-
Name
Tributylamine
- EINECS 203-058-7
- CAS No. 102-82-9
- Article Data141
- CAS DataBase
- Density 0.792 g/cm3
- Solubility 0.386 g/L (25 °C) in water
- Melting Point -70 °C(lit.)
- Formula C12H27N
- Boiling Point 215.3 °C at 760 mmHg
- Molecular Weight 185.353
- Flash Point 63.3 °C
- Transport Information UN 2542 6.1/PG 2
- Appearance pale yellow liquid with an amine odour
- Safety 26-36/37-45-61-36/37/39-28A
- Risk Codes 22-23/24-38-51/53-23/24/25
-
Molecular Structure
-
Hazard Symbols
 T,
T, N
N
- Synonyms Tributylamine(7CI,8CI);N,N-Dibutyl-1-butanamine;Tri-n-butylamine;Tris-n-butylamine;Tributylamine;
- PSA 3.24000
- LogP 3.68870
Synthetic route

| Conditions | Yield |
|---|---|
| With polymethylhydrosiloxane; bis(1,5-cyclooctadiene)diiridium(I) dichloride In tetrahydrofuran at 50℃; for 5h; | 99% |
| With NiO doped titania In butan-1-ol Reagent/catalyst; Irradiation; | 93% |
| With hydrogen; nickel at 120 - 140℃; |

| Conditions | Yield |
|---|---|
| With C42H44ClN4P2Ru(1+)*Cl(1-); potassium tert-butylate; ammonium formate at 100℃; for 72h; | 95% |
| With 1-hydroxytetraphenylcyclopentadienyl(tetraphenyl-2,4-cyclopentadien-1-one)-μ-hydrotetracarbonyldiruthenium(II); ammonium chloride; potassium hydroxide at 140℃; for 39h; Sealed tube; Inert atmosphere; | 99 %Chromat. |

| Conditions | Yield |
|---|---|
| With BER-CuSO4 In methanol for 3h; Ambient temperature; | 93% |
| With Dimethylphenylsilane In tetrahydrofuran at 20℃; for 24h; Inert atmosphere; | 88% |
| With bis(tri-n-butyltin) In tetrahydrofuran at 50℃; for 1h; | 84% |
| With BER-CuSO4 In methanol for 3h; Ambient temperature; other tertiary amine N-oxides and aromatic N-oxides, var. temp. and time; |
-
-
109-74-0
propyl cyanide

-
-
111-87-5
octanol

-
-
71-36-3
butan-1-ol

-
A
-
102-82-9
tributyl-amine

-
B
-
41145-51-1
octyldibutylamine

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 92% |


| Conditions | Yield |
|---|---|
| With 5-methyl-1,3,4-thiadiazol-2-amine; potassium hydroxide In ethanol; water at 25℃; for 1h; | 85% |
| With sodium hydroxide; urea at 120℃; under 3102.97 Torr; for 40h; | 76.5% |
| With ammonia |

| Conditions | Yield |
|---|---|
| palladium at 160℃; for 5h; | A 8% B 85% |
-
-
109-74-0
propyl cyanide

-
-
71-36-3
butan-1-ol

-
-
143-08-8
nonyl alcohol

-
A
-
102-82-9
tributyl-amine

-
B
-
93658-58-3
1-Dibutylamino-nonan

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 85% |


| Conditions | Yield |
|---|---|
| With hydrogen at 200℃; | A 13% B 2% C 85% |


| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 84% |


| Conditions | Yield |
|---|---|
| With chlorohydridocarbonylbis(tricyclohexylphosphine)ruthenium(II) In toluene at 120℃; for 24h; Catalytic behavior; Reagent/catalyst; Schlenk technique; Inert atmosphere; | 82% |
| With aluminium trichloride | |
| With palladium/alumina at 199.84℃; under 760.051 Torr; Kinetics; |

| Conditions | Yield |
|---|---|
| With ruthenium trichloride; triphenylphosphine In tetrahydrofuran at 185℃; for 8h; | 81% |
-
-
109-73-9
N-butylamine

-
A
-
102-82-9
tributyl-amine

-
B
-
130716-87-9
N-butylidenebutylamine

-
C
-
111-92-2
dibutylamine

| Conditions | Yield |
|---|---|
| With sodium nitroprusside at 20℃; N-alkylation; | A 3% B 8% C 81% |
| Rh on carbon In water for 1h; microwave irradiation; | A 9 % Chromat. B 57 % Chromat. C 34 % Chromat. |


| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 80% |


| Conditions | Yield |
|---|---|
| With tetrabutylammonium tetrafluoroborate In acetone at 320℃; Product distribution; in a gas chromatograph; other temperature; various concentration of reagent; | A 77% B n/a |
-
-
109-74-0
propyl cyanide

-
-
112-53-8
1-dodecyl alcohol

-
-
71-36-3
butan-1-ol

-
A
-
102-82-9
tributyl-amine

-
B
-
13590-84-6
di-n-butyl-dodecylamine

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 76% |

-
-
927-77-5
n-propylmagnesium bromide

-
-
121238-82-2
tris(benzotriazol-1-ylmethyl)amine

-
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 1.) room temperature, 1 h, 2.) reflux, 5 h; | 75% |

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 75% |


| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 75% |

-
-
109-74-0
propyl cyanide

-
-
36653-82-4
1-Hexadecanol

-
-
71-36-3
butan-1-ol

-
A
-
102-82-9
tributyl-amine

-
B
-
5675-43-4
Di-n-butyl-hexadecylamin

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 75% |

-
-
109-74-0
propyl cyanide

-
-
112-72-1
1-Tetradecanol

-
-
71-36-3
butan-1-ol

-
A
-
102-82-9
tributyl-amine

-
B
-
198066-94-3
dibutyl-tetradecyl-amine

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 240℃; under 7600 Torr; | A n/a B 70% |

-
-
40950-22-9
1,4-dibromobicyclo<2.2.1>heptane

-
A
-
102-82-9
tributyl-amine

-
B
-
279-23-2
norbornene

-
C
-
13474-70-9
1-bromonorbornane

-
D
-
67773-54-0
bis(1-norbornyl)mercury

| Conditions | Yield |
|---|---|
| With tetra-n-butylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; at 25°C, at mercury cathode, at -1.9 V; | A 24% B 68% C 4% D 3% |
| With tetra-n-butylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; at 25°C, at mercury cathode, at -1.8 V; | A 25% B 52% C 5% D 16% |
| With tetra-n-butylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; at 25°C, at mercury cathode, at -1.7 V; | A 20% B 45% C 4% D 17% |
| With tetra-n-butylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; at 25°C, at mercury cathode, at -1.6 V; | A 10% B 33% C 2% D 27% |

-
-
13965-02-1, 14177-93-6, 15692-07-6
cis-dichlorobis(triethylphosphine)platinum(II)

-
-
1923-70-2
tetrabutylammonium perchlorate

-
A
-
106-98-9
1-butylene

-
-
16842-17-4, 20436-52-6, 89254-73-9
trans-chlorohydridobis(triethylphosphine)platinum(II)

-
C
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| In acetonitrile; benzene Electrolysis; preelectrolyzing solvents/TBAP (activated alumina in soln.), adding cis-(PtCl2(PEt3)2), electrolyzing at -2.1 V to near zero current (Ar, 3h); filtering, evapg., reduction to dryness, adding charcoal, extg. (benzene), filtering, concg., chromy. (alumina, benzene/hexane), evapg.; gas chromy., elem. anal.; | A n/a B 63.3% C n/a |
| In acetonitrile; benzene Electrolysis; preelectrolyzing solvents/TBAP, adding cis-(PtCl2(PEt3)2), electrolyzing at -2.1 V to near zero current (Ar, 3h); same products when benzonitrile (5 - 10 equiv.) was added; filtering, evapg., reduction to dryness, adding charcoal, extg. (benzene), filtering, concg., chromy. (alumina, benzene/hexane), evapg.; gas chromy., elem. anal.; |

-
-
109-74-0
propyl cyanide

-
A
-
102-82-9
tributyl-amine

-
B
-
15431-00-2
butyl-1-idenedi-n-butylamine

-
C
-
111-92-2
dibutylamine

| Conditions | Yield |
|---|---|
| With hydrogen at 260℃; Flow reactor; | A 13% B 23% C 61.5% |

-
-
75-04-7
ethylamine

-
-
109-73-9
N-butylamine

-
A
-
102-82-9
tributyl-amine

-
B
-
13360-63-9
N-ethylbutylamine

-
C
-
4444-68-2
N,N-diethylbutylamine

-
D
-
4458-33-7
di-n-butylethylamine

-
E
-
111-92-2
dibutylamine

-
F
-
109-89-7
diethylamine

-
G
-
121-44-8
triethylamine

| Conditions | Yield |
|---|---|
| With hydrogen at 200℃; under 6000.6 Torr; Reagent/catalyst; Temperature; | A n/a B 60.7% C n/a D n/a E n/a F n/a G n/a |

-
-
40950-21-8
1,4-diiodobicyclo<2.2.1>heptane

-
A
-
102-82-9
tributyl-amine

-
B
-
279-23-2
norbornene

-
C
-
18313-42-3
1,1'-binorbornane

-
D
-
67773-54-0
bis(1-norbornyl)mercury

| Conditions | Yield |
|---|---|
| With tetra-n-butylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; at 25°C, at mercury cathode, at -1.9 V; | A 31% B 60% C 1% D 12% |
| With tetra-n-butylammonium perchlorate In N,N-dimethyl-formamide Electrolysis; at 25°C, at mercury cathode, at -1.8 V; | A 23% B 46% C 1% D 27% |


| Conditions | Yield |
|---|---|
| With {[(PCy3)(CO)RuH]4(μ-O)(μ-OH)2}; 4-tert-Butylcatechol In chlorobenzene at 130℃; for 16h; Glovebox; Schlenk technique; Sealed tube; chemoselective reaction; | A n/a B 46% |
| With bis(triphenylphosphine)platinum(II) dichloride; tin(ll) chloride In benzene at 180℃; for 5h; stainless steel reactor, var. reag.: PtCl2(PhCN)2; | A 8 % Chromat. B 74 % Chromat. |
| With tris(triphenylphosphine)ruthenium(II) chloride In methanol at 180℃; for 7h; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
111-86-4
n-Octylamine

-
-
109-73-9
N-butylamine

-
A
-
102-82-9
tributyl-amine

-
B
-
4088-42-0
N-n-butyl-N-n-octylamine

-
C
-
1120-48-5
n-dioctylamine

| Conditions | Yield |
|---|---|
| With platinum-nickel nanoclusters on activated carbon; hydrogen at 190℃; under 760.051 Torr; Flow reactor; chemoselective reaction; | A 14.5% B 28% C 40.3% |

-
-
16842-17-4, 20436-52-6, 89254-73-9
trans-chlorohydridobis(triethylphosphine)platinum(II)

-
-
1923-70-2
tetrabutylammonium perchlorate

-
A
-
106-98-9
1-butylene

-
-
118831-46-2
trans-{PtH(CH2CN)(PEt3)2}

-
C
-
102-82-9
tributyl-amine

-
D
-
7440-06-4
platinum

| Conditions | Yield |
|---|---|
| In acetonitrile; benzene byproducts: H2; Electrolysis; electrochemically reducing trans-(PtH(Cl)(PEt3)2) at -2.1 V vs Ag/AgCl in CH3CN/C6H6 (5/2, v/v), terminating electrolysis at constancy of current; 36% of hydrido complex recovered; 31P NMR; | A n/a B 38% C n/a D n/a |


| Conditions | Yield |
|---|---|
| at 120℃; for 6h; | 100% |
| In toluene at 70℃; for 24h; | 92% |
| at 70 - 80℃; | 78% |
-
-
102-82-9
tributyl-amine

-
-
19929-29-4
tetramethyl (dichloromethylene)bisphosphonate

| Conditions | Yield |
|---|---|
| In acetonitrile at 50℃; for 4h; | 100% |
-
-
102-82-9
tributyl-amine

-
-
121151-57-3
tetramethyl (dibromomethylene)bisphosphonate

| Conditions | Yield |
|---|---|
| In acetonitrile at 50℃; for 6h; | 100% |
-
-
102-82-9
tributyl-amine

-
-
133918-67-9
P,P-Bis(1-methylethyl) P',P'-dimethyl (dichloromethylene)bisphosphonate

| Conditions | Yield |
|---|---|
| In acetonitrile at 60℃; for 4h; | 100% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 72h; Reflux; | 100% |
| In 1,2-dichloro-ethane at 40℃; for 6h; | 98% |
| With meta-dinitrobenzene In acetonitrile at 75℃; for 7h; | 90% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether at 0℃; for 0.5h; | 100% |
| With hydrogenchloride In water at 135℃; | |
| With hydrogenchloride In water at 20℃; for 0.333333h; | |
| With chloro-trimethyl-silane In tetrahydrofuran; methanol for 0.5h; Inert atmosphere; Schlenk technique; |
-
-
61-82-5
3(5)-amino-1,2,4-triazole

-
-
262609-07-4
diethyl (2,4,6-trifluoro-phenyl)-malonate

-
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| Stage #1: 3(5)-amino-1,2,4-triazole; diethyl (2,4,6-trifluoro-phenyl)-malonate; tributyl-amine at 150℃; for 2h; Stage #2: With sodium hydroxide In water; toluene at 20 - 30℃; for 1.25h; | 100% |

-
-
888487-33-0
2-deoxy-2-fluoro-6-L-glycero-β-1-phosphoryl-D-heptoglucopyranose

-
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| In ethanol Product distribution / selectivity; | 100% |

| Conditions | Yield |
|---|---|
| In acetonitrile for 48h; Inert atmosphere; Reflux; | 100% |
-
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| With palladium on carbon; hydrogen In methanol; ethyl acetate at 20℃; under 1125.11 Torr; for 24h; | 100% |
-
-
888487-32-9
2-deoxy-1-O-dibenzylphosphoryl-2-fluoro-3,4,6,7-tetra-O-pivaloyl-L-glycero-β-D-gluco-heptopyranose

-
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| Stage #1: 2-deoxy-1-O-dibenzylphosphoryl-2-fluoro-3,4,6,7-tetra-O-pivaloyl-L-glycero-β-D-gluco-heptopyranose With palladium 10% on activated carbon; hydrogen; triethylamine In ethanol; ethyl acetate under 750.075 Torr; for 12h; Stage #2: With tetra(n-butyl)ammonium hydroxide In water at 20℃; for 26h; Inert atmosphere; Stage #3: tributyl-amine Further stages; | 100% |
-
-
102-82-9
tributyl-amine

-
-
88284-48-4
2-(trimethylsilyl)phenyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile at 20℃; for 16h; | 100% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 12h; | 100% |

| Conditions | Yield |
|---|---|
| In ethanol at 82℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| NiO nanoparticles at 100℃; for 10h; | 99% |
| at 25℃; under 7500600 Torr; for 18h; | |
| at 20.05℃; Kinetics; |

| Conditions | Yield |
|---|---|
| In acetonitrile for 48h; Inert atmosphere; Reflux; | 99% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 40℃; for 68h; | 99% |
| In dichloromethane at 20℃; for 48h; | 85% |
| In acetonitrile at 50℃; for 48h; | 82% |
| In acetonitrile at 40℃; for 24h; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 82℃; for 33h; Solvent; Temperature; Menshutkin Reaction; | 98.9% |
| In acetonitrile for 24h; Heating; | 80% |
| With ethanol |

| Conditions | Yield |
|---|---|
| With manganese(III) oxide; oxygen In tetrahydrofuran at 140℃; under 3000.3 Torr; for 24h; Reagent/catalyst; Pressure; Solvent; Temperature; Autoclave; Green chemistry; | 98% |
| With pyridine; copper(l) chloride In acetonitrile at 100℃; under 7500.75 - 22502.3 Torr; for 24h; Catalytic behavior; Pressure; Reagent/catalyst; Temperature; Autoclave; | 70% |
| With Eosin Y In ethanol for 10h; Irradiation; | 36% |
| With manganese(IV) oxide In benzene | |
| With copper(I) oxide; oxygen In ethanol; N,N-dimethyl-formamide at 120℃; under 4500.45 Torr; for 8h; Autoclave; | 93 %Spectr. |
-
-
102-82-9
tributyl-amine

-
-
220205-59-4
[Bis-(diisopropoxy-phosphoryl)-methyl]-phosphonic acid diethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: [Bis-(diisopropoxy-phosphoryl)-methyl]-phosphonic acid diethyl ester With trimethylsilyl bromide In dichloromethane silylation; Heating; Stage #2: tributyl-amine deesterification; | 98% |
-
-
102-82-9
tributyl-amine

| Conditions | Yield |
|---|---|
| With cross-linked polyvinylpyrrolidone*N2O4; dinitrogen tetraoxide In dichloromethane for 3h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); tetrabutylammomium bromide; 18O-labeled water; zinc(II) oxide In dimethyl sulfoxide at 100℃; for 16h; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; acetonitrile for 48h; Reflux; | 98% |
| In chloroform; acetonitrile for 96h; Reflux; Inert atmosphere; Schlenk technique; | 95% |
-
-
102-82-9
tributyl-amine

-
-
1493-13-6
trifluorormethanesulfonic acid

-
-
82052-14-0
tributylammonium triflate salt

| Conditions | Yield |
|---|---|
| In dichloromethane at 0℃; for 0.0833333h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; acetonitrile for 48h; Reflux; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; acetonitrile for 48h; Reflux; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; acetonitrile for 48h; Reflux; | 98% |
-
-
102-82-9
tributyl-amine

-
-
1373621-35-2
5,10,15,20-tetrakis[3-(4-bromobutoxy)phenyl]porphyrin magnesium(II)

| Conditions | Yield |
|---|---|
| In chloroform; acetonitrile at 70℃; for 90h; Inert atmosphere; Darkness; | 97% |

| Conditions | Yield |
|---|---|
| In chloroform; acetonitrile Reflux; Inert atmosphere; Schlenk technique; | 97% |
Tributylamine Chemical Properties
:
Tributylamine Specification
The Tributylamine is an organic compound with the formula C12H27N. The IUPAC name of this chemical is N,N-dibutylbutan-1-amine. With the CAS registry number 102-82-9, it is also named as 1-butanamine, N,N-dibutyl-. The product's categories are Amines; C11 to C38; Nitrogen Compounds; Analytical Reagents for General Use; Puriss p.a. ACS; T-Z, Puriss p.a. ACS. Besides, it is pale yellow liquid with an amine odour, which should be stored in a cool and well-ventilated place. It is used as oil additives, solvents, pharmaceutical intermediates, pesticides, emulsifier, plasticizer, mineral flotation agent and surface active agent.
Physical properties about Tributylamine are:
(1)ACD/LogP: 4.85; (2)ACD/LogD (pH 5.5): 1.76; (3)ACD/LogD (pH 7.4): 2.37; (4)ACD/BCF (pH 5.5): 2.34; (5)ACD/BCF (pH 7.4): 9.55; (6)ACD/KOC (pH 5.5): 8.51; (7)ACD/KOC (pH 7.4): 34.7; (8)#H bond acceptors: 1; (9)#Freely Rotating Bonds: 9; (10)Polar Surface Area: 3.24 Å2; (11)Index of Refraction: 1.437; (12)Molar Refractivity: 61.36 cm3; (13)Molar Volume: 233.8 cm3; (14)Polarizability: 24.32×10-24cm3; (15)Surface Tension: 27.4 dyne/cm; (16)Density: 0.792 g/cm3; (17)Flash Point: 63.3 °C; (18)Enthalpy of Vaporization: 45.16 kJ/mol; (19)Boiling Point: 215.3 °C at 760 mmHg; (20)Vapour Pressure: 0.149 mmHg at 25°C.
Preparation of Tributylamine:
This chemical can be prepared by butyl-methyl-amine. This reaction will need catalyst palladium black. The reaction time is 5 hours with reaction temperature of 160 °C. The yield is about 85%.
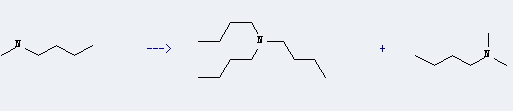
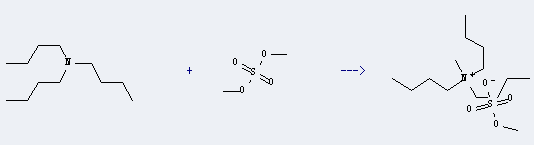
Uses of Tributylamine:
It can be used to produce methyl methyltributylammonium sulfate. The reaction time is 3 hours with reaction temperature of 80 °C. The yield is about 84.6%.
When you are using Tributylamine, please be cautious about it as the following:
It is harmful if swallowed and toxic by inhalation, in contact with skin and if swallowed. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. Besides, this chemical is irritating skin and toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. When you are using it, wear suitable gloves and eye/face protection and avoid release to the environment. Refer to special instructions/safety data sheet. After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer). In case of accident or if you feel unwell seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: N(CCCC)(CCCC)CCCC
(2)InChI: InChI=1/C12H27N/c1-4-7-10-13(11-8-5-2)12-9-6-3/h4-12H2,1-3H3
(3)InChIKey: IMFACGCPASFAPR-UHFFFAOYAY
(4)Std. InChI: InChI=1S/C12H27N/c1-4-7-10-13(11-8-5-2)12-9-6-3/h4-12H2,1-3H3
(5)Std. InChIKey: IMFACGCPASFAPR-UHFFFAOYSA-N
The toxicity data of Tributylamine is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 350mg/kg (350mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 42(12), Pg. 36, 1977. | |
| mammal (species unspecified) | LD50 | intraperitoneal | 107mg/kg (107mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: EXCITEMENT | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(11), Pg. 50, 1984. |
| mammal (species unspecified) | LD50 | oral | 888mg/kg (888mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: EXCITEMENT | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(11), Pg. 50, 1984. |
| mouse | LD50 | oral | 114mg/kg (114mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 42(12), Pg. 36, 1977. | |
| rabbit | LD50 | oral | 615mg/kg (615mg/kg) | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 42(12), Pg. 36, 1977. | |
| rabbit | LD50 | skin | 250uL/kg (0.25mL/kg) | Toxicology and Applied Pharmacology. Vol. 28, Pg. 313, 1974. | |
| rat | LCLo | inhalation | 75ppm/4H (75ppm) | Toxicology and Applied Pharmacology. Vol. 28, Pg. 313, 1974. | |
| rat | LD50 | oral | 114mg/kg (114mg/kg) | Trudy Leningradskogo Sanitarno-Gigienicheskogo Meditsinskogo Instituta. Vol. 115, Pg. 57, 1977. | |
| rat | LDLo | subcutaneous | 380mg/kg (380mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD BEHAVIORAL: COMA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Journal of Pharmacology and Experimental Therapeutics. Vol. 20, Pg. 435, 1923. |
Related Products
- Tributylamine
- 102-83-0
- 102830-75-1
- 102831-44-7
- 102831-92-5
- 102832-10-0
- 102832-11-1
- 1028343-10-3
- 10283-68-8
- 1028-38-2
- 102838-86-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View