-
Name
Rhodanine
- EINECS 205-505-1
- CAS No. 141-84-4
- Article Data33
- CAS DataBase
- Density 1.59 g/cm3
- Solubility soluble in hot water
- Melting Point 165-169 °C(lit.)
- Formula C3H3NOS2
- Boiling Point 218.2 °C at 760 mmHg
- Molecular Weight 133.195
- Flash Point 85.7 °C
- Transport Information UN 3288
- Appearance light yellow to beige fine crystalline powder
- Safety 22-26-39
- Risk Codes 22-41
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 4-Oxo-2-thionothiazolidine;4-Oxo-2-thioxothiazolidine;4-Thiazolidinone-2-thione;NSC 1899;Rhodaninicacid;Rhodanine(6CI,8CI);2-Thio-4-ketothiazolidine;2-Thioxo-1,3-thiazolidin-4-one;2-Thioxo-4-thiazolidinone;4-Oxo-1,3-thiazolidine-2-thione;4-Oxo-2-thiazolidinethione;
- PSA 86.49000
- LogP 0.46310
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: carbon disulfide With ammonium hydroxide In water at 40℃; for 0.5h; Stage #2: sodium monochloroacetic acid In water for 0.416667 - 0.583333h; Stage #3: With hydrogenchloride In water at 100 - 110℃; | 94% |
| Stage #1: carbon disulfide With ammonia Stage #2: sodium monochloroacetic acid at 80℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium acetate; acetic acid In N,N-dimethyl-formamide | A 83% B n/a |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In toluene at 110℃; for 8h; Catalytic behavior; Solvent; Reagent/catalyst; Temperature; | 77% |
| With sulfuric acid In toluene at 80℃; for 20h; Temperature; |

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 90 - 95℃; | 70% |

| Conditions | Yield |
|---|---|
| With triethylamine In water; acetone for 0.5h; | 68% |
| With triethylamine In water; acetone for 0.5h; Product distribution; Mechanism; Ambient temperature; other N-substituted dithiocarbamates investigated; | 68% |

| Conditions | Yield |
|---|---|
| In water for 0.333333h; Reflux; | 45% |
-
-
14064-10-9
diethyl 2-chloromalonate

-
-
513-74-6
ammonium dithiocarbamate

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite |

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
29596-83-6
(thiocarbamoylthio)acetic acid

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| beim Erhitzen auf den Schmelzpunkt; | |
| With water | |
| With acid |

| Conditions | Yield |
|---|---|
| With ethanol; hydrogen sulfide Zufuegen von Salzsaeure und folgend Eindampfen; |
-
-
899417-89-1
thiocarbamoylsulfanyl-acetic acid ethyl ester

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With ethanol | |
| With water |
-
-
55602-07-8
diethyl 2-thiocyanatomalonate

-
-
98-91-9
thiobenzoic acid

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With benzene Erhitzen des Reaktionsprodukts mit wss. HCl; |

| Conditions | Yield |
|---|---|
| With ethanol at 160℃; im Rohr; |
-
-
90669-10-6
thiocarbamoylsulfanyl-acetic acid anilide

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
540-72-7
sodium thiocyanide

-
-
3926-62-3
sodium monochloroacetic acid

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With water anschliessendes Erwaermen mit wss. HCl; |
-
-
1147550-11-5
ammonium thiocyanate

-
-
3926-62-3
sodium monochloroacetic acid

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With water anschliessendes Erwaermen mit wss. HCl; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ethanol |

| Conditions | Yield |
|---|---|
| With water |
-
-
98664-36-9
4-N'-acetylphenylhydrazono-2-thiazolidinethione

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol for 2h; Heating; |
-
-
98664-37-0
3,4,6,8-Tetrahydro-2-phenyl-2H-thiazolo<3,4-c>-1,2,4-triazin-6-thioxo-4-one

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol for 2h; Heating; |
-
-
98664-36-9
4-N'-Acetylphenylhydrazono-2-thiazolidinethione

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol for 2h; Heating; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ethanol |

| Conditions | Yield |
|---|---|
| With water folgend Eintragen in konz. Salzsaeure bei 80-90grad oder in Salzsaeure bei 90-95grad; |
-
-
64-17-5
ethanol

-
-
6326-83-6
bis(carboxymethyl)trithiocarbonate

-
-
7664-41-7
ammonia

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| at 25℃; bei unterschiedlichem Mengenverhaeltnis der Reaktionspartner und unterschiedlicher Reaktionsdauer; |

-
-
7664-93-9
sulfuric acid

-
-
90669-10-6
thiocarbamoylsulfanyl-acetic acid anilide

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| Stage #1: bis(carboxymethyl)trithiocarbonate With 1,1'-carbonyldiimidazole In tetrahydrofuran at 20℃; for 1.5h; Stage #2: With ammonia In tetrahydrofuran for 4h; Reflux; |
-
-
3926-62-3
sodium monochloroacetic acid

-
-
513-74-6
ammonium dithiocarbamate

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 85 - 90℃; | |
| With hydrogenchloride In water at 85 - 90℃; |
-
-
24066-82-8
ethyl 2-isothiocyanatoacetate

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 120℃; for 6h; |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
104-55-2
3-phenyl-propenal

-
-
15328-87-7
5-(3-phenyl-allylidene)-2-thioxo-thiazolidin-4-on e

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |
| With thiourea; urea at 110℃; for 0.1h; Knoevenagel condensation; Neat (no solvent); | 91% |
| With sodium hydroxide |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
43111-11-1
5-formyl-8-methoxypsoralen

-
-
105245-75-8
4-<(4-Oxo-2-thioxo-1,3-thiazolidin-5-yl)-methyleno>xanthotoxin

| Conditions | Yield |
|---|---|
| With sodium acetate In acetic acid for 2h; Heating; | 100% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
4649-09-6
1H-pyrrolo[2,3-b]pyridin-3-carbaldehyde

-
-
937012-09-4
(Z)-5-(1H-pyrrolo-[2,3-b]pyridin-3-ylmethylene)-2-thioxo-1,3-thiazolidin-4-one

| Conditions | Yield |
|---|---|
| With sodium acetate In acetic acid for 12h; Heating / reflux; | 100% |
| With sodium acetate; acetic acid for 5h; Reflux; | 88% |
| With sodium acetate In acetic acid for 5h; Heating / reflux; | 88% |
| With piperidine In ethanol at 70℃; for 16h; Knoevenagel Condensation; | 73% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
872576-87-9
4-ethoxy-6-formyl-quinoline-3-carbonitrile

-
-
872576-94-8
4-ethoxy-6-[4-oxo-2-thioxo-thiazolidin-(5Z)-ylidenemethyl]-quinoline-3-carbonitrile

| Conditions | Yield |
|---|---|
| With ammonium acetate In toluene for 6h; Knoevenagel Condensation; Heating / reflux; | 100% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
879323-78-1
4-ethoxy-quinoline-6-carbaldehyde

-
-
879324-55-7
5-[1-(4-ethoxy-quinolin-6-yl)-meth-(Z)-ylidene]-2-thioxo-thiazolidin-4-one

| Conditions | Yield |
|---|---|
| With acetic acid for 12h; Heating / reflux; | 100% |
-
-
439811-22-0
quinazoline-6-carbaldehyde

-
-
141-84-4
2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid at 130℃; for 12h; | 100% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
66-77-3
1-naphthaldehyde

-
-
181765-58-2
(Z)-5-(naphthalen-1'-ylmethylene)-2-thioxothiazolidin-4-one

| Conditions | Yield |
|---|---|
| With acetic acid; triethylamine In ethyl acetate at 85℃; for 3h; | 100% |
| With sodium acetate; acetic acid for 16h; Reflux; | 82% |
| With sodium acetate In acetic acid Reflux; | 76% |
| With piperidine In ethanol at 70℃; for 16h; Knoevenagel Condensation; | 59% |

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid In toluene for 4h; Reflux; Dean-Stark; | 100% |
| In isopropyl alcohol at 80 - 90℃; for 2h; | 75% |
| With piperidine; acetic acid for 0.333333h; Knoevenagel Condensation; Microwave irradiation; |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
60125-23-7, 3541-42-2
2'-hydroxycinnamaldehyde

-
-
18097-60-4
C12H9NO2S2

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
1794-45-2
2-chlorocinnamaldehyde

-
-
15328-88-8
5-[3-(2-chloro-phenyl)-allylidene]-2-thioxo-thiazolidin-4-one

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
1039844-65-9
C14H10Br2O2

-
-
893449-38-2
5-[[5-bromo-2-[(2-bromophenyl)methoxy]phenyl]methylene]-2-thioxo-4-thiazolidinone

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid In toluene Reflux; | 100% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
100-52-7
benzaldehyde

-
-
3806-42-6
5-benzylidene-2-thioxothiazolidin-4-one

| Conditions | Yield |
|---|---|
| With thiourea; urea at 110℃; for 0.0833333h; Knoevenagel condensation; Neat (no solvent); | 99% |
| With urea/choline chloride eutectic salt at 80℃; for 0.75h; Catalytic behavior; Temperature; Aldol Condensation; | 96% |
| With tetra(n-butyl)ammonium hydroxide In ethanol; water at 50℃; for 1.5h; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid for 16h; Heating; | 99% |
| With ammonium acetate In toluene at 110℃; for 24h; Inert atmosphere; Darkness; | 94% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 1h; Reflux; stereoselective reaction; | 99% |
| With ammonium acetate; acetic acid for 1 - 12h; Heating / reflux; |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 150℃; Microwave irradiation; | 99% |
| With magnesia In ethanol; water at 50℃; for 1.66667h; Knoevenagel Condensation; | 78% |


| Conditions | Yield |
|---|---|
| With water at 20℃; for 12h; | 99% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
39755-95-8
5-methoxyisatine

| Conditions | Yield |
|---|---|
| With water at 20℃; for 12h; | 99% |
Rhodanine Chemical Properties
Molecular Formula: C3H3NOS2
Molar Mass: 133.19 g/mol
EINECS: 205-505-1
Density: 1.59 g/cm3
Flash Point: 85.7 °C
Index of Refraction: 1.711
Boiling Point: 218.2 °C at 760 mmHg
Vapour Pressure: 0.128 mmHg at 25 °C
Melting Point: 165-169 °C(lit.)
Water Solubility: soluble
Appearance: light yellow to beige fine crystalline powder
Product categories of Rhodanine (141-84-4): Miscellaneous;Thiazoles, Isothiazoles & Benzothiazoles
Structure of Rhodanine (CAS NO.141-84-4):
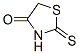
XLogP3-AA: 0.7
H-Bond Donor: 1
H-Bond Acceptor: 1
Systematic Name: 2-Thioxothiazolidin-4-one
SMILES: O=C1NC(=S)SC1
InChI: InChI=1/C3H3NOS2/c5-2-1-7-3(6)4-2/h1H2,(H,4,5,6)
InChIKey: KIWUVOGUEXMXSV-UHFFFAOYAP
Std. InChI: InChI=1S/C3H3NOS2/c5-2-1-7-3(6)4-2/h1H2,(H,4,5,6)
Std. InChIKey: KIWUVOGUEXMXSV-UHFFFAOYSA-N
Rhodanine Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MOD | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,202. | ||
| 2. | eye-rbt 750 µg/24H SEV | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,202. | ||
| 3. | orl-rat LD50:320 mg/kg | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,1093. | ||
| 4. | orl-mus LD50:225 mg/kg | FRZKAP Farmatsevtichnii Zhurnal (Kiev). 17 (1)(1962),36. | ||
| 5. | ipr-mus LD50:200 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD277-689 . | ||
| 6. | scu-mus LDLo:200 mg/kg | AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. 12 (1904),447. | ||
| 7. | ivn-mus LD50:32 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#04798 . |
Rhodanine Consensus Reports
Reported in EPA TSCA Inventory.
Rhodanine Safety Profile
Hazard Codes:  Xn
Xn
Risk Statements: 22-41
R22:Harmful if swallowed.
R41:Risk of serious damage to the eyes.
Safety Statements: 22-26-39
S22:Do not breathe dust.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S39:Wear eye / face protection.
RIDADR: 3288
WGK Germany: 3
RTECS: VI7700000
HazardClass: 6.1(a)
PackingGroup: II
HS Code: 29341000
Hazardous Substances Data: 141-84-4(Hazardous Substances Data)
Poison by ingestion, intraperitoneal, subcutaneous, and intravenous routes. A skin and severe eye irritant. May explode on rapid heating. When heated to decomposition it emits very toxic fumes of NOx and SOx.
Rhodanine Specification
Rhodanine (CAS NO.141-84-4) also can be called Rhodanine derivative ; 2-Thioxo-1,3-thiazolidin-4-one ; 4-Thiazolidinone, 2-thioxo- ; Rhodanic acid and 2-mercaptothiazolinone . Rhodanine (CAS NO.141-84-4) may explode on rapid heating. It is water soluble. Rhodanine may generate hydrogen sulfide with acids. It may generate heat and hydrogen gas with alkali metals, nitrides, hydrides, and other strong reducing agents.
Related Products
- Rhodanine
- Rhodanine-3-acetic acid
- 141849-62-9
- 141850-54-6
- 141852-65-5
- 141854-25-3
- 141-85-5
- 14185-99-0
- 14-18-6
- 14186-50-6
- 141-86-6
- 141871-02-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View