-
Name
Octamethylcyclotetrasiloxane
- EINECS 209-136-7
- CAS No. 556-67-2
- Article Data107
- CAS DataBase
- Density 0.95 g/cm3
- Solubility insoluble in water
- Melting Point 17-18 °C(lit.)
- Formula C8H24O4Si4
- Boiling Point 175 °C at 760 mmHg
- Molecular Weight 296.618
- Flash Point 63.2 °C
- Transport Information UN 1993 3/PG 3
- Appearance Colorless clear liquid
- Safety 36/37-46-51-61
- Risk Codes 53-62
-
Molecular Structure
-
Hazard Symbols
 N
N
- Synonyms Dimethylcyclosiloxane;Cyclic dimethylsiloxane tetramer;Dow Corning 244;KF 994;LS 8620;Mirasil CM 4;NSC 345674;NUCSilicone VS 7207;Octamethylcyclotetrasiloxane;DMC;
- PSA 36.92000
- LogP 2.87360
Synthetic route
-
-
433725-45-2
2,2,4,4,6,6-hexamethyl-8,8-divinylcyclotetrasiloxane

-
A
-
556-67-2
octamethylcyclotetrasiloxane

-
B
-
541-02-6
decamethylcyclopentasiloxane

-
D
copoly(dimethylsiloxane/divinylsiloxane), prepared by anionic polymerization of 2,2,4,4,6,6-hexamethyl-8,8-divinylcyclotetrasiloxane; monomer(s): 2,2,4,4,6,6-hexamethyl-8,8-divinylcyclotetrasiloxane -
copoly(dimethylsiloxane/divinylsiloxane), prepared by anionic polymerization of 2,2,4,4,6,6-hexamethyl-8,8-divinylcyclotetrasiloxane; monomer(s): 2,2,4,4,6,6-hexamethyl-8,8-divinylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| With P4-t-Bu at 80℃; for 0.333333h; Further byproducts given; | A n/a B n/a C n/a D 80% |

-
-
67-68-5
dimethyl sulfoxide

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
2373-51-5
chloro-methylsulfanyl-methane

-
B
-
541-05-9
Hexamethylcyclotrisiloxane

-
C
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
541-02-6
decamethylcyclopentasiloxane

-
E
-
540-97-6
dodecamethyl-cyclohexasiloxane

| Conditions | Yield |
|---|---|
| at 0℃; Mechanism; other diorganyldichlorosilanes; reactions in the presence of hexamethyldisiloxane, trimethylchlorosilane, tetramethoxysilane, tetraethoxysilane; | A 79% B 48% C 35% D 12% E 5% |

-
-
67-68-5
dimethyl sulfoxide

-
A
-
2373-51-5
chloro-methylsulfanyl-methane

-
B
-
541-05-9
Hexamethylcyclotrisiloxane

-
C
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
541-02-6
decamethylcyclopentasiloxane

| Conditions | Yield |
|---|---|
| With dimethylsilicon dichloride at 0℃; Further byproducts given; | A 79% B 48% C 35% D 5% |

-
-
1529-17-5
phenyltrimethylsilyl ether

-
A
-
556-67-2
octamethylcyclotetrasiloxane

-
B
-
3440-02-6
dimethyl-diphenoxy-silane

-
C
-
18406-79-6
C13H26O3Si3

| Conditions | Yield |
|---|---|
| With gallium(III) iodide at 175 - 180℃; for 3h; Yield given; Further byproducts given. Title compound not separated from byproducts; | A n/a B 79% C n/a D n/a |

-
-
19061-00-8
bis(trimethylsilyl)ketene

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| at 700℃; flash vacuum pyrolysis; | A 22% B 16% C 76% |

-
-
98991-82-3
(trimethylsilyl)(dimethylsilyl)ketene

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
1066-54-2
trimethylsilylacetylene

| Conditions | Yield |
|---|---|
| at 700℃; | A 22% B 16% C 76% |

-
-
87594-05-6
1-(1-Adamantyl)-2-diazo-2-(pentamethyldisilanyl)ethanone

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
82094-48-2
1-(trimethylsilylethynyl)adamantane

| Conditions | Yield |
|---|---|
| at 400℃; | A 21% B 20% C 73% |

-
-
52826-45-6
(benzylimino)triphenylphosphorane

-
-
65181-02-4
3,3-dimethyl-6-oxa-3-silabicyclo<3.1.0>hexane

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
113017-78-0
2,2,4,4-tetramethyl-6-vinyl-1,3-dioxa-2,4-disilacyclohexane

-
D
-
106-99-0
buta-1,3-diene

| Conditions | Yield |
|---|---|
| at 130℃; for 1h; | A n/a B n/a C 70% D n/a |
| at 130℃; for 1h; |

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| With water Autoclave; | A 63.7% B 27.2% |
| With water In toluene at 20℃; for 27h; | A 17 % Chromat. B 38% |
| With water Autoclave; | A 15.9% B 17.4% |
| With water In toluene at 20℃; for 27h; | A 4% B 44 % Chromat. |
| With tetrabutylammonium tetrafluoroborate; oxygen In tetrahydrofuran Product distribution; Further Variations:; Reagents; amount of electricity; type of electrolysis cell; Electrochemical reaction; |
-
-
541-05-9
Hexamethylcyclotrisiloxane

-
-
88703-87-1
1,2-silaoxetene 2,2-dimethyl-3-(trimethylsilyl)-4-adamantyl-1-oxa-2-silacyclobutene

-
A
-
556-67-2
octamethylcyclotetrasiloxane

-
B
-
82094-48-2
1-(trimethylsilylethynyl)adamantane

| Conditions | Yield |
|---|---|
| In benzene at 120℃; | A 48% B 62% |

-
-
1113-12-8
diallyl(dimethyl)silane

-
-
421-85-2
Trifluoromethanesulfonamide

-
A
-
556-67-2
octamethylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| With tert-butylhypochlorite; sodium iodide In acetonitrile at -30℃; for 24h; Inert atmosphere; Darkness; | A n/a B 28% C 61% |

-
-
15576-81-5
diiododimethylsilane

-
A
-
556-67-2
octamethylcyclotetrasiloxane

-
B
-
541-02-6
decamethylcyclopentasiloxane

| Conditions | Yield |
|---|---|
| With 3,3-dimethyl-butan-2-one; zinc In dichloromethane for 0.166667h; Ambient temperature; | A 60% B 18% |
-
-
75-78-5
dimethylsilicon dichloride

-
A
-
556-67-2
octamethylcyclotetrasiloxane

-
B
-
13452-94-3
Tetradecamethylcycloheptasilan

-
C
-
13452-92-1
decamethylcyclopentasilane

-
D
-
4098-30-0
dodecamethylcyclohexasilane

| Conditions | Yield |
|---|---|
| With tetrahydrofuran; lithium 0 deg C, 1 h, then 20-25 deg C, 2 h; Further byproducts given. Yields of byproduct given; | A n/a B n/a C 60% D n/a |
| With tetrahydrofuran; lithium 0 deg C, 1 h, then 20-25 deg C, 2 h; Further byproducts given. Yields of byproduct given; | A n/a B n/a C n/a D 60% |
| With tetrahydrofuran; lithium at -5 - 0℃; for 1h; Yield given. Further byproducts given. Yields of byproduct given; |

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
541-02-6
decamethylcyclopentasiloxane

| Conditions | Yield |
|---|---|
| With pyridine; sodium hydrogencarbonate In ethyl acetate at 20℃; for 3h; | A 60% B 20% C 15% |
| With water Autoclave; | A 51.3% B 29.2% C 8% |
| With composite catalyst at 150℃; under 675.068 Torr; for 0.5h; Temperature; Pressure; Reagent/catalyst; Large scale; | A n/a B 43% C n/a |

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
541-02-6
decamethylcyclopentasiloxane

-
D
-
540-97-6
dodecamethyl-cyclohexasiloxane

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide at 0℃; Further byproducts given; | A 48% B 35% C 12% D 5% |
| With tetrahydrofuran; lithium at 40 - 45℃; for 1h; Yield given. Further byproducts given. Yields of byproduct given; | |
| With hydrogenchloride In toluene at 20℃; for 4h; Yield given. Further byproducts given. Yields of byproduct given; |

-
-
17306-11-5
(allyl)(cyclopentadienyl)dimethylsilane

-
-
100-52-7
benzaldehyde

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
7338-50-3
6-phenylfulvene

| Conditions | Yield |
|---|---|
| A n/a B n/a C 47% |

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
107-50-6
tetradecamethylcycloheptasiloxane

-
B
-
541-05-9
Hexamethylcyclotrisiloxane

-
C
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
541-02-6
decamethylcyclopentasiloxane

-
E
-
540-97-6
dodecamethyl-cyclohexasiloxane

| Conditions | Yield |
|---|---|
| With H2O In water slow addn. of 4l (CH3)2SiCl2 to 12l H2O at 15-20°C withorous stirring; further products;; distn.;; | A n/a B 0.5% C 42% D 6.7% E 1.6% |
| With hydrogenchloride In toluene at 20℃; for 4h; Product distribution; diff. concentration of aq. HCl; diff. reaction time; other difunctional organochlorosilanes; | |
| With H2O In diethyl ether; water addn. of (CH3)2SiCl2 to ether-H2O; further products;; distn.;; |

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
556-67-2
octamethylcyclotetrasiloxane

-
B
-
541-02-6
decamethylcyclopentasiloxane

| Conditions | Yield |
|---|---|
| With water at 15 - 20℃; Autoclave; | A 42% B 6.7% |
-
-
34485-82-0
2,3-dimethyl-3,4-epoxy-1-butene

-
-
4098-30-0
dodecamethylcyclohexasilane

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
513-81-5
2,3-dimethyl-buta-1,3-diene

| Conditions | Yield |
|---|---|
| at 0℃; Irradiation; | A 3% B 4% C 38% D 25% |

-
-
681-84-5
tetramethylorthosilicate

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
2373-51-5
chloro-methylsulfanyl-methane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
84521-21-1
1,1-dimethyltetramethoxydisiloxane

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide at 0℃; | A n/a B n/a C 37% |

-
-
930-22-3
epoxybutene

-
-
4098-30-0
dodecamethylcyclohexasilane

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
106-99-0
buta-1,3-diene

| Conditions | Yield |
|---|---|
| at 0℃; Irradiation; | A 3% B 6% C 15% D 35% |

-
-
119-61-9
benzophenone

-
-
17306-11-5
(allyl)(cyclopentadienyl)dimethylsilane

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
2175-90-8
6,6-Diphenylfulvene

| Conditions | Yield |
|---|---|
| With quartz In benzene at 600℃; | A n/a B n/a C 28% |

-
-
78-10-4
tetraethoxy orthosilicate

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
2373-51-5
chloro-methylsulfanyl-methane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
53201-01-7
1,1-dimethyl-1,3,3,3-tetraethoxydisiloxane

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide at 0℃; | A n/a B n/a C 28% |

-
-
3277-26-7
1,1,3,3-Tetramethyldisiloxane

-
A
-
107-50-6
tetradecamethylcycloheptasiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
541-02-6
decamethylcyclopentasiloxane

-
D
-
540-97-6
dodecamethyl-cyclohexasiloxane

| Conditions | Yield |
|---|---|
| With iodine In dichloromethane for 0.333333h; Heating; Further byproducts given; | A 14% B 26% C 21% D 15% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
141-62-8
decamethyltetrasiloxane

-
C
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
141-63-9
dodecamethylpentasiloxane

| Conditions | Yield |
|---|---|
| With water In toluene at 20℃; for 27h; Further byproducts given; | A n/a B 22% C n/a D 10 % Chromat. |
| With water In toluene at 20℃; for 27h; Further byproducts given; | A n/a B 22 % Chromat. C n/a D 10% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
75-78-5
dimethylsilicon dichloride

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
141-62-8
decamethyltetrasiloxane

-
C
-
556-67-2
octamethylcyclotetrasiloxane

-
D
-
107-52-8
tetradecamethylhexasiloxane

| Conditions | Yield |
|---|---|
| With water In toluene at 20℃; for 27h; Further byproducts given; | A n/a B 22% C n/a D 6 % Chromat. |

-
-
4098-30-0
dodecamethylcyclohexasilane

-
A
-
541-05-9
Hexamethylcyclotrisiloxane

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
38041-04-2
octamethylcyclotetrasilane

-
D
-
13452-92-1
decamethylcyclopentasilane

| Conditions | Yield |
|---|---|
| With sodium perchlorate In tetrahydrofuran Photolysis; | A 73 % Chromat. B 20% C n/a D n/a |


-
A
-
107-46-0
Hexamethyldisiloxane

-
B
-
541-05-9
Hexamethylcyclotrisiloxane

-
C
-
556-67-2
octamethylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| In decane byproducts: H2S; Ar atmosphere; decompn. (165°C, 13 h); GLC, chromato-mass spectroscopy; | A 15% B 15% C 15% |

-
-
2943-62-6
1-chloro-1,1,3,3,3-pentamethyldisiloxane

-
A
-
16029-98-4
trimethylsilyl iodide

-
B
-
556-67-2
octamethylcyclotetrasiloxane

-
C
-
18297-87-5
1-chloroheptamethyltrisiloxane

-
D
-
14415-31-7
1-chloro-1,1,3,3,5,5,7,7,7-nonamethyltetrasiloxane

| Conditions | Yield |
|---|---|
| With sodium iodide In acetonitrile for 18h; Mechanism; Ambient temperature; other siloxanes; other reagent, var. temp.; | A 14% B 10% C n/a D n/a |


| Conditions | Yield |
|---|---|
| With aluminium(III) iodide at 140 - 170℃; for 5h; | 99% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
1152310-87-6
1,3-bis(3-azidopropyl)-1,1,3,3-tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 60℃; for 13h; | 98% |
-
-
3277-26-7
1,1,3,3-Tetramethyldisiloxane

-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
16881-77-9
(dimethoxy)methylsilane

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 20 - 60℃; for 15.25h; | 95% |


| Conditions | Yield |
|---|---|
| With China clay; sulfuric acid at 100℃; for 120h; | 94.3% |


| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 20 - 60℃; for 15.25h; | 94% |

-
-
3277-26-7
1,1,3,3-Tetramethyldisiloxane

-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
995-82-4
1,1,3,3,5,5,7,7,9,9,11,11-dodecamethylhexasiloxane

| Conditions | Yield |
|---|---|
| With Kg-23 catalyst at 80℃; for 12h; Inert atmosphere; | 93% |
| With potassium hydroxide In water; toluene at 50℃; for 2h; | 72.9% |
| With sulfuric acid | |
| With trimethylsilyl trifluoromethanesulfonate for 24h; |

| Conditions | Yield |
|---|---|
| In methanol | 92% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
865-37-2
dimethylaluminum hydride

-
-
254763-44-5
(CH3)2Si((OAl(CH3)2)2Si(CH3)2H)2

| Conditions | Yield |
|---|---|
| In hexane stirring (4 d); distn. (vac.); | 90% |

| Conditions | Yield |
|---|---|
| mixed in a quarts tube; heated to 180°C for 24 h; pressure (28 psig); purified by vac. distillation; recrystd. (cold pentane, or vac. sublimation); elem. anal.; | 90% |
-
-
556-67-2
octamethylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 4h; Time; Reflux; | 90% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
1152310-87-6
1,3-bis(3-azidopropyl)-1,1,3,3-tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 60℃; for 13h; | 90% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 4h; | 89% |
-
-
3277-26-7
1,1,3,3-Tetramethyldisiloxane

-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
33006-95-0
C20H62O9Si10

| Conditions | Yield |
|---|---|
| With Kg-23 catalyst In toluene at 80℃; for 12h; Inert atmosphere; | 86% |
| With sulfuric acid | |
| With trimethylsilyl trifluoromethanesulfonate for 24h; |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
88351-31-9
(E)-But-2-enedioic acid mono-(3-{3-[3-((E)-3-carboxy-acryloyloxy)-propyl]-1,1,3,3-tetramethyl-disiloxanyl}-propyl) ester

| Conditions | Yield |
|---|---|
| With KU-23 ion-exchange resin at 90℃; for 7h; | 86% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
A
-
16029-98-4
trimethylsilyl iodide

-
B
-
107-46-0
Hexamethyldisiloxane

-
C
-
74-88-4
methyl iodide

| Conditions | Yield |
|---|---|
| With aluminium(III) iodide at 140 - 170℃; for 5h; | A n/a B 83.5% C n/a |

-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
2554-06-5
2,4,6,8-tetramethyl-2,4,6,8-tetravinyl-cyclotetrasiloxane

-
-
18395-32-9
2-vinyl-2,4,4,6,6-pentamethylcyclotrisiloxane

| Conditions | Yield |
|---|---|
| With triphenylphosphine; potassium hydroxide at 95 - 145℃; for 2h; Temperature; Concentration; Reagent/catalyst; Autoclave; | 83.4% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
18132-72-4
1,3-bis<3-chloropropyl>-1,1,3,3-tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| Purolite CT-175 at 100℃; for 17h; | 83% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
88351-31-9
(E)-But-2-enedioic acid mono-(3-{3-[3-((E)-3-carboxy-acryloyloxy)-propyl]-1,1,3,3-tetramethyl-disiloxanyl}-propyl) ester

| Conditions | Yield |
|---|---|
| With KU-23 ion-exchange resin at 90℃; for 7h; | 82% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
70877-11-1
bis<4-(methacryloyloxy)butyl>tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In chloroform at 25℃; for 24h; | 82% |
-
-
556-67-2
octamethylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In chloroform at 25℃; for 24h; | 81.1% |
-
-
18001-60-0
1,1,3,3-tetraethoxy-1,3-dimethyldisiloxane

-
-
556-67-2
octamethylcyclotetrasiloxane

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 145℃; | 81% |
-
-
1066-35-9
dimethylmonochlorosilane

-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
995-82-4
1,1,3,3,5,5,7,7,9,9,11,11-dodecamethylhexasiloxane

| Conditions | Yield |
|---|---|
| With water; sulfur dioxide Ambient temperature; | 78% |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
1152310-87-6
1,3-bis(3-azidopropyl)-1,1,3,3-tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 20 - 80℃; for 20.25h; | 77% |

Octamethylcyclotetrasiloxane Chemical Properties
The Molecular Structure of Octamethylcyclotetrasiloxane (CAS NO.556-67-2):
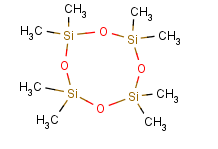
Empirical Formula: C8H24O4Si4
Molecular Weight: 296.6158
Nominal Mass: 296 Da
Average Mass: 296.6158 Da
Monoisotopic Mass: 296.075163 Da
Index of Refraction: 1.423
Molar Refractivity: 79.02 cm3
Molar Volume: 310 cm3
Surface Tension: 18.4 dyne/cm
Density: 0.95 g/cm3
Flash Point: 63.2 °C
Enthalpy of Vaporization: 39.44 kJ/mol
Boiling Point: 175 °C at 760 mmHg
Vapour Pressure: 1.57 mmHg at 25°C
Appearance: Colorless to brown liquid
IUPAC Name: 2,2,4,4,6,6,8,8-octamethyl-1,3,5,7,2,4,6,8-tetraoxatetrasilocane
Product Categories: Chloro;Silicon Compounds;Organics;Dichlorosilanes;Dichlorosilanes (for Polysilanes);Functional Materials;Reagent for High-Performance Polymer Research;Si (Classes of Silicon Compounds);Si-Cl Compounds;Silicon Compounds (for Synthesis);Synthetic Organic Chemistry;Chloro Silanes
Storage temp: Store at 0-5°C
Water Solubility: reacts
Sensitive: Moisture Sensitive
Stability: Stable. Reacts violently with water and alcohols. Highly flammable. Incompatible with strong oxidizing agents, water, alcohols, caustics, ammonia
Octamethylcyclotetrasiloxane Uses
Octamethylcyclotetrasiloxane (CAS NO.556-67-2) is commonly used as raw materials of organic silicon, also used in the electronics industry.
Octamethylcyclotetrasiloxane Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MLD | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,1230. | ||
| 2. | eye-rbt 500 mg/24H MLD | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,1230. |
Octamethylcyclotetrasiloxane Consensus Reports
Reported in EPA TSCA Inventory.
Octamethylcyclotetrasiloxane Safety Profile
A skin irritant. When heated to decomposition it emits acrid smoke and irritating vapors.
Hazard Codes:  Xi
Xi Xn
Xn F
F N
N
Risk Statements: 20-59-36/37/38-11-67-65-63-48/20-38-20/21-50/53
R20: Harmful by inhalation
R59: Dangerous to the ozone layer
R36/37/38: Irritating to eyes, respiratory system and skin
R11: Highly flammable
R67: Vapours may cause drowsiness and dizziness
R65: Harmful: may cause lung damage if swallowed
R63: Possible risk of harm to the unborn child
R38: Irritating to skin
R20/21: Harmful by inhalation and in contact with skin
R50/53: Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
Safety Statements: 60-61-62-36/37-59
S60: This material and its container must be disposed of as hazardous waste
S61: Avoid release to the environment. Refer to special instructions / safety data sheets
S62: If swallowed, do not induce vomitting; seek medical advice immediately and show this container or label
S36/37: Wear suitable protective clothing and gloves
S59: Refer to manufacturer / supplier for information on recovery / recycling
RIDADR: UN 2924 3/PG 2
WGK Germany: 3
F: 3-10-19-21
TSCA: Yes
HazardClass: 3
PackingGroup: II
HS Code: 29310095
Related Products
- Octamethylcyclotetrasiloxane
- 55667-43-1
- 55668-34-3
- 55668-46-7
- 5567-15-7
- 55672-83-8
- 55672-92-9
- 55674-63-0
- 55674-67-4
- 55676-21-6
- 55676-22-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View