inert atmosphere; | 99% |
-
-
764-01-2
methyl propargyl alcohol

-
-
813-19-4
bis(tri-n-butyltin)

-
-
121882-41-5, 124607-17-6, 850354-34-6
(2E)-3-methyl-3-tributylstannylprop-2-en-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: bis(tri-n-butyltin) With n-butyllithium; copper(l) cyanide In tetrahydrofuran; hexane at -78 - -40℃; for 0.583333h; Inert atmosphere; Stage #2: methyl propargyl alcohol In tetrahydrofuran; methanol; hexane at -78 - -10℃; Inert atmosphere; | 99% |
| With n-butyllithium; copper(l) cyanide In tetrahydrofuran; methanol at -78 - 20℃; | 99% |
-
-
813-19-4
bis(tri-n-butyltin)

-
-
15898-43-8
sodium 4-acetamidobenzenesulfinate

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; Inert atmosphere; | 99% |

-
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| With lithium chloride; bis(dibenzylideneacetone)-palladium(0); tri tert-butylphosphoniumtetrafluoroborate In 1,4-dioxane at 50℃; for 4h; Inert atmosphere; | 99% |
-
-
764-01-2
methyl propargyl alcohol

-
-
813-19-4
bis(tri-n-butyltin)

-
-
850354-34-6, 121882-41-5, 124607-17-6
3-(tributylstannyl)but-2-en-1-ol

| Conditions | Yield |
|---|---|
| With n-butyllithium; copper(l) cyanide In tetrahydrofuran at -10℃; Inert atmosphere; regioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); N-ethyl-N,N-diisopropylamine In isopropyl alcohol at 23℃; | 99% |

| Conditions | Yield |
|---|---|
| With methyl iodide; dichloro bis(acetonitrile) palladium(II) In N,N,N,N,N,N-hexamethylphosphoric triamide 20°C, molar ratio distannane/halide 1.3:1, 5 min; | A n/a B 98% |
-
-
609-73-4
o-nitroiodobenzene

-
-
813-19-4
bis(tri-n-butyltin)

-
-
79062-30-9
2-(tri-n-butylstannyl)-1-nitrobenzene

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In toluene byproducts: (C4H9)3SnI; soln. of Bu3SnSnBu3, Pd(PPh3)4, aryl iodide in toluene heated to 80°C for 72 h under Ar; evapd., washed with aq. KF, extd. (C6H6), dried over Na2SO4, chromd. (SiO2, hexane); elem. anal.; | 98% |
| With palladium diacetate; tricyclohexylphosphine In neat (no solvent) at 110℃; under 760.051 Torr; for 24h; Sealed tube; | 67% |
-
-
813-19-4
bis(tri-n-butyltin)

-
-
15946-37-9
sodium 3-chlorobenzenesulfinate

-
-
24344-58-9
tributyl(3-chlorophenyl)stannane

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; Inert atmosphere; | 98% |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
72170-87-7
(+)-(2S)-N-[cyclohex-2-en-1-yliden]-2-(methoxymethyl)pyrrolidin-1-amine

-
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| With NH4Cl In tetrahydrofuran addn. of the distannane to n-BuLi in abs. THF, react. for 15 min, cooling to -100°C, warming within 3 h to -78°C, dropwise addn. of the pyrrolidine compd. and addn. of satd. NH4Cl; evapn., dissolving in Et2O, washing with H2O and satd. NaCl soln., drying (MgSO4), concn. and column chromy. (silica gel, petroleum ether-Et2O); de 42 %; | 97% |


| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tricyclohexylphosphine In 1,4-dioxane at 110℃; for 3h; Reagent/catalyst; Inert atmosphere; | 97% |
-
-
106-38-7
para-bromotoluene

-
-
813-19-4
bis(tri-n-butyltin)

-
A
-
1461-23-0
tributyltin bromide

-
B
-
31614-66-1
tributyl(p-tolyl)stannane

| Conditions | Yield |
|---|---|
| With palladium diacetate; tricyclohexylphosphine In neat (no solvent) at 110℃; under 760.051 Torr; for 24h; Sealed tube; | A n/a B 97% |


| Conditions | Yield |
|---|---|
| With bis(tri-tert-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; Temperature; | 97% |
| With bis(tri-t-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; Solvent; Temperature; Reagent/catalyst; Inert atmosphere; | 97% |
-
-
813-19-4
bis(tri-n-butyltin)

-
-
824-79-3
sodium 4-methylbenzenesulfinate

-
-
31614-66-1
tributyl(p-tolyl)stannane

| Conditions | Yield |
|---|---|
| With bis(tri-tert-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; | 97% |
| With bis(tri-t-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; Inert atmosphere; | 97% |
-
-
591-50-4
iodobenzene

-
-
813-19-4
bis(tri-n-butyltin)

-
A
-
92-52-4
biphenyl

-
B
-
960-16-7
tributylphenylstannane

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In toluene byproducts: Bu3SnI; at 115°C for 15 h in sealed thick wall Pyrex glass ampoules; | A 3.5% B 96% |
| bis(triphenylphosphine)palladium dibromide In toluene byproducts: Bu3SnI; at 115°C for 15 h in sealed thick wall Pyrex glass ampoules; | A 2% B 94% |

-
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In toluene for 96h; Reflux; Darkness; Inert atmosphere; | 96% |
-
-
813-19-4
bis(tri-n-butyltin)

-
-
824-80-6
sodium 4-fluorobenzenesulfinate

-
-
17151-47-2
tributyl(4-fluorophenyl)stannane

| Conditions | Yield |
|---|---|
| With bis(tri-tert-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; | 96% |
| With bis(tri-t-butylphosphine)palladium(0); silver carbonate In N,N-dimethyl acetamide at 140℃; for 1h; Inert atmosphere; | 96% |

-
-
813-19-4
bis(tri-n-butyltin)

| Conditions | Yield |
|---|---|
| With lithium chloride; bis(dibenzylideneacetone)-palladium(0); tri tert-butylphosphoniumtetrafluoroborate In 1,4-dioxane at 50℃; for 4h; Inert atmosphere; | 96% |
Hexabutyldistannane Specification
The Distannane,1,1,1,2,2,2-hexabutyl-, with the CAS registry number 813-19-4, is also known as Hexabutyldistannane. It belongs to the product categories of Classes of Metal Compounds; Sn (Tin) Compounds; Typical Metal Compounds. Its EINECS registry number is 212-383-3. This chemical's molecular formula is C24H54Sn2 and molecular weight is 580.11. What's more, both its IUPAC name and systematic name are the same which is called Tributyl-tributylstannyl-stannane.
Physical properties about Distannane,1,1,1,2,2,2-hexabutyl- are: (1)ACD/LogP: 16.70; (2)# of Rule of 5 Violations: 2; (3)ACD/LogD (pH 5.5): 16.7; (4)ACD/LogD (pH 7.4): 16.7; (5)#H bond acceptors: 0; (6)#H bond donors: 0; (7)#Freely Rotating Bonds: 19; (8)Polar Surface Area: Å2; (9)Flash Point: 240.1 °C; (10)Enthalpy of Vaporization: 70.65 kJ/mol; (11)Boiling Point: 471.8 °C at 760 mmHg; (12)Vapour Pressure: 1.29E-08 mmHg at 25 °C.
Preparation of Distannane,1,1,1,2,2,2-hexabutyl-: this chemical can be prepared by Anthracene-9,10-dicarbonitrile with Benzyl-tributyl-stannane. This reaction needs reagent benzene and other condition of irradiation. The yield is 28 %.
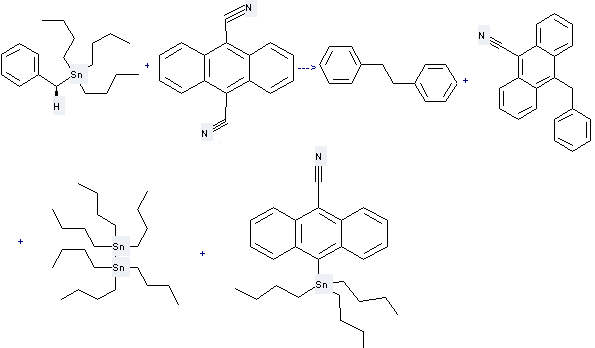
Uses of Distannane,1,1,1,2,2,2-hexabutyl-: it is used to produce other chemicals. For example, it can react with 4-Methylquinoline trifluoroacetate to get 2-Butyl-4-methylquinoline. The reaction occurs with reagent acetonitrile and other condition of irradiation. The yield is 30 %.

When you are dealing with this chemical, you should be very careful. This chemical may cause damage to health and present an immediate or delayed danger to one or more components of the environment. It is toxic by inhalation and if swallowed. What's more, this chemical is very toxic to aquatic organisms and it may cause long-term adverse effects in the aquatic environment. Therefore, you should obtain special instructions before use it and avoid releasing to the environment. In addition, you should dispose of this material and its container as hazardous waste. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
(1) SMILES: CCCC[Sn](CCCC)(CCCC)[Sn](CCCC)(CCCC)CCCC
(2) InChI: InChI=1/6C4H9.2Sn/c6*1-3-4-2;;/h6*1,3-4H2,2H3;;/rC24H54Sn2/c1-7-13-19-25(20-14-8-2,21-15-9-3)26(22-16-10-4,23-17-11-5)24-18-12-6/h7-24H2,1-6H3
(3) InChIKey: REDSKZBUUUQMSK-NRFXJKAKAK
Related Products
- Hexabutyldistannane
- 81320-19-6
- 81321-10-0
- 81321-14-4
- 81321-15-5
- 81321-37-1
- 813222-82-1
- 81322-67-0
- 81323-58-2
- 81325-81-7
- 81329-90-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View