-
Name
Propylthiouracil
- EINECS 200-103-2
- CAS No. 51-52-5
- Article Data17
- CAS DataBase
- Density 1.24 g/cm3
- Solubility 1.1 g/L in water
- Melting Point 218-220 °C(lit.)
- Formula C7H10N2OS
- Boiling Point 355.2 °C at 760 mmHg
- Molecular Weight 170.235
- Flash Point 168.6 °C
- Transport Information
- Appearance white crystalline powder
- Safety 36/37-45
- Risk Codes 22-40
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Uracil,6-propyl-2-thio- (7CI,8CI);2-Mercapto-4-hydroxy-6-n-propylpyrimidine;2-Mercapto-4-hydroxy-6-propylpyrimidine;2-Mercapto-6-propylpyrimidin-4-ol;2-Thio-4-oxo-6-propyl-1,3-pyrimidine;2-Thio-6-propyl-1,3-pyrimidin-4-one;6-Propyl-2-thio-2,4(1H,3H)pyrimidinedione;6-Propyl-2-thiouracil;6-Propylthiouracil;6-n-Propyl-2-thiouracil;6-n-Propylthiouracil;NSC 6498;NSC 70461;PTU (thyreostatic);Procasil;Propacil;Propycil;Propyl-Thiorist;Propyl-Thyracil;Propylthiorit;Propylthiouracil;Prothiucil;Prothiurone;Prothycil;Prothyran;Protiural;Thiuragyl;Thyreostat II;
- PSA 80.74000
- LogP 1.38500
Synthetic route

| Conditions | Yield |
|---|---|
| Reflux; | 81% |
| With potassium hydroxide In ethanol at 80℃; for 5h; | 41% |
| With sodium ethanolate |

| Conditions | Yield |
|---|---|
| With potassium carbonate In water at 70 - 105℃; for 3h; | 78% |
| With sodium ethanolate In ethanol at 100℃; Inert atmosphere; | 33% |
| Heating; |

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 94% |

-
-
51-52-5
propylthiouracil

-
-
14542-93-9
1,1,3,3-tetramethylbutane isonitrile

-
-
762-42-5
dimethyl acetylenedicarboxylate

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 93% |

-
-
51-52-5
propylthiouracil

-
-
119072-54-7, 2769-71-3
2,6-dimethylphenyl isonitrile

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 93% |

-
-
51-52-5
propylthiouracil

-
-
119072-54-7, 2769-71-3
2,6-dimethylphenyl isonitrile

-
-
762-42-5
dimethyl acetylenedicarboxylate

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 92% |

-
-
51-52-5
propylthiouracil

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
79-11-8
chloroacetic acid

-
-
67-64-1
acetone

-
-
100-46-9
benzylamine

-
-
1379463-56-5
2-(N-benzyl-2-((6-oxo-4-propyl-1,6-dihydropyrimidin-2-yl)thio)acetamido)-N-(tert-butyl)-2-methylpropanamide

| Conditions | Yield |
|---|---|
| Stage #1: propylthiouracil; chloroacetic acid at 20℃; for 0.5h; Ionic liquid; Stage #2: acetone; benzylamine at 20℃; for 0.25h; Ugi condensation; Ionic liquid; Stage #3: tert-butylisonitrile at 20℃; for 5h; Ugi condensation; Ionic liquid; | 90% |

-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
79-11-8
chloroacetic acid

-
-
67-64-1
acetone

-
-
100-46-9
benzylamine

-
-
1379463-51-0
2-(N-benzyl-2-((6-oxo-4-propyl-1,6-dihydropyrimidin-2-yl)thio)acetamido)-N-cyclohexyl-2-methylpropanamide

| Conditions | Yield |
|---|---|
| Stage #1: propylthiouracil; chloroacetic acid at 20℃; for 0.5h; Ionic liquid; Stage #2: acetone; benzylamine at 20℃; for 0.25h; Ugi condensation; Ionic liquid; Stage #3: Cyclohexyl isocyanide at 20℃; for 5h; Ugi condensation; Ionic liquid; | 89% |


| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide for 6h; Heating; Green chemistry; | 89% |


| Conditions | Yield |
|---|---|
| With laccase from Agaricus bisporus In aq. phosphate buffer; ethanol at 20℃; for 13h; pH=6; Green chemistry; Enzymatic reaction; chemoselective reaction; | 88% |
-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 87% |

-
-
51-52-5
propylthiouracil

-
-
448224-88-2
2-bromo-N-[4-(4-chloro-phenyl)-1,3-thiazol-2-yl]-acetamide

| Conditions | Yield |
|---|---|
| With potassium phosphate; sodium iodide In N,N-dimethyl-formamide at 60℃; for 1h; | 87% |
-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
4755-72-0, 39266-56-3
Chloro-phenyl-acetic acid

-
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide at 30℃; for 6h; Green chemistry; | 87% |

-
-
51-52-5
propylthiouracil

-
-
13345-08-9
6-propyl(1H,3H)pyrimidine-2,4-dione

| Conditions | Yield |
|---|---|
| With chloroacetic acid In water for 5h; Heating; | 86% |
| With chloroacetic acid | |
| With chloroacetic acid Heating; | |
| With chloroacetic acid |

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 20℃; for 0.166667h; | 85% |
-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
108-94-1
cyclohexanone

-
-
79-11-8
chloroacetic acid

-
-
100-46-9
benzylamine

-
-
1379463-54-3
1-(N-benzyl-2-((6-oxo-4-propyl-1,6-dihydropyrimidin-2-yl)thio)acetamido)-N-cyclohexylcyclohexanecarboxamide

| Conditions | Yield |
|---|---|
| Stage #1: propylthiouracil; chloroacetic acid at 20℃; for 0.5h; Ionic liquid; Stage #2: cyclohexanone; benzylamine at 20℃; for 0.25h; Ugi condensation; Ionic liquid; Stage #3: Cyclohexyl isocyanide at 20℃; for 5h; Ugi condensation; Ionic liquid; | 85% |

-
-
51-52-5
propylthiouracil

-
-
14542-93-9
1,1,3,3-tetramethylbutane isonitrile

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 85% |


| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 5h; | 85% |

-
-
51-52-5
propylthiouracil

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
79-11-8
chloroacetic acid

-
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide at 30℃; for 6h; Green chemistry; | 85% |

-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
75-04-7
ethylamine

-
-
79-11-8
chloroacetic acid

-
-
67-64-1
acetone

-
-
1379463-49-6
N-cyclohexyl-2-(N-ethyl-2-((6-oxo-4-propyl-1,6-dihydropyrimidin-2-yl)thio)acetamido)-2-methylpropanamide

| Conditions | Yield |
|---|---|
| Stage #1: propylthiouracil; chloroacetic acid at 20℃; for 0.5h; Ionic liquid; Stage #2: ethylamine; acetone at 20℃; for 0.25h; Ugi condensation; Ionic liquid; Stage #3: Cyclohexyl isocyanide at 20℃; for 5h; Ugi condensation; Ionic liquid; | 83% |

-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
79-11-8
chloroacetic acid

-
-
96-22-0
pentan-3-one

| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide at 30℃; for 6h; Green chemistry; | 83% |

-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
4755-72-0, 39266-56-3
Chloro-phenyl-acetic acid

-
-
96-22-0
pentan-3-one

| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide at 30℃; for 6h; Green chemistry; | 83% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20 - 40℃; for 4h; | 82% |
| With potassium hydroxide | |
| With sodium hydroxide In water at 20℃; |
-
-
51-52-5
propylthiouracil

-
-
37746-78-4
4-bromo-trans-crotonic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 1h; | 82% |
-
-
51-52-5
propylthiouracil

-
-
931-53-3
Cyclohexyl isocyanide

-
-
67-64-1
acetone

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide at 30℃; for 6h; Green chemistry; | 82% |

-
-
51-52-5
propylthiouracil

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
4755-72-0, 39266-56-3
Chloro-phenyl-acetic acid

-
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With octyl methyl imidazolium bromide at 30℃; for 6h; Green chemistry; | 81% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 81% |
-
-
51-52-5
propylthiouracil

-
-
123-31-9
hydroquinone

-
-
1611453-74-7
2-(2,5-dihydroxyphenylsulfanyl)-6-propyl-1H-pyrimidin-4-one

| Conditions | Yield |
|---|---|
| In aq. phosphate buffer; water; N,N-dimethyl-formamide at 25℃; pH=6; Michael Addition; Electrolysis; | 80% |

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 5h; | 80% |

-
-
51-52-5
propylthiouracil

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
75-04-7
ethylamine

-
-
79-11-8
chloroacetic acid

-
-
67-64-1
acetone

-
-
1379463-57-6
N-(tert-butyl)-2-(N-ethyl-2-((6-oxo-4-propyl-1,6-dihydropyrimidin-2-yl)thio)acetamido)-2-methylpropanamide

| Conditions | Yield |
|---|---|
| Stage #1: propylthiouracil; chloroacetic acid at 20℃; for 0.5h; Ionic liquid; Stage #2: ethylamine; acetone at 20℃; for 0.25h; Ugi condensation; Ionic liquid; Stage #3: tert-butylisonitrile at 20℃; for 5h; Ugi condensation; Ionic liquid; | 79% |

-
-
51-52-5
propylthiouracil

-
-
38838-06-1
methyl 5-deoxy-5-iodo-2,3-isopropylidene-β-D-ribofuranoside

-
-
676557-71-4
methyl 5-deoxy-2,3-O-isopropylidene-5-(6-propyluracil-2-yl)thio-β-D-ribofuranoside

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 60℃; for 1h; | 78% |
Propylthiouracil Consensus Reports
Propylthiouracil Specification
The Propylthiouracil, with the CAS registry number 51-52-5, is also known as 6-n-Propylthiouracil. It belongs to the product categories of Pyridines, Pyrimidines, Purines and Pteredines; Intermediates & Fine Chemicals; Pharmaceuticals; API's. Its EINECS registry number is 200-103-2. This chemical's molecular formula is C7H10N2OS and molecular weight is 170.2321. Its IUPAC name is called 6-propyl-2-sulfanylidene-1H-pyrimidin-4-one. What's more, this chemical's classification codes are Antimetabolites; Antithyroid Agents; Drug / Therapeutic Agent; Hormone Antagonists; Hormones, Hormone Substitutes, and Hormone Antagonists; Human Data; Mutation Data; Noxae; Reproductive Effect; Thyroid Inhibitor; Tumor Data. Meanwhile, it can be used as anti-thyroid drugs and in biochemical research. Propylthiouracil is a thioamide drug used to treat hyperthyroidism (including Graves' disease) by decreasing the amount of thyroid hormone produced by the thyroid gland.
Physical properties of Propylthiouracil: (1)ACD/LogP: 1.37; (2)ACD/LogD (pH 5.5): 1.37; (3)ACD/LogD (pH 7.4): 1.3; (4)ACD/BCF (pH 5.5): 6.49; (5)ACD/BCF (pH 7.4): 5.48; (6)ACD/KOC (pH 5.5): 132.69; (7)ACD/KOC (pH 7.4): 111.93; (8)#H bond acceptors: 3; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 2; (11)Index of Refraction: 1.596; (12)Molar Refractivity: 46.62 cm3; (13)Molar Volume: 137 cm3; (14)Surface Tension: 54.9 dyne/cm; (15)Density: 1.24 g/cm3; (16)Flash Point: 168.6 °C; (17)Enthalpy of Vaporization: 62.4 kJ/mol; (18)Boiling Point: 355.2 °C at 760 mmHg; (19)Vapour Pressure: 1.56E-05 mmHg at 25°C.
Preparation of Propylthiouracil: this chemical can be prepared by β-oxo-hexanoate with thiourea via condensation reaction. Meanwhile, it can also be prepared by butyryl ethyl acetate and thiourea via condensation reaction.
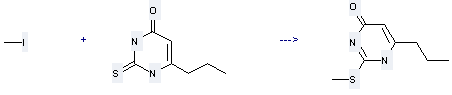
Uses of Propylthiouracil: it can be used to produce 2-methylsulfanyl-6-propyl-1H-pyrimidin-4-one with iodomethane at temperature of 60 °C. This reaction will need reagent aq. NaOH and solvent ethanol. The yield is about 55%.
When you are using this chemical, please be cautious about it. This chemical is harmful and irritant. Harmful if swallowed. Whenever you will contact it, please wear suitable protective clothing and gloves. In case of accident or if you feel unwell seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: CCCC1=CC(=O)NC(=S)N1
(2)InChI: InChI=1S/C7H10N2OS/c1-2-3-5-4-6(10)9-7(11)8-5/h4H,2-3H2,1H3,(H2,8,9,10,11)
(3)InChIKey: KNAHARQHSZJURB-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| child | TDLo | oral | 165mg/kg/2W-I (165mg/kg) | LIVER: "HEPATITIS (HEPATOCELLULAR NECROSIS), DIFFUSE" | Drug Intelligence and Clinical Pharmacy. Vol. 19, Pg. 669, 1985. |
| child | TDLo | oral | 900mg/kg/60D- (900mg/kg) | LIVER: "HEPATITIS (HEPATOCELLULAR NECROSIS), DIFFUSE" LIVER: "JAUNDICE, OTHER OR UNCLASSIFIED" | Annals of Internal Medicine. Vol. 82, Pg. 228, 1975. |
| child | TDLo | oral | 1785mg/kg/17W (1785mg/kg) | LIVER: "JAUNDICE, OTHER OR UNCLASSIFIED" KIDNEY, URETER, AND BLADDER: OTHER CHANGES IN URINE COMPOSITION | Journal of Pediatric Gastroenterology and Nutrition. Vol. 7, Pg. 776, 1988. |
| man | TDLo | oral | 42857ug/kg/10 (42.857mg/kg) | KIDNEY, URETER, AND BLADDER: "CHANGES IN TUBULES (INCLUDING ACUTE RENAL FAILURE, ACUTE TUBULAR NECROSIS)" KIDNEY, URETER, AND BLADDER: INTERSTITIAL NEPHRITIS KIDNEY, URETER, AND BLADDER: URINE VOLUME DECREASED | American Journal of Kidney Diseases. Vol. 24, Pg. 575, 1994. |
| man | TDLo | oral | 116mg/kg/11W- (116mg/kg) | LIVER: OTHER CHANGES | Archives of Internal Medicine. Vol. 135, Pg. 319, 1975. |
| man | TDLo | oral | 257mg/kg (257mg/kg) | PERIPHERAL NERVE AND SENSATION: FLACCID PARALYSIS WITHOUT ANESTHESIA (USUALLY NEUROMUSCULAR BLOCKAGE) | Postgraduate Medical Journal. Vol. 73, Pg. 445, 1997. |
| mouse | LDLo | unreported | 750ug/kg (0.75mg/kg) | Drugs in Japan Vol. 6, Pg. 732, 1982. | |
| rat | LD50 | oral | 1250mg/kg (1250mg/kg) | Nature. Vol. 157, Pg. 659, 1946. | |
| rat | LDLo | intraperitoneal | 400mg/kg (400mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 97, Pg. 478, 1949. | |
| rat | LDLo | unreported | 750ug/kg (0.75mg/kg) | Drugs in Japan Vol. 6, Pg. 732, 1982. | |
| women | LDLo | oral | 84mg/kg/2W-I (84mg/kg) | LIVER: "HEPATITIS (HEPATOCELLULAR NECROSIS), DIFFUSE" | Archives of Internal Medicine. Vol. 142, Pg. 838, 1982. |
| women | LDLo | oral | 480mg/kg/9W-I (480mg/kg) | VASCULAR: OTHER CHANGES BLOOD: AGRANULOCYTOSIS | Southern Medical Journal. Vol. 75, Pg. 1297, 1982. |
| women | LDLo | unreported | 6mg/kg (6mg/kg) | VASCULAR: SHOCK BLOOD: CHANGES IN SPLEEN LIVER: "HEPATITIS (HEPATOCELLULAR NECROSIS), ZONAL" | New England Journal of Medicine. Vol. 249, Pg. 814, 1953. |
| women | TDLo | oral | 693mg/kg/11W- (693mg/kg) | LIVER: "HEPATITIS, FIBROUS (CIRRHOSIS, POST-NECROTIC SCARRING)" LIVER: "JAUNDICE, OTHER OR UNCLASSIFIED" | Southern Medical Journal. Vol. 87, Pg. 943, 1994. |
| women | TDLo | oral | 900mg/kg/21W- (900mg/kg) | LIVER: OTHER CHANGES | Archives of Internal Medicine. Vol. 140, Pg. 1184, 1980. |
Related Products
- Propylthiouracil
- 5152-54-5
- 51527-18-5
- 51527-19-6
- 51527-58-3
- 51527-73-2
- 51527-97-0
- 51528-02-0
- 51528-20-2
- 51528-22-4
- 51529-01-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View