-
Name
Lactonitrile
- EINECS 201-163-2
- CAS No. 78-97-7
- Article Data66
- CAS DataBase
- Density 0.991
- Solubility
- Melting Point -40.15°C
- Formula C3H5 N O
- Boiling Point 90 ºC (17 mmHg)
- Molecular Weight 71.0788
- Flash Point 77 °C
- Transport Information UN 3276
- Appearance Straw colored liquid.
- Safety Poison by ingestion, skin contact, and subcutaneous routes. Moderately toxic by inhalation. In the presence of alkali, it evolves HCN. Combustible when exposed to heat or flame; can react vigorously with oxidizing materials. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of CN− and NOx. See also NITRILES.
- Risk Codes R23/25;R27
-
Molecular Structure
-
Hazard Symbols

- Synonyms Lactonitrile(6CI,7CI,8CI); (?à)-2-Hydroxypropanenitrile;(?à)-2-Hydroxypropionitrile;2-Hydroxypropanenitrile; 2-Hydroxypropionitrile; Acetaldehyde, cyanohydrin;Acetocyanohydrin; NSC 7764; a-Hydroxypropionitrile
- PSA 44.02000
- LogP -0.10922
Synthetic route

| Conditions | Yield |
|---|---|
| With triethylamine at 5 - 10℃; Product distribution / selectivity; | 99% |
| With triethylamine at 0 - 10℃; pH=2 - 3; | 99.54% |
| With sodium acetate; acetic acid; 2-hydroxy-propionitrile |

| Conditions | Yield |
|---|---|
| With sodium hydrogensulfite In water at 0℃; for 2h; | 80% |
| With acetic acid In diethyl ether; water | 22% |
-
-
114711-04-5
2-[(tert-butyldimethylsilyl)oxy]propanenitrile

-
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With P(MeNCH2CH2)3N In dimethyl sulfoxide at 80℃; for 19h; desilylation; | 80% |

| Conditions | Yield |
|---|---|
| With ytterbium(III) chloride; 2,6-bis[4′-isopropyloxazolin-2′-yl]pyridine In acetonitrile for 2h; Ambient temperature; | 61% |
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With tetrachloromethane; copper(II) choride dihydrate; chromium(III) acetylacetonate; water at 110℃; for 6h; Sealed tube; | 46% |
| With acetic acid; lithium iodide In [D3]acetonitrile at 20℃; for 3h; Irradiation; Inert atmosphere; |
-
-
74-90-8
hydrogen cyanide

-
-
151-50-8
potassium cyanide

-
-
75-07-0
acetaldehyde

-
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| at 25 - 30℃; |

-
-
74-90-8
hydrogen cyanide

-
-
75-07-0
acetaldehyde

-
A
-
107-13-1
acrylonitrile

-
B
-
78-97-7
2-hydroxy-propionitrile

-
-
74-90-8
hydrogen cyanide

-
-
67-66-3
chloroform

-
-
75-07-0
acetaldehyde

-
-
130-95-0
Quinine

-
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| at 25℃; |


| Conditions | Yield |
|---|---|
| With ammonium chloride In diethyl ether | |
| With Dowex-H+ In water for 24h; Ambient temperature; |
-
-
17374-27-5
1,5-dimethylimidazolidine-2,4-dione

-
A
-
16752-54-8
methylaminopropionitrile

-
B
-
57768-53-3
α-methylaminodipropionitrile

-
D
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; Equilibrium constant; K as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |

-
-
16752-54-8
methylaminopropionitrile

-
A
-
17374-27-5
1,5-dimethylimidazolidine-2,4-dione

-
B
-
57768-53-3
α-methylaminodipropionitrile

-
D
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With sodium carbonate at 50℃; Product distribution; | |
| With sodium hydrogencarbonate at 30℃; Rate constant; Equilibrium constant; k as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |

-
-
72-19-5
L-threonine

-
-
127-65-1
chloroamine-T

-
A
-
70-55-3
toluene-4-sulfonamide

-
B
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium chloride In water at 29.9 - 44.9℃; for 24h; Kinetics; Mechanism; Thermodynamic data; effect of +>, ->, and ; with HClO4 and H2SO4 also; solvent isotope effect; variation of ionic strength and addition of the product, p-toluenesulfonamide; decrease of dielectric constant by methanol; ΔH(excit.), ΔS(excit.), ΔG(excit.); |


| Conditions | Yield |
|---|---|
| With perchloric acid; N-chlorobenzotriazole; chloride In water at 29.9℃; Kinetics; Mechanism; |

| Conditions | Yield |
|---|---|
| With Candida tropicalis at 30℃; for 96h; Quantum yield; Hydrolitic efficiency of other microorganisms; Penicillium notatum, Aspergillus flavas, Pseudomonas fluorescens; |
-
-
2869-25-2
2,2'-iminodipropionitrile

-
B
-
78-97-7
2-hydroxy-propionitrile

-
C
-
51806-98-5, 95596-57-9, 2134-48-7
2-aminopropanenitrile

-
D
-
67337-69-3
methyl-5 hydantoine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; Equilibrium constant; K as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; chloroamine-T In water at 29.9 - 44.9℃; Thermodynamic data; Rate constant; Ea, log A, -ΔS(excit.), ΔH(excit.), ΔG(excit.); further acid concentration, further acid and alkaline media; |
-
-
57768-53-3
α-methylaminodipropionitrile

-
A
-
17374-27-5
1,5-dimethylimidazolidine-2,4-dione

-
B
-
16752-54-8
methylaminopropionitrile

-
D
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; Equilibrium constant; K as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |

-
-
151-50-8
potassium cyanide

-
-
918-04-7
potassium 1-hydroxyethane-1-sulfonate

-
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| In water |

-
A
-
2869-25-2
2,2'-iminodipropionitrile

-
B
-
78-97-7
2-hydroxy-propionitrile

-
C
-
51806-98-5, 95596-57-9, 2134-48-7
2-aminopropanenitrile

-
D
-
67337-69-3
methyl-5 hydantoine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; Equilibrium constant; K as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |


-
A
-
17374-27-5
1,5-dimethylimidazolidine-2,4-dione

-
B
-
16752-54-8
methylaminopropionitrile

-
C
-
57768-53-3
α-methylaminodipropionitrile

-
D
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; Equilibrium constant; K as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |

-
-
497-19-8
sodium carbonate

-
-
51806-98-5, 95596-57-9, 2134-48-7
2-aminopropanenitrile

-
A
-
2869-25-2
2,2'-iminodipropionitrile

-
B
-
4744-01-8
α-carboxyaminopropionamide

-
D
-
78-97-7
2-hydroxy-propionitrile

-
E
-
67337-69-3
methyl-5 hydantoine

-
F
-
4726-84-5
alanine amide

| Conditions | Yield |
|---|---|
| With ammonia In water at 50℃; Product distribution; Kinetics; pH=8.9 and 9.5; |

-
-
51806-98-5, 95596-57-9, 2134-48-7
2-aminopropanenitrile

-
A
-
2869-25-2
2,2'-iminodipropionitrile

-
C
-
78-97-7
2-hydroxy-propionitrile

-
D
-
67337-69-3
methyl-5 hydantoine

| Conditions | Yield |
|---|---|
| With sodium carbonate at 50℃; Product distribution; | |
| With sodium hydrogencarbonate at 30℃; Rate constant; Equilibrium constant; k as a function of pH and concentration of carbonate, borate, phosphate and ammonia; |

-
-
67337-69-3
methyl-5 hydantoine

-
A
-
2869-25-2
2,2'-iminodipropionitrile

-
C
-
78-97-7
2-hydroxy-propionitrile

-
D
-
51806-98-5, 95596-57-9, 2134-48-7
2-aminopropanenitrile

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; Equilibrium constant; K as a function of pH and the concentration of carbonate, borate, phosphate and ammonia; |


| Conditions | Yield |
|---|---|
| With perchloric acid; sodium bismuthate; hydrogen fluoride In water at 35℃; for 3h; Kinetics; Further Variations:; Temperatures; Oxidation; |

| Conditions | Yield |
|---|---|
| With pyridine; acetic acid; 2-hydroxy-propionitrile |

| Conditions | Yield |
|---|---|
| at 25 - 60℃; Rate constant; | |
| at 25 - 60℃; Equilibrium constant; |


| Conditions | Yield |
|---|---|
| at 25 - 30℃; |


| Conditions | Yield |
|---|---|
| at 200 - 225℃; Nickel-Kieselgur-Katalysator; |


| Conditions | Yield |
|---|---|
| With formaldehyd; perchloric acid; bromamine T In water at 29.9℃; Kinetics; Mechanism; Thermodynamic data; E(activ.), ΔG(excit.), ΔH(excit.), ΔS(excit.); other temperatures and solvent, effect of acid concentration, no influence of ionic strength and tosylamine addition; |
-
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| In diethyl ether; ethanol | 99% |
| In diethyl ether; ethanol | 815 g (99%) |
-
-
78-97-7
2-hydroxy-propionitrile

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
33695-65-7
1-cyanoethyl p-toluenesulfonate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; toluene at 0 - 25℃; | 98.34% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 0 - 10℃; | 98.34% |
-
-
98-60-2
4-chlorobenzenesulfonyl chloride

-
-
78-97-7
2-hydroxy-propionitrile

-
-
33695-77-1
p-chlorobenzenesulfonic acid 1-cyanoethyl ester

| Conditions | Yield |
|---|---|
| With sodium carbonate at 0 - 25℃; | 97.52% |
-
-
124-63-0
methanesulfonyl chloride

-
-
78-97-7
2-hydroxy-propionitrile

-
-
6839-20-9
2-[(methylsulfonyl)oxy]propanenitrile

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 25℃; for 24h; | 96.78% |
| With triethylamine In dichloromethane at 0℃; for 0.5h; Mesylation; | 91% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In toluene at 0 - 10℃; for 24h; | 96.01% |
| With triethylamine In dichloromethane at 0 - 20℃; for 3.5h; Substitution; | 88% |
-
-
78-97-7
2-hydroxy-propionitrile

-
-
598-81-2, 2043-43-8, 65144-02-7, 89673-71-2
(+/-)-lactamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-propionitrile With Caproamide; acetic acid at 50℃; for 0.5h; Schlenk technique; Inert atmosphere; Stage #2: With palladium (II) nitrate at 50℃; for 0.166667h; Schlenk technique; Inert atmosphere; | 96% |
| With CF3O3S(1-)*C36H35ClFeOP3Pt(1+); water; silver trifluoromethanesulfonate In tetrahydrofuran at 40℃; for 12h; Catalytic behavior; Inert atmosphere; | 93% |
| With [RuCl2(η3:η3-C10H16){PMe2(OH)}]; water at 20℃; for 72h; Inert atmosphere; Schlenk technique; Sealed tube; | 85% |

| Conditions | Yield |
|---|---|
| In pyridine at 20℃; for 2h; | 96% |
| With triethylamine In dichloromethane at 0 - 20℃; | 85% |
-
-
78-97-7
2-hydroxy-propionitrile

| Conditions | Yield |
|---|---|
| With hydrogenchloride; thiophosphate sodium salt hydrate In water; water-d2 at 50℃; for 24h; pH=6.5; Sealed tube; | 96% |
-
-
78-97-7
2-hydroxy-propionitrile

-
-
98-09-9
benzenesulfonyl chloride

-
-
10531-15-4
1-cyanoethyl benzenesulfonate

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran at 0 - 20℃; for 24h; | 95.2% |
-
-
421-83-0
trifluoromethane sulfonyl chloride

-
-
78-97-7
2-hydroxy-propionitrile

-
-
1403873-31-3
1-cyanoethyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With potassium carbonate at 0 - 25℃; for 20h; | 94.13% |
-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
78-97-7
2-hydroxy-propionitrile

-
-
114711-04-5
2-[(tert-butyldimethylsilyl)oxy]propanenitrile

| Conditions | Yield |
|---|---|
| With triethylamine; 2,8,9-Trimethyl-2,5,8,9-tetraaza-1-phospha-bicyclo[3.3.3]undecane 1-oxide In [D3]acetonitrile at 25℃; for 6h; | 94% |
| With P(MeNCH2CH2)3N; triethylamine In acetonitrile at 24℃; for 0.2h; | 90% |
| With 1H-imidazole In N,N-dimethyl-formamide Ambient temperature; | 70% |
-
-
78-97-7
2-hydroxy-propionitrile

-
-
586-75-4
4-chlorobenzoyl chloride

-
-
1569086-99-2
1-cyanoethyl 4-bromobenzoate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 94% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ammonium hydroxide; acetone | 92% |

| Conditions | Yield |
|---|---|
| With boron trifluoride at 0℃; for 18h; Addition; | 91% |
-
-
79-37-8
oxalyl dichloride

-
-
78-97-7
2-hydroxy-propionitrile

-
-
125849-94-7
3,5-dichloro-6-methyl-2(H)-1,4-oxazin-2-one

| Conditions | Yield |
|---|---|
| With triethylamine hydrochloride In chlorobenzene at 90℃; for 3h; | 90% |
| With triethylamine hydrochloride In chlorobenzene at 90℃; for 3h; Inert atmosphere; | 90% |
| Stage #1: oxalyl dichloride; 2-hydroxy-propionitrile In chlorobenzene at 0℃; Stage #2: With triethylamine hydrochloride In chlorobenzene at 0 - 90℃; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether at -5 - 26℃; for 18h; | 90% |
| With hydrogenchloride In diethyl ether at 0 - 5℃; for 48h; |

| Conditions | Yield |
|---|---|
| In ethanol for 18h; Reflux; | 90% |
| With titanium(IV) isopropylate In toluene at 40℃; for 18h; Inert atmosphere; Glovebox; | 56% |
-
-
109-92-2
ethyl vinyl ether

-
-
78-97-7
2-hydroxy-propionitrile

-
-
53411-95-3
2-<(1'-ethoxy)ethoxy> propionitrile

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 80℃; for 3h; | 88% |
| With acid |
-
-
78-97-7
2-hydroxy-propionitrile

-
-
52046-55-6
2,N-dihydroxy-propionamidine

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydroxylamine hydrochloride In ethanol at 20℃; | 87% |
| With hydroxylamine hydrochloride; sodium ethanolate | |
| With hydroxylamine hydrochloride; water; sodium carbonate |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water at -10 - -5℃; for 4h; Reflux; | 85.58% |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
78-97-7
2-hydroxy-propionitrile

-
-
1403873-31-3
1-cyanoethyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0℃; for 0.25h; Inert atmosphere; | 82% |
-
-
506-87-6
ammonium carbonate

-
-
78-97-7
2-hydroxy-propionitrile

-
A
-
39121-77-2
α-ureido-propionamide

-
B
-
67337-69-3
methyl-5 hydantoine

| Conditions | Yield |
|---|---|
| In water at 50℃; for 4h; pH=7.9; | A 20% B 80% |


| Conditions | Yield |
|---|---|
| In diethyl ether for 0.25h; Heating; | 80% |
-
-
1603-41-4
(5-methyl-pyridin-2-yl)amine

-
-
78-97-7
2-hydroxy-propionitrile

-
-
1176904-05-4
2-[(2,6-dimethylimidazo[1,2-a]pyridine-3-yl)amino]propanenitrile

| Conditions | Yield |
|---|---|
| Stage #1: (5-methyl-pyridin-2-yl)amine; 2-hydroxy-propionitrile With silica-gel-supported sulfuric acid at 20℃; Stage #2: With ammonia In water | 80% |
-
-
78-97-7
2-hydroxy-propionitrile

-
-
105-53-3
diethyl malonate

-
-
139003-80-8
ethyl 4-amino-2,5-dihydro-5-methyl-2-oxo-3-furancarboxylate

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In ethyl acetate for 48h; Ambient temperature; | 79% |
Lactonitrile Chemical Properties
IUPAC Name: 2-Hydroxypropanenitrile
CAS: 78-97-7
EINECS: 201-163-2
Following is the Molecular Structure of Lactonitrile (78-97-7):
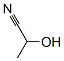
SMILES: N#CC(C)O
Molecular Formula: C3H5NO
Molecular Weight: 71.08
Melting Point:<21℃
Boiling Point: 183.4℃ at 760mmHg
Flash Point: 64.7℃
Molar Volume: 69.3cm3
Density: 1.025g/cm3
Molar Refractivity: 17.35cm3
Surface Tension: 39.6dyne/cm
Polarizability: 6.87 10-24cm3
Enthalpy of Vaporization: 48.84kJ/mol
Vapour Pressure: 0.222mmHg at 25℃
Refractive Index: n20/D 1.404
Lactonitrile Uses
Solvent, intermediate in production of Ethyl acetate and Lactic acid .
Lactonitrile Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| frog | LDLo | subcutaneous | 300mg/kg (300mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 5, Pg. 161, 1899. |
| mouse | LD50 | intraperitoneal | 15mg/kg (15mg/kg) | BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Toxicology and Applied Pharmacology. Vol. 84, Pg. 533, 1986. |
| rabbit | LD50 | skin | 20uL/kg (0.02mL/kg) | American Industrial Hygiene Association Journal. Vol. 30, Pg. 470, 1969. | |
| rabbit | LDLo | subcutaneous | 5200ug/kg (5.2mg/kg) | PERIPHERAL NERVE AND SENSATION: SPASTIC PARALYSIS WITH OR WITHOUT SENSORY CHANGE LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 5, Pg. 161, 1899. |
| rat | LCLo | inhalation | 125ppm/4H (125ppm) | American Industrial Hygiene Association Journal. Vol. 30, Pg. 470, 1969. | |
| rat | LD50 | oral | 87mg/kg (87mg/kg) | American Industrial Hygiene Association Journal. Vol. 30, Pg. 470, 1969. |
Lactonitrile Consensus Reports
EPA Extremely Hazardous Substances List. Reported in EPA TSCA Inventory. Cyanide and its compounds are on the Community Right-To-Know List.
Lactonitrile Safety Profile
Hazard Codes: ![]() T+ Very toxic, T Toxic.
T+ Very toxic, T Toxic.
Risk Statements: 23/25-27-25-20
R23/25: Toxic by inhalation and if swallowed.
R27: Very Toxic in contact with skin.
R25: Toxic if swallowed.
R20: Harmful by inhalation.
Safety Statements: 13-36/37/39-45-23
S13: Keep away from food, drink and animal feeding stuffs.
S36/27/39: Wear suitable protective clothing, gloves and eye/face protection.
S45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible).
S23: Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer).
RIDADR: UN 3276 6.1/PG 1
WGK: Germany 3
RTECS: OD8225000
HazardClass: 6.1(a)
PackingGroup: II
Poison by ingestion, skin contact, and subcutaneous routes. Moderately toxic by inhalation. In the presence of alkali, it evolves HCN .Combustible when exposed to heat or flame; can react vigorously with oxidizing materials. To fight fire, use foam, CO2 , dry chemical.
Lactonitrile Specification
Lactonitrile (78-97-7),which also can be called for 2-Hydroxypropanenitrile ; 2-Hydroxypropannitril ; 2-Hydroxypropannitril ; 2-Hydroxypropionitrile ; Acetaldehyde ; Cyanohydrin ; Acetocyanohydrin ; Ethylidene cyanohydrin ; Lactonitrile ; Laktonitril ; Laktonitril ; Propanenitrile,2-hydroxy- ; Propionitrile,2-hydroxy- ; Alpha-hydroxypropionitrile . Lactonitrile (78-97-7) is a straw colored liquid and it's very soluble in water.When heated to decomposition it emits toxic fumes of CN− and NOx .This chemical should be stored in a cool, dry, dark location in a tightly sealed container or cylinder. Keep away from incompatible materials, ignition sources and untrained individuals. Secure and label area. Protect containers/cylinders from physical damage.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View