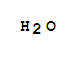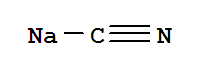This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| With acetic acid In water at 30℃; Microreactor technology; Continuous flow system; | 99% |
| With sodium metabisulfite In water at 20℃; for 0.5h; | |
| Stage #1: acetone With sodium hydrogen sulfite In water at 0℃; for 1h; Stage #2: potassium cyanide In water for 14h; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sulfuric acid; sodium cyanide In water at 10℃; for 1.5h; pH=7.3; | 95% |
| With pyridine | |
| With quinoline |
-
-
19355-69-2
2-amino-2-cyanopropane

-
A
-
18992-13-7
2-cyano-2-nitropropane

-
B
-
40561-27-1
1-cyano-1-methylethylnitrate

-
C
-
58102-56-0
tris-(1-cyano-1-methyl-ethyl)-hydroxylamine

-
D
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With perchloric acid; sodium nitrite at 25℃; Product distribution; var. α-aminonitriles; other conditions, also radical scavengers; | A 47.4% B 0.4% C 13.9% D 37.3% |


| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 20℃; | 29% |

| Conditions | Yield |
|---|---|
| With aluminum oxide at 150 - 300℃; Beim Leiten von Aceton-Dampf im den Gemisch; | |
| With dipotassium hydrogenphosphate; kieselguhr at 60℃; Beim Leiten von Aceton-Dampf im den Gemisch; |

| Conditions | Yield |
|---|---|
| at 20℃; Kinetics; Verlauf der Reaktion; auch in anderen Loesungsmitteln; | |
| at 20℃; Rate constant; und in anderen Loesungsmitteln; |

-
-
60-29-7
diethyl ether

-
-
74-90-8
hydrogen cyanide

-
-
67-64-1
acetone

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
74-90-8
hydrogen cyanide

-
-
143-33-9
sodium cyanide

-
-
67-64-1
acetone

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
74-90-8
hydrogen cyanide

-
-
151-50-8
potassium cyanide

-
-
67-64-1
acetone

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile


| Conditions | Yield |
|---|---|
| With sulfur dioxide; water | |
| With sulfuric acid | |
| With ammonium chloride In diethyl ether; water at 0 - 10℃; for 1h; | |
| With sulfuric acid at 10 - 15℃; |

| Conditions | Yield |
|---|---|
| With methanol; sulfuric acid | |
| Stage #1: acetone With sodium hydrogensulfite In water at 0℃; for 2h; Stage #2: potassium cyanide In water at 20℃; |

| Conditions | Yield |
|---|---|
| With sodium disulfite man gibt nach dem Erkalten eine gesaettigte waessr.Kaliumcyanidloesung hinzu; |

| Conditions | Yield |
|---|---|
| at 20℃; Kinetics; Verlauf der Reaktion; |


| Conditions | Yield |
|---|---|
| With sodium hydrogensulfite |


| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilyl cyanide; acetone With zinc(II) iodide Addition; Stage #2: With hydrogenchloride Substitution; |
-
-
532-28-5, 613-88-7
(RS)-mandelonitrile

-
-
67-64-1
acetone

-
A
-
100-52-7
benzaldehyde

-
B
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With Amberlite IRA904 OH(1-) In toluene at 25℃; for 5h; Equilibrium constant; |


| Conditions | Yield |
|---|---|
| In dimethylsulfoxide-d6; chloroform-d1 at 25℃; Kinetics; Product distribution; Further Variations:; Solvents; | A n/a B 53 % Spectr. |

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| In water |
-
-
773837-37-9
sodium cyanide

-
-
3483-11-2
lithium acetoacetate

-
A
-
89417-57-2
2-Hydroxy-2-methyl-bernsteinsaeuremonoamid

-
B
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
C
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 0 - 20℃; for 48h; pH=4.5; |

-
-
108-94-1
cyclohexanone

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
931-97-5
1-hydroxy-1-cyclohexanecarbonitrile

| Conditions | Yield |
|---|---|
| With Hevea brasiliensis (S)-hydroxynitrile lyase pH=4.5; aq. buffer; Enzymatic reaction; | 100% |
| With titanium(IV) isopropylate; dl-3-(2-hydroxy-1-naphthylidene)-imino-ε-caprolactam (Nap-ACL) In dichloromethane for 3h; Ambient temperature; | 99% |
| With ytterbium(III) isopropoxide In tetrahydrofuran for 0.5h; Ambient temperature; | 94% |
-
-
122-78-1
phenylacetaldehyde

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
50353-47-4
phenyl acetaldehyde cyanohydrin

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol for 2h; Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| With citrate buffer In di-isopropyl ether for 13h; Ambient temperature; mandelonitrile lyase, pH 5.5; | 100% |
| oxynitrilase; | 82% |
-
-
112-44-7
undecylaldehyde

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
130273-76-6
(RS)-2-hydroxy-dodecanenitrile

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate for 21h; Ambient temperature; | 100% |
| With triethylamine for 1.5h; Ambient temperature; | 99% |
-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
1885-14-9
phenyl chloroformate

-
-
130312-20-8
1-cyano-1-methyl-ethylphenylcarbonate

| Conditions | Yield |
|---|---|
| With pyridine for 0.5h; | 100% |
-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
2043-61-0
cyclohexanecarbaldehyde

-
-
100007-62-3, 107485-34-7, 4354-47-6
2-cyclohexyl-2-hydroxyacetonitrile

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate for 44h; Ambient temperature; | 100% |
| With titanium(IV) isopropylate; dl-3-(2-hydroxy-1-naphthylidene)-imino-ε-caprolactam (Nap-ACL) In dichloromethane for 2.5h; Ambient temperature; | 99% |
| With ytterbium(III) isopropoxide In tetrahydrofuran for 0.5h; Ambient temperature; | 86% |
-
-
66086-70-2
3-(4-nitro-phenyl)-6-phenyl-[1,2,4]triazine 4-oxide

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 0.333333h; Heating; | 100% |
-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 0.333333h; Heating; | 100% |
-
-
2627-86-3
(S)-1-phenyl-ethylamine

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
882182-35-6
2-methyl-2-(((1S)-1-phenylethyl)amino)propanenitrile

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 100% |
| In methanol at 20℃; for 10h; | 47% |
-
-
104062-70-6
(1-formyl-pentyl)-carbamic acid tert-butyl ester

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
180779-12-8
[1-(cyano-hydroxy-methyl)-pentyl]-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 20h; | 100% |
| With triethylamine In dichloromethane at 20℃; |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In tetrahydrofuran at 20℃; for 2h; | 100% |
-
-
63-05-8
Androstenedione

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
77881-13-1
17β-cyano-17α-hydroxy-4-androsten-3-one

| Conditions | Yield |
|---|---|
| With sodium carbonate In methanol; water at 17 - 40℃; for 29h; pH=8 - 9; | 100% |
| With water; sodium carbonate In methanol at 38 - 42℃; for 4h; pH=8 - 9; | 100% |
| With methanol; potassium hydroxide at 50℃; for 6h; Autoclave; Large scale; | 95.9% |
-
-
1134620-82-8
(2S)-(7-methyl-4,5,6,7-tetrahydro-2H-indazol-4-yl)propanal

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; Inert atmosphere; | 100% |

-
-
31007-06-4
2-(4-methoxyphenyl)propionitrile

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 25h; Inert atmosphere; regioselective reaction; | 100% |
-
-
622-97-9
1-ethenyl-4-methylbenzene

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
75920-45-5
(+/-)-2-(4'-methylphenyl)-propionitrile

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 25h; Inert atmosphere; regioselective reaction; | 100% |
-
-
405-99-2
para-fluorostyrene

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
51965-61-8
4-fluoro-α-methylbenzeneacetonitrile

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 25h; Inert atmosphere; regioselective reaction; | 100% |
-
-
2628-16-2
p-acetoxystyrene

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
1132795-53-9
2-(4-acetoxyphenyl)propanenitrile

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 40h; Inert atmosphere; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 25h; Inert atmosphere; regioselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With 3-[bis-(4-methoxy-phenyl)-phosphanyl]-2H-isoquinolin-1-one; N-{6-[bis-(4-methoxy-phenyl)-phosphanyl]-pyridin-2-yl}-2,2-dimethyl-propionamide; bis(1,5-cyclooctadiene)nickel (0) In toluene at 35℃; for 25h; Inert atmosphere; regioselective reaction; | 100% |

-
-
35666-81-0
2-methyl-2-(morpholin-4-yl)propanenitrile

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 24h; | 100% |
| In acetone at 20℃; for 24h; | 100% |
| at 100℃; for 1h; | 76% |
-
-
105499-11-4
2-(Boc-amino)propionaldehyde

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
103865-01-6
C9H16N2O3

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 20h; | 100% |
-
-
123285-52-9
2-(S)-[N-(tert-butoxycarbonyl)amino]-3-methylbutanal

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
163218-15-3
C11H20N2O3

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 20h; | 100% |
-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
346690-97-9
N-(tert-butoxycarbonyl)-2-aminobutanal

-
-
166196-01-6
tert-butyl (1-cyano-1-hydroxybutan-2-yl)carbamate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 20h; | 100% |
-
-
1256750-19-2
tert-butyl (1,1-dideutero-3-oxo-1-(perdeuterocyclobutyl)propan-2-yl)carbamate

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
1256750-20-5
tert-butyl (1,1-dideutero-3-cyano-3-hydroxy-1-(perdeuterocyclobutyl)propan-2-yl)carbamate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 15h; | 100% |

-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

| Conditions | Yield |
|---|---|
| With triphenyl phosphite; tetrakis(triphenylphosphite)nickel(0) In toluene at 100℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With nickel(II) acetate tetrahydrate; triphenylphosphine; zinc In N,N-dimethyl-formamide at 90℃; for 15h; Reagent/catalyst; Solvent; Autoclave; | 100% |
| With nickel(II) chloride hexahydrate; 1,3-bis-(diphenylphosphino)propane; zinc In N,N-dimethyl-formamide at 90℃; under 3750.38 Torr; for 15h; Catalytic behavior; Reagent/catalyst; Solvent; Autoclave; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphite)nickel(0); phosphorous acid trimethyl ester In toluene at 100℃; for 1h; stereoselective reaction; | 100% |
-
-
75-86-5
2-hydroxy-2-methylpropanenitrile

-
-
19355-69-2
2-amino-2-cyanopropane

-
-
34241-39-9
azobisisobutyronitrile

| Conditions | Yield |
|---|---|
| 99.8% |

| Conditions | Yield |
|---|---|
| With potassium permanganate; manganese dioxide; sulfuric acid In water; acetone | 99.5% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-2-methylpropanenitrile With sulfuric acid; water; 1-Nitropropane at 40 - 55℃; for 0.5h; Stage #2: With water; 1-Nitropropane at 130℃; for 1h; Product distribution / selectivity; | 99.2% |
| Stage #1: 2-hydroxy-2-methylpropanenitrile With nitromethane; sulfuric acid; water at 40 - 55℃; for 0.25 - 0.5h; Stage #2: With nitromethane; water at 120 - 130℃; for 0.5 - 1h; Product distribution / selectivity; | 98% |
| Stage #1: 2-hydroxy-2-methylpropanenitrile With Nitroethane; sulfuric acid; water at 40 - 55℃; for 0.333333h; Stage #2: With Nitroethane; water at 120 - 130℃; for 0.75 - 1h; Product distribution / selectivity; | 98% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View