-
Name
Isopentyl acetate
- EINECS 204-662-3
- CAS No. 123-92-2
- Article Data124
- CAS DataBase
- Density 0.879 g/cm3
- Solubility Slightly soluble in water: 0.20 g/100 mL
- Melting Point -78 °C
- Formula C7H14O2
- Boiling Point 142.1 °C at 760 mmHg
- Molecular Weight 130.187
- Flash Point 25 °C
- Transport Information UN 1104 3/PG 3
- Appearance colourless liquid with a smell of bananas
- Safety 23-25-2-36/37/39-26-16
- Risk Codes 10-66-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 1-Butanol,3-methyl-, acetate (9CI);Acetic acid, isoamyl ester (3CI);Isopentyl alcohol,acetate (6CI,8CI);3-Methyl-1-butanol acetate;3-Methylbutyl ethanoate;Acetic acid 3-methyl-1-butylester;Acetic acid 3-methylbutyl ester;Acetic acid isopentyl ester;Bananaoil;Isoamyl alcohol acetate;Isoamyl ethanoate;Isopentyl acetate;Isopentyl ethanoate;NSC 9260;Pear oil;i-Amyl acetate;iso-Amyl acetate;iso-Pentyl acetate;
- PSA 26.30000
- LogP 1.59560
Synthetic route

| Conditions | Yield |
|---|---|
| copper methanesulfonate In cyclohexane at 85 - 90℃; for 2.5h; | 99% |
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 98.1% |
| With H+ Amberlyst 15 at 120℃; for 1h; | 97% |

| Conditions | Yield |
|---|---|
| With pseudomonas fuorescens lipase immobilized on multiwall carbon nano-tubes at 50℃; for 4h; Green chemistry; | 99% |
| With Rasta resin-(1,5,7-triazabicyclo[4.4.0]dec-5-ene)[RR-TBD] In tetrahydrofuran at 20 - 60℃; | 91% |
| With steapsin lipase In hexane at 55℃; for 24h; Enzymatic reaction; | 99 %Chromat. |
| With candida rugosa lipase immobilized on nanostructured tin dioxide In decane at 50℃; for 72h; Kinetics; Temperature; | |
| With H-ZSM-5 zeolite: In toluene at 20 - 90℃; for 3h; Catalytic behavior; |

| Conditions | Yield |
|---|---|
| cerium triflate In acetonitrile at 20℃; for 0.5h; | 98% |
| With iron zirconium phosphate In neat (no solvent) at 40℃; for 0.166667h; Green chemistry; | 95% |
| With Cu(2+)*Zr(4+)*2PO4(3-) = CuZr(PO4)2 at 60℃; for 0.333333h; | 95% |

| Conditions | Yield |
|---|---|
| With lipolase In carbon dioxide at 40℃; under 150012 Torr; Rate constant; Product distribution; other enzyme (Novozym); lipase catalysed esterification of isoamyl alcohol by ammonium acetate in supercritical CO2 and by acetic acid in hexane; effect of reagent concentrations, temperature and pressure on product yield; | 96% |
| With lipolase In carbon dioxide at 40℃; under 150012 Torr; | 96% |

| Conditions | Yield |
|---|---|
| for 3h; Heating; | 85% |
| With candida rugosa lipase immobilized on nanostructured tin dioxide In decane at 50℃; for 72h; | |
| With recombinant acyltransferase from Mycobacterium smegmatis In aq. phosphate buffer at 25℃; for 0.5h; pH=8; Concentration; Green chemistry; Enzymatic reaction; | |
| With Octanoic acid; dihydrogen peroxide at 60℃; for 24h; Time; |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 30℃; for 0.916667h; Neat (no solvent); | 51% |
| With diethyl ether; magnesium | |
| With sodium hydride 1.) 2 h, reflux, 2.) 1 h, reflux; Multistep reaction; | |
| With coal fly ash supported phosphomolybdic acid at 120℃; Temperature; Microwave irradiation; Green chemistry; |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride |

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With boron trifluoride |
-
-
60-29-7
diethyl ether

-
-
79-20-9
acetic acid methyl ester

-
-
4548-78-1
(3-methylbutyl)magnesium bromide

-
-
123-92-2
3-methyl-1-butyl acetate

-
-
506-96-7
Acetyl bromide

-
-
628-04-6
1-ethoxy-3-methyl-butane

-
A
-
74-96-4
ethyl bromide

-
B
-
123-92-2
3-methyl-1-butyl acetate

-
C
-
141-78-6
ethyl acetate

-
D
-
107-82-4
i-pentyl bromide

| Conditions | Yield |
|---|---|
| at 200 - 210℃; |

-
-
544-01-4
isopentyl ether

-
-
507-02-8
acetyl iodide

-
A
-
541-28-6
isopentyl iodide

-
B
-
123-92-2
3-methyl-1-butyl acetate

| Conditions | Yield |
|---|---|
| at 25℃; unter Lichtausschluss; |


| Conditions | Yield |
|---|---|
| With silver containing copper MnO-catalyst at 270 - 280℃; | |
| With uranium(VI) trioxide; copper(II) oxide at 270℃; |

| Conditions | Yield |
|---|---|
| With acetylcoenzyme A at 25℃; for 1h; Product distribution; brewers' yeast (Saccharomyces uvarum), also further yeasts or Hansenula anomala, pH 7.5; | |
| Veresterung; das Produkt enthaelt in der Hauptsache Isoamylacetat und aktive Amylacetate, neben geringen Mengen von Acetaten einiger Alkohole mit 6 und 7 Kohlenstoffatomen und unbekannten hochsiedenden Verbindungen; |

| Conditions | Yield |
|---|---|
| tetraethylammonium chloride In acetonitrile at 60℃; Rate constant; | |
| In N,N-dimethyl-formamide; toluene at 60℃; Rate constant; Thermodynamic data; Ea; ΔH333, ΔS333; | |
| With acetic acid |
-
-
39782-38-2
3-methyl-1-vinyloxybutane

-
A
-
123-92-2
3-methyl-1-butyl acetate

-
B
-
100386-52-5
chloro-acetaldehyde diisopentylacetal

| Conditions | Yield |
|---|---|
| With water; chlorine |
-
-
17458-90-1
3-methylbuta-1,2-dien-1-yl acetate

-
-
123-92-2
3-methyl-1-butyl acetate

| Conditions | Yield |
|---|---|
| Hydrogenation; |
-
-
123-51-3
i-Amyl alcohol

-
-
75-07-0
acetaldehyde

-
-
25016-92-6
aluminium triisopentylate

-
-
123-92-2
3-methyl-1-butyl acetate


| Conditions | Yield |
|---|---|
| With potassium permanganate; oxygen unter Druck; | |
| With sulfuric acid; oxygen unter Druck; |
-
-
555-75-9
aluminum ethoxide

-
-
75-07-0
acetaldehyde

-
-
590-86-3
isovaleraldehyde

-
A
-
108-64-5
Ethyl isovalerate

-
B
-
123-92-2
3-methyl-1-butyl acetate


| Conditions | Yield |
|---|---|
| With aluminum ethoxide |
-
-
36595-99-0
benzenesulfonic acid isopentyl ester

-
-
64-19-7
acetic acid

-
-
123-92-2
3-methyl-1-butyl acetate

| Conditions | Yield |
|---|---|
| With Petroleum ether |
-
-
4548-78-1
(3-methylbutyl)magnesium bromide

-
-
141-78-6
ethyl acetate

-
A
-
64-17-5
ethanol

-
B
-
64029-94-3
methyl-diisopentyl-carbinol

-
C
-
123-92-2
3-methyl-1-butyl acetate

-
-
544-01-4
isopentyl ether

-
-
5435-44-9, 22243-66-9
(E)-3-Ureido-but-2-enoic acid ethyl ester

-
-
75-36-5
acetyl chloride

-
A
-
107-84-6
1-chloro-3-methylbutane

-
B
-
123-92-2
3-methyl-1-butyl acetate

-
C
-
563-45-1
3-Methyl-1-butene


| Conditions | Yield |
|---|---|
| With zinc(II) chloride bei Siedetemperatur; |

-
-
108-95-2
phenol

-
A
-
123-92-2
3-methyl-1-butyl acetate

-
B
-
5032-03-1
(3-methyl-butyl)-phenol

-
C
-
141-78-6
ethyl acetate

-
D
-
103-73-1
Phenetole

| Conditions | Yield |
|---|---|
| at 200℃; |

-
-
123-51-3
i-Amyl alcohol

-
-
7664-93-9
sulfuric acid

-
-
75-07-0
acetaldehyde

-
-
123-92-2
3-methyl-1-butyl acetate

| Conditions | Yield |
|---|---|
| unter Druck; |



| Conditions | Yield |
|---|---|
| analog entsteht mit Aethylenglykol dessen Monoacetat; |

-
-
123-92-2
3-methyl-1-butyl acetate

-
-
2269-22-9
aluminum tri-sec-butoxide

-
-
25016-92-6
aluminium triisopentylate

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: sec-Bu-OAc; N2 atmosphere; exchange reaction (111°C); distn. of butyl ester; | 90% |

| Conditions | Yield |
|---|---|
| With lipase PS IM Amano; triethylamine at 20℃; | 89% |
-
-
10026-04-7, 53609-55-5
tetrachlorosilane

-
-
123-92-2
3-methyl-1-butyl acetate

-
-
4607-64-1
tetrakis-(3-methyl-butoxy)-silane

| Conditions | Yield |
|---|---|
| 88% | |
| 88% |
-
-
123-92-2
3-methyl-1-butyl acetate

-
-
618-36-0
rac-methylbenzylamine

-
-
36065-27-7
N-α-phenylethylacetamide

| Conditions | Yield |
|---|---|
| With hydrogen; sodium carbonate; lipase B from Candida antarctica In toluene at 60℃; under 760.051 Torr; for 24h; Sealed tube; Enzymatic reaction; | 82% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 100℃; for 2.5h; Inert atmosphere; | 73% |
-
-
123-92-2
3-methyl-1-butyl acetate

| Conditions | Yield |
|---|---|
| With fluorine In chloroform; trichlorofluoromethane at -75℃; | 70% |

| Conditions | Yield |
|---|---|
| With 1-ethoxy-4-nitropyridinium tetrafluoroborate In acetone for 5h; Irradiation; | 67% |
| With N-hydroxyphthalimide; nitromethane; iodic acid In water at 80℃; for 12h; Inert atmosphere; Sealed tube; | 44% |
| With 4-nitroperbenzoic acid Rate constant; velocity constants relativ to 11 (OOCCH3=H); |

| Conditions | Yield |
|---|---|
| With 2,4,6-triphenylpyrylium tetrafluoroborate; 1,2-dibromomethane at 50℃; for 24h; Irradiation; | 67% |
-
-
123-92-2
3-methyl-1-butyl acetate

-
-
42070-98-4
1-(p-tolyl)ethylamine

-
-
92520-13-3
N-(1-(4-methylphenyl)ethyl)acetamide

| Conditions | Yield |
|---|---|
| With hydrogen; sodium carbonate; lipase B from Candida antarctica In toluene at 100℃; under 760.051 Torr; for 24h; Temperature; Sealed tube; Enzymatic reaction; | 66% |

| Conditions | Yield |
|---|---|
| With iodic acid In acetonitrile at 80℃; for 24h; Inert atmosphere; Sealed tube; | 49% |

| Conditions | Yield |
|---|---|
| at 750℃; under 0.007 Torr; | 15% |
| at 500℃; | |
| at 600℃; | |
| at 700℃; im Stickstoffstrom an Glaswolle; | |
| at 500℃; Pyrolysis; |

| Conditions | Yield |
|---|---|
| With bromomagnesium diethylamide |
-
-
681-84-5
tetramethylorthosilicate

-
-
123-92-2
3-methyl-1-butyl acetate

-
-
4607-64-1
tetrakis-(3-methyl-butoxy)-silane

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| at 240℃; |
-
-
36794-64-6
2,3,3-trimethylbutan-1-ol

-
-
123-92-2
3-methyl-1-butyl acetate

-
-
102439-06-5
acetic acid-(2,3,3-trimethyl-butyl ester)

| Conditions | Yield |
|---|---|
| With sulfuric acid |
Isoamyl acetate Consensus Reports
Reported in EPA TSCA Inventory.
Isoamyl acetate Standards and Recommendations
OSHA PEL: TWA 100 ppm
ACGIH TLV: TWA 150 ppm
DFG MAK: 50 ppm
Isoamyl acetate Analytical Methods
For occupational chemical analysis use NIOSH: Esters I, 1450.
Isoamyl acetate Specification
The Isoamyl acetate, with the CAS registry number 123-92-2,is also known as Albichthol; Albichtol; Albikhtol; Ammonium bithiolicum. It belongs to the product categories of Analytical Chemistry;Solvents for HPLC & Spectrophotometry. Its EINECS number is 204-662-3. This chemical's molecular formula is C7H14O2 and molecular weight is 130.18. What's more,Its systematic name is Isoamyl acetate. It is a Oily liquid which is highly flammable and insoluble in water.It is also used to test the effectiveness of respirators or gas masks because it has a strong smell which is generally not experienced as unpleasant and that can be detected at low concentrations, and has low toxicity.
Physical properties about Isoamyl acetate are:
(1)ACD/LogP: 2.158; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.16; (4)ACD/LogD (pH 7.4): 2.16; (5)ACD/BCF (pH 5.5): 25.70; (6)ACD/BCF (pH 7.4): 25.70; (7)ACD/KOC (pH 5.5): 355.50; (8)ACD/KOC (pH 7.4): 355.50; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 4; (12)Index of Refraction: 1.404; (13)Molar Refractivity: 36.212 cm3; (14)Molar Volume: 147.942 cm3; (15)Surface Tension: 25.5960006713867 dyne/cm; (16)Density: 0.88 g/cm3; (17)Flash Point: 25 °C; (18)Enthalpy of Vaporization: 37.926 kJ/mol; (19)Boiling Point: 142.099 °C at 760 mmHg; (20)Vapour Pressure: 5.67799997329712 mmHg at 25°C.
You can still convert the following datas into molecular structure:
(1)SMILES:O=C(OCCC(C)C)C;
(2)Std. InChI:InChI=1S/C7H14O2/c1-6(2)4-5-9-7(3)8/h6H,4-5H2,1-3H3;
(3)Std. InChIKey:MLFHJEHSLIIPHL-UHFFFAOYSA-N.
Production of Isoamyl acetate:
Isoamyl acetate (CAS NO.123-92-2) can be prepared in the laboratory by the acid catalyzed Fischer esterification reaction between isoamyl alcohol and glacial acetic acid as shown in the reaction equation below. Typically, concentrated sulfuric acid is used as the catalyst. The reactants are refluxed in order to promote the reaction, the acid is neutralized and extracted, and the product is dried, then distilled.
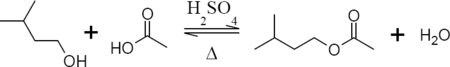
Safety Information of Isoamyl acetate:
The Isoamyl acetate is irritating to eyes, respiratory system and skin.Repeated exposure may cause skin dryness or cracking. So it should Keep out of the reach of children.And it is highly flammable, so it should be keep away from sources of ignition - No smoking .When you use it ,wear suitable protective clothing, gloves and eye/face protection,and do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer) ,and avoid contact with eyes.In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
The toxicity data of Isoamyl acetate are as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| cat | LCLo | inhalation | 35gm/m3 (35000mg/m3) | BEHAVIORAL: GENERAL ANESTHETIC BEHAVIORAL: FOOD INTAKE (ANIMAL) KIDNEY, URETER, AND BLADDER: PROTEINURIS | Archiv fuer Gewerbepathologie und Gewerbehygiene. Vol. 5, Pg. 1, 1933. |
| guinea pig | LDLo | subcutaneous | 5gm/kg (5000mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE BEHAVIORAL: GENERAL ANESTHETIC | Archiv fuer Gewerbepathologie und Gewerbehygiene. Vol. 5, Pg. 1, 1933. |
| rabbit | LD50 | oral | 7422mg/kg (7422mg/kg) | Industrial Medicine and Surgery. Vol. 41, Pg. 31, 1972. | |
| rabbit | LD50 | skin | > 5gm/kg (5000mg/kg) | National Technical Information Service. Vol. OTS0573010, | |
| rat | LD50 | oral | 16600mg/kg (16600mg/kg) | Yakkyoku. Pharmacy. Vol. 32, Pg. 1241, 1981. |
Related Products
- Isoamyl acetate
- Isoamyl benzoate
- Isoamyl butyrate
- Isoamyl butyrate
- Isoamyl carbamate
- Isoamyl cinnamate
- Isoamyl geranate
- Isoamyl iodide
- Isoamyl nitrite
- Isoamyl octanoate
- 1239262-52-2
- 12392-64-2
- 123927-75-3
- 1239-29-8
- 123931-04-4
- 1239-31-2
- 1239319-82-4
- 123932-43-4
- 123-93-3
- 123934-38-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View