-
Name
Formic acid isopropyl ester
- EINECS 210-901-2
- CAS No. 625-55-8
- Article Data33
- CAS DataBase
- Density 0.884 g/cm3
- Solubility slightly soluble in water, completely miscible with alcohol, ether, and most organic solvents.
- Melting Point -93 °C
- Formula C4H8O2
- Boiling Point 70.8 °C at 760 mmHg
- Molecular Weight 88.1063
- Flash Point 7°F
- Transport Information UN 1281
- Appearance Clear colorless liquid
- Safety 16-33-36
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Formicacid, isopropyl ester (6CI,7CI,8CI);Iso-propyl methanoate;Isopropyl formate;
- PSA 26.30000
- LogP 1.20370
Synthetic route

| Conditions | Yield |
|---|---|
| With phosphotungstic acid at 70℃; for 0.75h; | 95% |
| With trifluoromethanesulfonic acid based UiO-67 metal organic framework at 90℃; for 18h; | |
| at 120℃; under 22502.3 Torr; for 4h; Equilibrium constant; Temperature; Concentration; Autoclave; |

| Conditions | Yield |
|---|---|
| With phosphotungstic acid at 70℃; for 0.75h; | 95% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 12h; Ambient temperature; | 92% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 3h; Ambient temperature; | 90% |

| Conditions | Yield |
|---|---|
| With [bis({2‐[bis(propan‐2‐yl)phosphanyl]ethyl})amine](borohydride)(carbonyl)(hydride)iron(II); acetone In toluene at 140℃; under 13689.1 Torr; for 4h; Autoclave; Inert atmosphere; | 83% |
-
-
536-74-3
phenylacetylene

-
-
67-63-0
isopropyl alcohol

-
A
-
625-55-8
isopropyl formate

-
B
-
939-48-0
isopropyl benzoate

| Conditions | Yield |
|---|---|
| With bis-[(trifluoroacetoxy)iodo]benzene at 60℃; for 15h; Sealed tube; | A n/a B 67% |


| Conditions | Yield |
|---|---|
| With 1H-imidazole for 24h; Ambient temperature; | 62% |
| With 1H-imidazole Yield given; |

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; boron trifluoride at 100℃; |

| Conditions | Yield |
|---|---|
| at 200℃; |
-
-
24425-00-1
diisopropyl pyrocarbonate

-
-
625-55-8
isopropyl formate

| Conditions | Yield |
|---|---|
| With pyridine N-oxide In 1,2-dichloro-benzene |

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide at 130℃; Product distribution; Rate constant; Mechanism; other temperatures.; |
-
-
4447-60-3
Triisopropoxymethan

-
A
-
74-98-6
propane

-
B
-
625-55-8
isopropyl formate

-
C
-
6482-34-4
bis-2-propyl carbonate

-
D
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide In chlorobenzene at 140℃; for 4h; Kinetics; Mechanism; Product distribution; various temperatures, rates of formation; |


| Conditions | Yield |
|---|---|
| Thermodynamic data; Mechanism; calculated (BEBO and equibonding method) activation energies for hydrogen atom transfer reaction, anodic formylation; |
-
-
3397-76-0, 100508-13-2
N,N-Dimethylformamide Hydrochloride

-
-
67-63-0
isopropyl alcohol

-
A
-
75-29-6
isopropyl chloride

-
B
-
625-55-8
isopropyl formate

| Conditions | Yield |
|---|---|
| at 100℃; for 0.833333h; |


| Conditions | Yield |
|---|---|
| With air; methyl nitrite; nitrogen(II) oxide at 24.85℃; under 740 Torr; Product distribution; Irradiation; |

| Conditions | Yield |
|---|---|
| Geschwindigkeit; |

-
-
78-84-2
isobutyraldehyde

-
A
-
187737-37-7
propene

-
B
-
74-98-6
propane

-
C
-
625-55-8
isopropyl formate

-
D
-
67-63-0
isopropyl alcohol

-
E
-
67-64-1
acetone

-
F
-
79-31-2
isobutyric Acid

| Conditions | Yield |
|---|---|
| With oxygen; manganese(II) In propionic acid at 45℃; Product distribution; Mechanism; |

-
-
78-84-2
isobutyraldehyde

-
A
-
625-55-8
isopropyl formate

-
B
-
124-38-9
carbon dioxide

-
C
-
13122-72-0
2-methylperoxypropionic acid

-
D
-
67-63-0
isopropyl alcohol

-
E
-
67-64-1
acetone

-
F
-
79-31-2
isobutyric Acid

| Conditions | Yield |
|---|---|
| With oxygen at 10℃; for 2.66667h; Product distribution; Mechanism; other temperatures 20 and 30 deg C, various reaction time; | A 2.73 % Chromat. B n/a C 4.36 % Chromat. D 0.18 % Chromat. E 0.87 % Chromat. F 78.38 % Chromat. G n/a |

-
-
78448-33-6
1-isopropoxy-2-methyl-propane

-
A
-
1712-64-7
isopropyl nitrate

-
B
-
625-55-8
isopropyl formate

-
C
-
110-19-0
2-methylpropyl acetate

-
D
-
42125-48-4
acetic acid-(β-hydroxy-isobutyl ester)

-
E
-
78-84-2
isobutyraldehyde

-
F
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With air; methyl nitrite; nitric oxide Product distribution; Rate constant; Mechanism; Ambient temperature; Irradiation; OH radical initiated oxidation of title comp. under laboratory conditions related to troposphere; |

-
-
187737-37-7
propene

-
-
124-38-9
carbon dioxide

-
A
-
625-55-8
isopropyl formate

-
B
-
67-63-0
isopropyl alcohol

-
C
-
67-64-1
acetone

-
D
-
75-56-9, 16033-71-9
methyloxirane

| Conditions | Yield |
|---|---|
| With stainless steel; oxygen; isobutyraldehyde In carbon dioxide at 55℃; under 142514 Torr; for 18h; Epoxidation; Oxidation; Further byproducts.; |


| Conditions | Yield |
|---|---|
| Substitution; |

| Conditions | Yield |
|---|---|
| Conversion of starting material; |

| Conditions | Yield |
|---|---|
| With water; difluoro[4-(trifluoromethyl)phenyl]-λ3-bromane In dichloromethane at 0℃; for 1h; Baeyer-Villiger type oxidation; Inert atmosphere; | 81 %Chromat. |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; for 1h; |
-
-
124-38-9
carbon dioxide

-
-
67-63-0
isopropyl alcohol

-
A
-
2568-89-0
diisopropoxymethane

-
B
-
625-55-8
isopropyl formate

| Conditions | Yield |
|---|---|
| Stage #1: isopropyl alcohol With aluminium(III) triflate; [Ru(1,1,1-tris(diphenylphosphinomethyl)ethane)(trimethylene methane)] for 0.0833333h; Inert atmosphere; Schlenk technique; Stage #2: carbon dioxide With hydrogen at 80℃; under 60006 Torr; for 18h; Catalytic behavior; Schlenk technique; Autoclave; |

-
-
124-38-9
carbon dioxide

-
-
67-63-0
isopropyl alcohol

-
A
-
67-56-1
methanol

-
B
-
2568-89-0
diisopropoxymethane

-
C
-
625-55-8
isopropyl formate

| Conditions | Yield |
|---|---|
| With cobalt(II) tetrafluoroborate hexahydrate; hydrogen; bis(trifluoromethanesulfonyl)amide; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In tetrahydrofuran at 80℃; under 15001.5 - 60006 Torr; for 22h; Catalytic behavior; Schlenk technique; Autoclave; |


| Conditions | Yield |
|---|---|
| With cobalt(II) tetrafluoroborate hexahydrate; hydrogen; bis(trifluoromethanesulfonyl)amide; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In tetrahydrofuran at 100℃; under 15001.5 - 60006 Torr; for 22h; Catalytic behavior; Schlenk technique; Autoclave; |

-
-
201230-82-2
carbon monoxide

-
-
6831-82-9
potassium isopropoxide

-
-
67-63-0
isopropyl alcohol

-
-
625-55-8
isopropyl formate

| Conditions | Yield |
|---|---|
| at 30℃; under 78307.8 Torr; for 24h; Autoclave; Inert atmosphere; Large scale; | 26800 g |

-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

-
-
67-63-0
isopropyl alcohol

-
A
-
67-56-1
methanol

-
B
-
625-55-8
isopropyl formate

-
C
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With [bis({2‐[bis(propan‐2‐yl)phosphanyl]ethyl})amine](bromo)(carbonyl)(hydride)iron(II); potassium tert-butylate In tetrahydrofuran at 140℃; Inert atmosphere; Schlenk technique; Green chemistry; |

-
-
628-35-3
ethylene glycol monoformate

-
-
67-63-0
isopropyl alcohol

-
A
-
67-56-1
methanol

-
B
-
625-55-8
isopropyl formate

-
C
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| With [bis({2‐[bis(propan‐2‐yl)phosphanyl]ethyl})amine](bromo)(carbonyl)(hydride)iron(II); potassium tert-butylate In tetrahydrofuran at 140℃; for 3h; Inert atmosphere; Schlenk technique; Green chemistry; | A 62 %Chromat. B 18 %Chromat. C 85 %Chromat. |


| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 10h; | 94% |


| Conditions | Yield |
|---|---|
| With iron (III) perchlorate monohydrate In neat (no solvent) at 80℃; for 5h; Ritter Amidation; | 90% |

| Conditions | Yield |
|---|---|
| With (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; 4-cyano-1-isopropoxypyridin-1-ium trifluoromethanesulfonate In ethanol; acetonitrile at 20℃; for 1h; Inert atmosphere; Irradiation; | 90% |

-
-
625-55-8
isopropyl formate

-
-
120014-30-4
debenzyldonepezil

| Conditions | Yield |
|---|---|
| Stage #1: debenzyldonepezil With potassium carbonate In methanol; water at 20℃; for 2h; Stage #2: isopropyl formate at 50℃; for 2h; | 88% |

| Conditions | Yield |
|---|---|
| With boron trifluoride In Hexadecane at 110℃; for 8h; Autoclave; | 88% |

| Conditions | Yield |
|---|---|
| With 1-(3-sulfopropyl)pyridinium phosphotungstate In neat (no solvent) at 70℃; for 0.166667h; Microwave irradiation; Sealed tube; Inert atmosphere; | 82% |

| Conditions | Yield |
|---|---|
| In benzene byproducts: CO2; ratio of HCOOCHMe2/trans-Mo(N2)2(dpe)2 10/1, refluxed under nitrogen for 30 min; cooling, addn. of n-hexane, filtered, washed with ether and n-hexane, dried in vac.; | A 7% B 55% C 6% D 2% E 81% |


| Conditions | Yield |
|---|---|
| With titanium tetrachloride; triethylamine In dichloromethane at -40 - 5℃; for 1.16667h; Temperature; Inert atmosphere; stereoselective reaction; | 81% |

| Conditions | Yield |
|---|---|
| With tributyl-amine; titanium tetrachloride In dichloromethane at -78 - 5℃; for 1.16667h; Inert atmosphere; stereoselective reaction; | 81% |

| Conditions | Yield |
|---|---|
| With 1-(3-sulfopropyl)pyridinium phosphotungstate In neat (no solvent) at 70℃; for 0.166667h; Microwave irradiation; Sealed tube; Inert atmosphere; | 78% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
625-55-8
isopropyl formate

-
-
51795-72-3
diethyl 3-methylbut-2-enylphosphonate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; lithium diisopropyl amide In tetrahydrofuran at -70℃; | 72% |

-
-
625-55-8
isopropyl formate

-
-
88016-30-2
(methoxycarbonyl)methyl p-toluenesulfonate

| Conditions | Yield |
|---|---|
| With titanium tetrachloride; triethylamine In dichloromethane at -45℃; for 1h; Inert atmosphere; stereoselective reaction; | 72% |

| Conditions | Yield |
|---|---|
| dodecacarbonyl-triangulo-triruthenium; P(C4H9)3 In pyridine at 180℃; for 10h; | 68% |
-
-
625-55-8
isopropyl formate

-
-
88016-31-3
(trifluoromethanesulfonyloxy)acetic acid methyl ester

| Conditions | Yield |
|---|---|
| With titanium tetrachloride; triethylamine In dichloromethane at -78 - 5℃; for 1.16667h; Inert atmosphere; stereoselective reaction; | 65% |
-
-
625-55-8
isopropyl formate

-
-
402-43-7
p-trifluoromethylphenyl bromide

-
-
444993-17-3
4-trifluoromethyl-benzoic acid isopropyl ester

| Conditions | Yield |
|---|---|
| Stage #1: p-trifluoromethylphenyl bromide With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: isopropyl formate In tetrahydrofuran; hexane at -78℃; for 3h; Stage #3: With iodine; potassium carbonate; isopropyl alcohol In tetrahydrofuran; hexane at -78 - 20℃; for 14h; | 60% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(II) bis(trifluoromethanesulfonate) at 130℃; for 12h; Sealed tube; | 51% |
-
-
625-55-8
isopropyl formate

-
-
120226-50-8
N-pivaloyl-piperonylmethylamine

| Conditions | Yield |
|---|---|
| Stage #1: N-pivaloyl-piperonylmethylamine With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: isopropyl formate In tetrahydrofuran for 0.5h; | 50% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride; triethylamine In dichloromethane at -40 - 5℃; for 1.16667h; Inert atmosphere; stereoselective reaction; | 50% |
-
-
625-55-8
isopropyl formate

-
-
108963-96-8
(S)-1-tert-butoxycarbonyl-5-methoxycarbonyl-pyrrolidin-2-one

-
-
127706-22-3
γ-formyl-N-Boc-L-pyroglutamic acid methyl ester

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran; hexane at -78 - -40℃; | 46% |

| Conditions | Yield |
|---|---|
| With (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; 4-cyano-1-isopropoxypyridin-1-ium trifluoromethanesulfonate In ethanol at 20℃; for 1h; Inert atmosphere; Irradiation; | 45% |
-
-
625-55-8
isopropyl formate

-
-
84358-13-4
N-[(tert-butoxy)carbonyl]piperidine-4-carboxylic acid

-
-
913264-42-3
tert-butyl 4-(3-isopropyl-1,2,4-oxadiazol-5-yl)piperidine-1-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: isopropyl formate; N-[(tert-butoxy)carbonyl]piperidine-4-carboxylic acid With 4-methyl-morpholine In ethyl acetate at 0 - 20℃; for 12h; Stage #2: With N-hydroxybutyramide In N,N-dimethyl-formamide at 110℃; for 7h; | 39.5% |
-
-
625-55-8
isopropyl formate

-
-
98-83-9
isopropenylbenzene

-
-
27751-80-0
isopropyl 3-hydroxy-3-phenyl-propanoate

| Conditions | Yield |
|---|---|
| With potassium dihydrogenphosphate; (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; 4-cyano-1-isopropoxypyridin-1-ium trifluoromethanesulfonate In ethanol; acetonitrile at 20℃; for 1h; Inert atmosphere; Irradiation; | 35% |
Isopropyl formate Consensus Reports
Reported in EPA TSCA Inventory.
Isopropyl formate Specification
The Isopropyl formate, with the CAS registry number 625-55-8, is also known as Formic acid, 1-methylethyl ester. It belongs to the product categories of Organics; C2 to C5; Carbonyl Compounds; Esters. Its EINECS registry number is 210-901-2. This chemical's molecular formula is C4H8O2 and molecular weight is 88.1051. Its IUPAC name is called propan-2-yl formate. Isopropyl formate is used as a spice for food and it can also be used as a fungicide, disinfectant and bactericide.
Physical properties of Isopropyl formate: (1)ACD/LogP: 0.65; (2)ACD/LogD (pH 5.5): 0.65; (3)ACD/LogD (pH 7.4): 0.65; (4)ACD/BCF (pH 5.5): 1.82; (5)ACD/BCF (pH 7.4): 1.82; (6)ACD/KOC (pH 5.5): 53.51; (7)ACD/KOC (pH 7.4): 53.51; (8)#H bond acceptors: 2; (9)#Freely Rotating Bonds: 2; (10)Index of Refraction: 1.373; (11)Molar Refractivity: 22.47 cm3; (12)Molar Volume: 98.6 cm3; (13)Surface Tension: 23.5 dyne/cm; (14)Density: 0.893 g/cm3; (15)Enthalpy of Vaporization: 31.23 kJ/mol; (16)Boiling Point: 70.8 °C at 760 mmHg; (17)Vapour Pressure: 127 mmHg at 25°C.
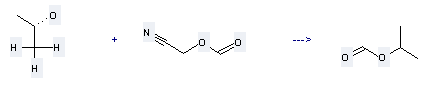
Preparation of Isopropyl formate: this chemical can be prepared by propan-2-ol and formyloxy-acetonitrile. This reaction will need reagent imidazole. The reaction time is 1 day with ambient temperature. The yield is about 62%.
Uses of Isopropyl formate: it can be used to produce propan-2-ol at temperature of 180 °C. This reaction will need catalyst Ru3(CO)12/P(C4H9)3 and solvent pyridine with reaction time of 10 hours. The yield is about 68%.

When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. You should keep it away from sources of ignition - No smoking. Whenever you will contact it, please wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: CC(C)OC=O
(2)InChI: InChI=1S/C4H8O2/c1-4(2)6-3-5/h3-4H,1-2H3
(3)InChIKey: RMOUBSOVHSONPZ-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 1400ug/kg (1.4mg/kg) | "Pesticide Index," Frear, E.H., ed., State College, PA, College Science Pub., 1969Vol. 4, Pg. 256, 1969. |
Related Products
- ISOPROPYL γ-FLUOROBUTYRATE
- Isopropyl (2S)-2-hydroxypropanoate
- Isopropyl 2-(3-nitrobenzylidene)acetoacetate
- Isopropyl 2,4-dichlorophenoxyacetate
- Isopropyl 2-bromo-2-methylpropanoate
- Isopropyl 2-cyanoacetate
- Isopropyl 2-methylbutanoate
- Isopropyl 3-amino-4-methylbenzoate
- Isopropyl 3-aminocrotonate
- Isopropyl 4-hydroxypiperidine-1-carboxylate
- 62555-84-4
- 625-56-9
- 625-57-0
- 62558-08-1
- 625-59-2
- 62559-23-3
- 625-60-5
- 62561-03-9
- 625-61-6
- 62561-74-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View