-
Name
Octadecanamide
- EINECS 204-693-2
- CAS No. 124-26-5
- Article Data54
- CAS DataBase
- Density 0.868 g/cm3
- Solubility 102μg/L at 20℃
- Melting Point 98-102 °C(lit.)
- Formula C18H37NO
- Boiling Point 408.2 °C at 760 mmHg
- Molecular Weight 283.498
- Flash Point 207.5 °C
- Transport Information
- Appearance light yellow granules
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Stearamide(6CI,7CI,8CI);Adogen 42;Advawax 290;Alflow S 10;Amide AP;Amide S;Amide S (binder);Amide T;Armid 18;Armoslip 18LF;Atmer SA 1750;Crodamide S;Crodamide SR;Diamid AP 1;EBF;Fatty Amide S;Fatty Amide T;G 270;Hidorin F792;Hidorin M 7;Hymicron G 270;Hymicron L 271;Kao Fatty Amide S;KawaslipVL;Kemamide S;L 271;Lutamide S;NSC 66462;Neutron 2;Neutron 2S;Selosol 2M;Stearic acid amide;Stearic amide;Stearoylamide;Stearoylamine;Stearylamide;Uniwax 1750;Stearamide;
- PSA 43.09000
- LogP 6.43350
Synthetic route

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate; ammonia at 165℃; for 7h; Reagent/catalyst; Temperature; Large scale; | 98.4% |
| With ammonium bicarbonate; formamide for 0.15h; Irradiation; | 82% |
| Stage #1: stearic acid With 1,1'-carbonyldiimidazole In dichloromethane at 20℃; for 0.5h; Stage #2: With ammonia In dichloromethane for 12h; | 23% |
-
-
120158-01-2
Octadecanoyl-carbamic acid tert-butyl ester

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane for 1h; Ambient temperature; | 97% |
-
-
77165-65-2
octadecanoyl azide

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| With zinc(II) tetrahydroborate In 1,2-dimethoxyethane for 0.5h; Ambient temperature; | 95% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N-hydroxy-ethanamine; water at 100℃; for 5h; | 90% |
| With sodium hydroxide; poly(ethylene glycol)-400 for 0.0166667h; microwave irradiation; | 78% |
| (i) HCl, SnCl2, Et2O, (ii) H2O; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With fluorenone imine; oxygen In toluene for 60h; Reflux; | 51% |

| Conditions | Yield |
|---|---|
| With ammonia; water at 180℃; |

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| at 130 - 150℃; |

-
A
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
B
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| at 130 - 150℃; |
-
-
102899-77-4
(stearoylamino-methylsulfanyl)-acetic acid

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| at 180 - 190℃; |

| Conditions | Yield |
|---|---|
| at 160℃; |


| Conditions | Yield |
|---|---|
| at 160℃; | |
| With diammonium phosphate at 195℃; | |
| at 160℃; | |
| at 205 - 210℃; |

| Conditions | Yield |
|---|---|
| With diethyl ether; ammonia | |
| With ammonia | |
| With ammonia |
-
-
40652-01-5
N-(1-Cyanethyl)-stearoylamid

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| (thermolysis); |

| Conditions | Yield |
|---|---|
| at 135 - 200℃; Kinetics; |

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| at 230℃; |
-
-
102899-77-4
(stearoylamino-methylsulfanyl)-acetic acid

-
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: SOCl2 2: NH3 View Scheme |

-
-
38428-49-8
N-Boc-octadecylamine

-
A
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 98 percent / ruthenium dioxide hydrate, 10percent aqueous NaIO4 / ethyl acetate / 3 h / Ambient temperature 2: 97 percent / trifluoroacetic acid / CH2Cl2 / 1 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 95 percent / triethylamine / H2O; dioxane / 6 h / Ambient temperature 2: 98 percent / ruthenium dioxide hydrate, 10percent aqueous NaIO4 / ethyl acetate / 3 h / Ambient temperature 3: 97 percent / trifluoroacetic acid / CH2Cl2 / 1 h / Ambient temperature View Scheme |
-
-
53422-22-3
acetonin

-
-
144-55-8
sodium hydrogencarbonate

-
-
112-76-5
Stearoyl chloride

-
-
124-26-5
stearamide

-
-
124-40-3
dimethyl amine

-
-
2733-91-7
methyl (9Z,12S,13R)-12,13-epoxy-9-octadecenoate

-
A
-
301-02-0
cis-9-octadecenoamide

-
B
-
1323108-64-0
N,N-dimethyl-(12S,13R)-epoxy-cis-9-octadecenyl amide

-
C
-
629-54-9
Palmitamide

-
D
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl amine; methyl (9Z,12S,13R)-12,13-epoxy-9-octadecenoate With sodium methylate In methanol for 2h; Reflux; Stage #2: In methanol at 0℃; for 24.25h; |


| Conditions | Yield |
|---|---|
| With anthracene at 20℃; Kinetics; Reagent/catalyst; Photolysis; |
-
-
933460-58-3
1-{(2S)-2-[(diisopropoxyphosphoryl)methoxy-3-(triphenylmethoxy)]propyl}-5-azacytosine

-
-
21132-76-3
docosanoyl chloride

-
A
-
1608459-53-5
(S,E)-(8-amino-3-(hydroxymethyl)-6,10-dioxo-2-oxa-5,7,9-triazahentriacont-7-en-1-yl)phosphonic acid

-
B
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: pyridine / 48 h / 20 °C 2: acetonitrile / 24 h / 20 °C View Scheme |

-
-
2857-97-8
trimethylsilyl bromide

-
-
1608457-43-7
1-(2S)-2-[(diisopropoxyphosphoryl)methoxy-3-(triphenylmethoxy)propyl-N4-docosanoyl-5-azacytosine]

-
A
-
1608459-53-5
(S,E)-(8-amino-3-(hydroxymethyl)-6,10-dioxo-2-oxa-5,7,9-triazahentriacont-7-en-1-yl)phosphonic acid

-
B
-
124-26-5
stearamide

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 24h; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: acetic acid methyl ester; sulfuric acid View Scheme |

| Conditions | Yield |
|---|---|
| With ammonia at 180℃; for 6h; Autoclave; | 91 %Chromat. |

| Conditions | Yield |
|---|---|
| With triethylamine; trifluoroacetyl chloride In dichloromethane | 95% |
| at 295℃; for 1h; Temperature; | 86% |
| With trichloromethyl chloroformate; benzene |

| Conditions | Yield |
|---|---|
| Stage #1: stearamide With titanium(IV) isopropylate In toluene at 20 - 100℃; Stage #2: With hydrogenchloride In diethyl ether; toluene | 90% |
| Yield given. Multistep reaction; |
-
-
27126-76-7
[hydroxy(tosyloxy)iodo]benzene

-
-
124-26-5
stearamide

-
-
67038-06-6
heptadecylammonium p-toluenesulfonate

| Conditions | Yield |
|---|---|
| In acetonitrile at 65 - 70℃; for 0.0333333h; | 89.5% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 2h; Heating; | 89% |

| Conditions | Yield |
|---|---|
| With cerium(IV) oxide In 1,3,5-trimethyl-benzene at 165℃; for 22h; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With niobium(V) oxide; water In neat (no solvent) for 20h; Reflux; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With lithium-tris(diethylamino)hydridoaluminate In tetrahydrofuran for 12h; Ambient temperature; | 83% |
| Multi-step reaction with 2 steps 1: thionyl chloride 2: tin (II)-chloride; hydrogen chloride; diethyl ether / und beim Verseifen des Reaktionsprodukts mit warmem Wasser View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitrobenzaldehdye; stearamide With copper(I) bromide In tetrachloromethane; acetonitrile at 20℃; for 0.25h; Inert atmosphere; Stage #2: With N-Bromosuccinimide In tetrachloromethane; acetonitrile at 90℃; for 15h; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(I) bromide In decane at 80℃; for 6h; | 78% |

| Conditions | Yield |
|---|---|
| Stage #1: stearamide With borane tetrahydrofuran In toluene at 0 - 20℃; for 1h; Stage #2: leelamine In toluene Reflux; | 77.68% |
-
-
696-59-3
cis,trans-2,5-dimethoxytetrahydrofuran

-
-
124-26-5
stearamide

-
A
-
86734-18-1
1-Pyrrol-1-yl-octadecan-1-one

-
B
-
638-65-3
stearonitrile

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide In toluene at 60 - 110℃; for 1.25h; | A 75% B 25% |


| Conditions | Yield |
|---|---|
| Stage #1: stearamide With triethylamine; methyl chloroformate In dichloromethane at 0 - 20℃; for 2h; Stage #2: ethanolamine In dichloromethane at 0 - 20℃; for 20h; | 74% |
-
-
86428-38-8
1,4-dichloro-1,4-dimethoxybutane

-
-
124-26-5
stearamide

-
-
86734-18-1
1-Pyrrol-1-yl-octadecan-1-one

| Conditions | Yield |
|---|---|
| With Amberlyst A-21 resin In acetonitrile at 55℃; for 16h; | 72% |

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-ethane at 100℃; for 24h; | 71% |

| Conditions | Yield |
|---|---|
| With n-butyllithium; cyclopentyl methyl ether In hexane at 115℃; for 24h; Reagent/catalyst; Inert atmosphere; | 65% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; copper(I) bromide In ethyl acetate at 50℃; for 12h; | 42% |

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; copper(I) bromide In ethyl acetate at 25℃; for 6h; | 42% |

| Conditions | Yield |
|---|---|
| In toluene Heating; | 25% |

| Conditions | Yield |
|---|---|
| at 150℃; |

| Conditions | Yield |
|---|---|
| at 150℃; |


| Conditions | Yield |
|---|---|
| With acetic acid | |
| With acetic acid |
Octadecanamide Chemical Properties
EINECS: 204-693-2
Molecular Formula: C18H37NO
Molecular Weight: 283.49
Nominal Mass: 283 Da
Average Mass: 283.4925 Da
Monoisotopic Mass: 283.287515 Da
Index of Refraction: 1.457
Molar Refractivity: 89 cm3
Molar Volume: 326.4 cm3
Surface Tension: 32.9 dyne/cm
Density: 0.868 g/cm3
Flash Point: 207.5 °C
Enthalpy of Vaporization: 66.02 kJ/mol
Boiling Point: 408.2 °C at 760 mmHg
Vapour Pressure: 7.11E-07 mmHg at 25 °C
Melting Point: 98-102 °C(lit.)
Appearance: Light yellow granules
Structure of Octadecanamide (CAS NO.124-26-5):
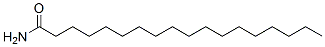
IUPAC Name: Octadecanamide
Canonical SMILES: CCCCCCCCCCCCCCCCCC(=O)N
InChI: InChI=1S/C18H37NO/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h2-17H2,1H3,(H2,19,20)
InChIKey: LYRFLYHAGKPMFH-UHFFFAOYSA-N
Product Category of Octadecanamide (CAS NO.124-26-5): Color Former & Related Compounds;Functional Materials;Sensitizer
Octadecanamide Uses
Octadecanamide (CAS NO.124-26-5) is commonly used as smoothness agent and release agent for PVC, polyolefin, polystyrene and other plastics.
Octadecanamide Toxicity Data With Reference
Octadecanamide (CAS NO.124-26-5) hasn't been listed as a carcinogen by NTP, IARC, ACGIH, or CA Prop 65. You can see actual entry in RTECS for complete information. The toxicological properties have not been fully investigated.
Octadecanamide Consensus Reports
Reported in EPA TSCA Inventory.
Octadecanamide Safety Profile
Questionable carcinogen with experimental tumorigenic data. When heated to decomposition it emits toxic fumes of NOx.
Safety Statements: 22-24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 1
Octadecanamide Specification
Octadecanamide , its cas register number is 124-26-5. It also can be called Stearamide ; Kemamide S ; and Stearylamide .It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. While, it's inhaled: Remove from exposure and move to fresh air immediately. Then you have the ingesting of the product : Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
In addition, Octadecanamide (CAS NO.124-26-5) is not compatible with strong oxidizing agents, strong acids, and you must not take it with incompatible materials. It should be exposured to moist air or water. And also prevent it to broken down into hazardous decomposition products: Nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
Related Products
- Octadecanamide
- Octadecanamide, N-((4-hydroxy-3-methoxyphenyl)methyl)-
- Octadecanamide,N-(4-hydroxyphenyl)-
- Octadecanamide,N-(hydroxymethyl)-
- Octadecanamide,N-[2-(3,4-dihydroxyphenyl)ethyl]-
- Octadecanamide,N-[2-[(2-hydroxyethyl)amino]ethyl]-
- Octadecanamide,N-[3-(dimethylamino)propyl]-
- Octadecanamide,N-[9-(diethylamino)-5H-benzo[a]phenoxazin-5-ylidene]-
- 12427-38-2
- 12427-42-8
- 124276-32-0
- 124276-61-5
- 124276-95-5
- 12428-54-5
- 124-28-7
- 124-30-1
- 124312-73-8
- 12431-56-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View