-
Name
PROPARGYLALDEHYDE DIETHYL ACETAL
- EINECS 233-430-4
- CAS No. 10160-87-9
- Article Data30
- CAS DataBase
- Density 0.909 g/cm3
- Solubility Slightly soluble in water.
- Melting Point
- Formula C7H12O2
- Boiling Point 139 °C at 760 mmHg
- Molecular Weight 128.171
- Flash Point 32.2 °C
- Transport Information
- Appearance Clear light yellow liquid
- Safety 16-26-36
- Risk Codes 10-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Propiolaldehyde diethyl acetal;Propyne, 3,3-diethoxy-;1,1-Diethoxyprop-2-yne;2-Propyn-1-al diethyl acetal;3,3-Diethoxy-1-propyne;3,3-Diethoxypropyne;NSC 45019;Propynaldiethyl acetal;
- PSA 18.46000
- LogP 1.01870
Synthetic route
-
-
10160-86-8
2,3-dibromo-1,1-diethoxypropane

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With potassium hydroxide; tetraoctyl ammonium bromide In Petroleum ether at 80℃; for 6h; | 79% |
| With ammonia; sodium amide In diethyl ether at -34℃; for 0.25h; | 75% |
| With ammonia; sodium amide at -30℃; | 75% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride In dichloromethane Ambient temperature; | 70% |
-
-
64-17-5
ethanol

-
-
40745-72-0
1-bromo-2,2-dichlorocyclopropane

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride In dichloromethane Ambient temperature; | 57% |

| Conditions | Yield |
|---|---|
| With sulfur dioxide; calcium chloride |
-
-
408320-07-0
2,2-dibromo-propionaldehyde diethylacetal

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; tert-butyl alcohol at 100℃; |
-
-
32798-36-0
6,6-diethoxy-3-methyl-hex-4-yn-3-ol

-
-
124-41-4
sodium methylate

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| at 150℃; under 20 Torr; |
-
-
122-51-0
orthoformic acid triethyl ester

-
-
74-86-2
acetylene

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| (i) EtMgBr, (ii) /BRN= 605384/; Multistep reaction; | |
| beim Behandeln von Acetylen-monomagnesiumbromid; |
-
-
408320-07-0
2,2-dibromo-propionaldehyde diethylacetal

-
-
865-47-4
potassium tert-butylate

-
-
75-65-0
tert-butyl alcohol

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
22834-33-9
3-(1-ethoxy-prop-2-ynyloxy)-propenal

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| (heating); |
-
-
122-51-0
orthoformic acid triethyl ester

-
-
107-02-8
acrolein

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| (i) (bromination), (ii) /BRN= 605384/, (iii) NaNH2, liq. NH3; Multistep reaction; |
-
-
593-61-3
bromoethyne

-
-
14444-77-0
diethyl phenyl orthoformate

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With magnesium 1.) THF, 2.) THF, RT; Yield given. Multistep reaction; |

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 160℃; |
-
-
122-51-0
orthoformic acid triethyl ester

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With diethyl ether |

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With potassium hydroxide |
-
-
10160-86-8
2,3-dibromo-1,1-diethoxypropane

-
A
-
17592-40-4
2-bromo-3,3-diethoxypropene

-
B
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
107-19-7
propargyl alcohol

-
-
122-51-0
orthoformic acid triethyl ester

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; In(OSO2CF3)3 In chloroform for 42h; Heating; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 19.0 g / ZnCl2 / aq. ethanol / 1 h / 15 °C 2: 75 percent / NaNH2; NH3 / diethyl ether / 0.25 h / -34 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 80 percent / HCl gas, TsOH / ethanol / 70 Torr 2: 1.) Mg / 1.) THF, 2.) THF, RT View Scheme |

-
-
122-51-0
orthoformic acid triethyl ester

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: NaNH2, liq. NH3 View Scheme |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 82℃; for 10h; Time; Large scale; | 60 kg |
-
-
108-29-2
5-methyl-dihydro-furan-2-one

-
A
-
106-98-9
1-butylene

-
B
-
10160-87-9
Propiolaldehyde diethyl acetal

-
C
-
541-47-9
3-Methylbutenoic acid

-
D
-
930-56-3
1-(2-methyl-cyclopropyl)-ethanone

| Conditions | Yield |
|---|---|
| With niobium(v) loaded on MFI-type zeolite at 320℃; under 760.051 Torr; Reagent/catalyst; |

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
78-84-2
isobutyraldehyde

-
-
86517-10-4
6,6-Diethoxy-2-methyl-hex-4-yn-3-ol

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; cyclohexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: isobutyraldehyde In tetrahydrofuran; cyclohexane at -78℃; for 3h; Stage #3: With hydrogenchloride In tetrahydrofuran; cyclohexane; water | 100% |
| With n-butyllithium In diethyl ether; hexane for 2h; Ambient temperature; | 55% |
-
-
73568-25-9
2-chloro-3-quinoline carboxaldehyde

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
872596-83-3
2-(3,3-diethoxy-prop-1-ynyl)-quinoline-3-carbaldehyde

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride In N,N-dimethyl-formamide at 20℃; Sonogashira reaction; | 100% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
625-99-0
3-iodochlorobenzene

-
-
1354931-76-2
1-chloro-3-(3,3-diethoxyprop-1-ynyl)benzene

| Conditions | Yield |
|---|---|
| With triethylamine; copper(l) iodide; [1,4-bis(diphenylphosphino)butane] palladium(ll) dichloride at 20℃; for 4h; Sonogashira Coupling; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 4h; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 25℃; for 4h; Sonogashira Cross-Coupling; Inert atmosphere; |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
16088-62-3
(S)-Propylene oxide

-
-
141207-06-9
(+)-6,6-diethoxy-4-hexyn-2-ol

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide; hexane at -30 - 20℃; for 4h; | 99% |
| With N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium In tetrahydrofuran; hexane for 0.5h; Ambient temperature; | 90.6% |
-
-
637-81-0
ethyl-2-azidoacetate

-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| Amberlyst A-21*CuI In dichloromethane at 20℃; for 12h; Huisgen cycloaddition; | 99% |
| With copper(II) loaded mesoporous SBA-15 In dichloromethane at 20℃; Huisgen Cycloaddition; regioselective reaction; | 66% |
| With copper(I) iodide adsorbed on Wyoming montmorillonite at 20℃; for 18h; Huisgen's cycloaddition; Neat (no solvent); |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
72320-38-8
3-azidopropan-1-ol

| Conditions | Yield |
|---|---|
| Amberlyst A-21*CuI In dichloromethane at 20℃; for 12h; Huisgen cycloaddition; | 99% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
622-79-7
benzyl azide

-
-
133735-81-6
1-benzyl-4-(diethoxymethyl)-1H-1,2,3-triazole

| Conditions | Yield |
|---|---|
| Amberlyst A-21*CuI In dichloromethane at 20℃; for 12h; Huisgen cycloaddition; | 99% |
| With Amberlyst A-21*copper(I) iodide In dichloromethane at 20℃; for 12h; Huisgen reaction; Automated synthesizer; Combinatorial reaction / High throughput screening (HTS); regioselective reaction; | 99% |
| With copper(ll) sulfate pentahydrate; sodium L-ascorbate In dichloromethane; water at 20℃; for 12h; | 98% |
-
-
5332-25-2
6-bromoquinoline

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1197377-23-3
6-(3,3-diethoxyprop-1-ynyl)quinoline

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; triphenylphosphine; palladium diacetate In N,N-dimethyl-formamide at 90℃; Inert atmosphere; | 99% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1224697-06-6
4-diethoxymethyl-1-(3-trifluoroacetamidopropyl)-1,2,3-triazole

| Conditions | Yield |
|---|---|
| With Amberlyst A-21*copper(I) iodide In dichloromethane at 20℃; for 12h; Huisgen reaction; Automated synthesizer; Combinatorial reaction / High throughput screening (HTS); regioselective reaction; | 99% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1449566-55-5
(S)-2-(tert-butyldiphenylsilyloxy)tetradecanal

-
-
1449521-29-2
(S)-5-(tert-butyldiphenylsilyloxy)-1,1-diethoxyheptadec-2-yn-4-ol

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -28 - 0℃; for 1h; Stage #2: (S)-2-(tert-butyldiphenylsilyloxy)tetradecanal In tetrahydrofuran; hexane at -78 - 20℃; for 1.5h; | 99% |
-
-
455-19-6
4-Trifluoromethylbenzaldehyde

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1539295-60-7
4,4-diethoxy-1-(4-(trifluoromethyl)phenyl)but-2-yn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 4-Trifluoromethylbenzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 99% |
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: 4-Trifluoromethylbenzaldehyde In tetrahydrofuran; hexane at 25℃; for 16h; | 79% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
19277-54-4
p-chlorophenyldiazomethane

-
-
96251-77-3
1-chloro-4-(4,4-diethoxybuta-1,2-dien-1-yl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With copper(l) iodide; triethylamine In 1,4-dioxane for 0.166667h; Flow reactor; Stage #2: p-chlorophenyldiazomethane In 1,4-dioxane; dichloromethane at 20℃; for 0.166667h; Flow reactor; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; Inert atmosphere; Stage #2: C19H29FN3OP In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; diastereoselective reaction; | 98% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1094159-79-1
N,N′-bis[6-(3,3-dimethylbutyrylamino)pyridin-2-yl]-5-iodoisophthalamide

-
-
1239955-22-6
5-(3,3-diethoxyprop-1-ynyl)-N1,N3-bis(6-(3,3-dimethylbutyrylamino)pyridin-2-yl)isophthalamide

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; diisopropylamine In tetrahydrofuran at 20℃; for 20h; Sonogashira coupling; | 98% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1443233-72-4
C23H48O5Si2

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -78 - 0℃; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; Inert atmosphere; Stage #2: C19H30N3OP In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; diastereoselective reaction; | 97% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
167971-82-6
(4S)-4-tert-butoxy-3,4-dihydro-2H-pyrrole 1-oxide

-
-
1255710-21-4
C15H27NO4

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; triethylamine In acetonitrile at 0 - 20℃; for 24h; Kinugasa reaction; Inert atmosphere; | 97% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); caesium carbonate; XPhos In tetrahydrofuran at 60℃; for 5.5h; Sonogashira coupling; Inert atmosphere; Sealed tube; stereoselective reaction; | 97% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1123-56-4
2,6-dimethylbenzaldehyde

-
-
1539295-63-0
1-(2,6-dimethylphenyl)-4,4-diethoxybut-2-yn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 2,6-dimethylbenzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 97% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
2101-86-2
p-methylazidobenzene

-
-
113934-25-1
1-(4-methylphenyl)-1H-1,2,3-triazole-4-carboxaldehyde

| Conditions | Yield |
|---|---|
| With copper(II) sulfate; sodium L-ascorbate In water; tert-butyl alcohol at 70℃; for 24h; Solvent; | 97% |
-
-
600-00-0
ethyl 2-bromoisobutyrate

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
1071146-03-6
non-1-en-3-yn-2-ylbenzene

| Conditions | Yield |
|---|---|
| With copper(I) thiophene-2-carboxylate; C45H50N3O2P; caesium carbonate In diethyl ether at 20℃; for 24h; Schlenk technique; Inert atmosphere; enantioselective reaction; | 97% |

-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
51985-30-9, 137845-37-5, 144381-72-6
4-Methyl-N-[(E)-3-phenyl-prop-2-en-(E)-ylidene]-benzenesulfonamide

-
-
233770-16-6
(E)-N-(4,4'-diethoxy-1-styrylbut-2-ynyl)-4-toluenesulfonamide

| Conditions | Yield |
|---|---|
| With n-butyllithium; zinc(II) chloride In tetrahydrofuran; hexane 1.) -78 deg C, 30 min; 2.) Et2O, -78 deg C, 30 min then 0 deg C, 5 h; | 96% |
| With n-butyllithium; zinc(II) chloride In diethyl ether at -78℃; Yield given; |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
610-97-9
o-iodo-methyl-benzoic acid

-
-
181419-89-6
1-(2-methoxycarbonylphenyl)-3,3-diethoxyprop-1-yne

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal; o-iodo-methyl-benzoic acid With trans-bis(triphenylphosphine)palladium dichloride; triethylamine at 20℃; for 0.166667h; Sonogashira Cross-Coupling; Inert atmosphere; Stage #2: With copper(l) iodide at 50℃; for 4h; Sonogashira Cross-Coupling; Inert atmosphere; | 96% |
| With copper(l) iodide; bis(acetato)bis(triphenylphosphine)palladium(0); diethylamine In N,N-dimethyl-formamide for 2h; Ambient temperature; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; Inert atmosphere; Stage #2: C19H29BrN3OP In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; diastereoselective reaction; | 96% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
100-10-7
4-dimethylamino-benzaldehyde

-
-
1314664-65-7
C16H23NO3

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at 0℃; Inert atmosphere; Stage #2: 4-dimethylamino-benzaldehyde In tetrahydrofuran; hexane at 0℃; Inert atmosphere; | 96% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
1539295-61-8
4,4-diethoxy-1-(4-methoxyphenyl)but-2-yn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 96% |
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: 4-methoxy-benzaldehyde In tetrahydrofuran; hexane at 25℃; for 16h; | 96% |

| Conditions | Yield |
|---|---|
| With copper(I) thiophene-2-carboxylate; C55H70N3O2P; caesium carbonate In diethyl ether at 20℃; for 36h; Inert atmosphere; Schlenk technique; enantioselective reaction; | 96% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
100-52-7
benzaldehyde

-
-
53366-79-3
(+/-)-4-hydroxy-4-phenylbut-2-ynal diethyl acetal

| Conditions | Yield |
|---|---|
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: benzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 95% |
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: benzaldehyde In tetrahydrofuran; hexane at 25℃; for 16h; | 95% |
| Stage #1: Propiolaldehyde diethyl acetal With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: benzaldehyde In tetrahydrofuran; hexane at -78 - -40℃; for 3h; | 94% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
157200-04-9
4-methyl-6-phenyl-3-trifluoromethanesulfonylpyridazine

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; diisopropylamine In tetrahydrofuran for 3h; Ambient temperature; | 95% |
-
-
10160-87-9
Propiolaldehyde diethyl acetal

-
-
216005-94-6
{2-[(R)-1-(3,4-Dimethoxy-phenyl)-3-iodo-4-oxo-cyclohex-2-enyl]-ethyl}-methyl-carbamic acid methyl ester

-
-
216005-95-7
{2-[(R)-3-(3,3-Diethoxy-prop-1-ynyl)-1-(3,4-dimethoxy-phenyl)-4-oxo-cyclohex-2-enyl]-ethyl}-methyl-carbamic acid methyl ester

| Conditions | Yield |
|---|---|
| With diisopropylamine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In tetrahydrofuran at 0℃; for 0.666667h; | 95% |
Propynal diethyl acetal Specification
The 1-Propyne,3,3-diethoxy-, with the CAS registry number 10160-87-9, is also known as 3,3-diethoxypropyne. Its EINECS number is 233-430-4. It belongs to the product categories of Acetylenes; Functionalized Acetylenes. This chemical's molecular formula is C7H12O2 and molecular weight is 128.17. What's more, its systematic name is 3,3-Diethoxyprop-1-yne. Its storage temperature is 2-8 °C.
Physical properties of 1-Propyne,3,3-diethoxy- are: (1)ACD/LogP: 1.67; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.67; (4)ACD/LogD (pH 7.4): 1.67; (5)ACD/BCF (pH 5.5): 10.88; (6)ACD/BCF (pH 7.4): 10.88; (7)ACD/KOC (pH 5.5): 192.1; (8)ACD/KOC (pH 7.4): 192.1; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 18.46 Å2; (13)Index of Refraction: 1.421; (14)Molar Refractivity: 35.75 cm3; (15)Molar Volume: 140.9 cm3; (16)Polarizability: 14.17×10-24 cm3; (17)Surface Tension: 28.7 dyne/cm; (18)Density: 0.909 g/cm3; (19)Flash Point: 32.2 °C; (20)Enthalpy of Vaporization: 36.07 kJ/mol; (21)Boiling Point: 139 °C at 760 mmHg; (22)Vapour Pressure: 8.16 mmHg at 25 °C.
Preparation: this chemical can be prepared by 2,3-dibromo-1,1-diethoxy-propane at the temperature of 80 °C. This reaction will need reagent solid potassium hydroxyde and solvent petroleum ether with the reaction time of 6 hours. This reaction will also need catalyst tetraoctylammonium bromide. The yield is about 79%.

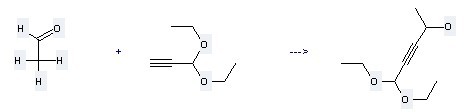
Uses of 1-Propyne,3,3-diethoxy-: it can be used to produce 5,5-diethoxy-pent-3-yn-2-ol at the ambient temperature. It will need reagents acetaldehyde, BuLi and solvents diethyl ether, hexane with the reaction time of 2 hours. The yield is about 70%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. It is flammable, so you should keep it away from sources of ignition - No smoking. It is in case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)SMILES: C#CC(OCC)OCC
(2)InChI: InChI=1/C7H12O2/c1-4-7(8-5-2)9-6-3/h1,7H,5-6H2,2-3H3
(3)InChIKey: RGUXEWWHSQGVRZ-UHFFFAOYAB
Related Products
- Propynal diethyl acetal
- 101-61-1
- 10161-33-8
- 10161-34-9
- 1016167-62-6
- 101622-50-8
- 101622-51-9
- 1016241-62-5
- 1016241-80-7
- 1016241-87-4
- 101625-35-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View