Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:81-81-2
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name:warfarin CAS:81-81-2 Grade:For chemical industry, scientific research and export Product Quality 12 years of chemical raw materials Mature operation of the industry System stability Data storage Security without vulnerability Product
Cas:81-81-2
Min.Order:25 Kilogram
FOB Price: $1.0 / 2.0
Type:Other
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:81-81-2
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Siwei Development Group Ltd.
Product name: Warfarin CAS No.: 81-81-2 Molecule Formula:C19H16O4 Molecule Weight:308.33 Purity: 98.0% Package: 25kg/drum Description:White crystals or white crystalline powder Manufacture Standards:Enterprise Standard
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api, intermediat
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Antimex Chemical Limied
Our own factory produces direct sales with absolute price advantage Application:Pharmaceutical industry Transportation:By sea Port:Shanghai/tianjin
Cas:81-81-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Appearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Shanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
Shenzhen Foris Technology Co. LTD
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
LEAP CHEM Co., Ltd.
Best Seller, High Quality, Competitive Price, Fast Delivery, Quick ResponseAppearance:powder, or liquid Storage:Stored in room temperature, ventilated place Package:Bottle, barrel, cargo, container, etc. Application:Pharmaceuticals, intermediates, AP
Hangzhou KieRay Chem Co.,Ltd.
Hangzhou KieRaychem Co.,Ltd.is located in Yuhang District of Hangzhou City and specialized in the chemical product customization, development, sales, import and export. Current business is focused on fine chemicals, pharmaceutical materials and inter
Pure Chemistry Scientific Inc.
Warfarin Application:80010283
Shenzhen Sumshine Biotech Co., Ltd.
1. Our staff are all biomedical related majors with rich experience in the pharmaceutical industry, and can provide you with more professional services.2. Our supplier has a good quality management system, and the quality of products is reliable and
Synthetic route

| Conditions | Yield |
|---|---|
| With C23H24N2O6S*Li(1+) In tetrahydrofuran; dimethyl sulfoxide at 0℃; for 48h; Reagent/catalyst; Michael Addition; | 99% |
| With lipase In water at 50℃; for 168h; Temperature; Michael Addition; Enzymatic reaction; | 99.1% |
| With N-ethyl-N,N-diisopropylamine In water for 20h; Michael Addition; Reflux; | 93% |

| Conditions | Yield |
|---|---|
| With polystyrene-divinylbenzene support prepared cyclohexanol as porogen with immobilized 1,5,7-triazabicyclo[4.4.0]dec-5-ene at 100℃; for 96h; Reagent/catalyst; Solvent; Temperature; Time; Michael Addition; Green chemistry; | 96% |
| With L-proline In dimethyl sulfoxide at 20℃; for 15h; Michael addition; | 85% |
| With rac-Pro-OH In dimethyl sulfoxide at 20℃; for 24h; | 80% |
| With cetyltrimethylammonim bromide; 1,4-dihydropyridine-enolate In water at 20℃; for 10h; Michael addition; | 50% |

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; for 16h; | 84% |

-
-
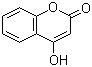
1076-38-6
4-hydroxy[1]benzopyran-2-one

-
-
5381-93-1, 86734-67-0, 86734-69-2, 127707-68-0
1-hydroxy-1-phenyl-3-butanone

-
-
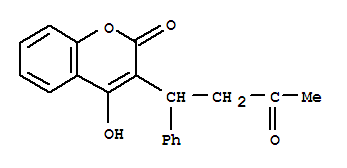
81-81-2
warfarin

| Conditions | Yield |
|---|---|
| With cyclopentene In chlorobenzene; toluene at 100℃; for 8h; Inert atmosphere; regioselective reaction; | 83% |
-
-
86795-49-5
3-(phenyl-λ3-iodanylidene)chromane-2,4-dione

-
-
1896-62-4
(E)-benzalacetone

-
-
81-81-2
warfarin

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 12h; | 76% |
-
-
38063-51-3
(+/-)-Warfarin 4-methyl ether

-
-
81-81-2
warfarin

| Conditions | Yield |
|---|---|
| With boron tribromide In dichloromethane -78 up to 0 deg C; |
-
-
102077-97-4
2-methoxywarfarin

-
-
81-81-2
warfarin

| Conditions | Yield |
|---|---|
| With boron tribromide In dichloromethane -78 up to 0 deg C; |

-
-
1076-38-6
4-hydroxy[1]benzopyran-2-one

-
-
116-11-0
2-Methoxypropene

-
-
100-52-7
benzaldehyde

-
-
81-81-2
warfarin

| Conditions | Yield |
|---|---|
| Stage #1: 4-hydroxy[1]benzopyran-2-one; 2-Methoxypropene; benzaldehyde With ethylenediamine diacetic acid In 1,4-dioxane at 90℃; for 4h; tandem Knoevenagel-hetero-Diels-Alder reaction; Stage #2: With hydrogenchloride; silica gel In water; trifluoroacetic acid at 20℃; |


| Conditions | Yield |
|---|---|
| In water |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1-n-butyl-3-methylimidazolim bromide; bovine serum albumin; tetrabutylammomium bromide / 4 h / 60 °C / Inert atmosphere; Enzymatic reaction 2: 48 h View Scheme | |
| Multi-step reaction with 2 steps 1: sodium hydroxide / water / Microwave irradiation 2: N-ethyl-N,N-diisopropylamine / water / 20 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: Fe3O4(at)L-proline/Pd2 NCs / 24 h / 70 °C / Sealed tube 2: water / 12 h / 100 °C View Scheme |
-
-
81-81-2
warfarin

-
-
14602-86-9
(1R,2S,5R)-menthyl chloroformate

| Conditions | Yield |
|---|---|
| With TEA In 1,2-dichloro-ethane for 0.5h; Ambient temperature; | 100% |
-
-
81-81-2
warfarin

-
-
593-56-6
N-methoxylamine hydrochloride

-
-
1103877-12-8
4-hydroxy-3-(3-(methoxyimino)-1-phenylbutyl)-2H-1-benzopyran-2-one

| Conditions | Yield |
|---|---|
| With pyridine In methanol at 20℃; for 18h; | 99% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide In pyridine at 55 - 60℃; for 5h; air; | A 98% B n/a |
-
-
81-81-2
warfarin

-
-
19493-09-5
Methylenetriphenylphosphorane

-
-
92824-18-5
4-hydroxy-3-(3-methyl-1-phenyl-3-butenyl)-2H-1-benzopyran-2-one

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; dimethyl sulfoxide | 95% |
| In tetrahydrofuran; dimethyl sulfoxide | 65% |
-
-
81-81-2
warfarin

-
-
99-61-6
3-nitro-benzaldehyde

-
-
1189759-41-8
4-hydroxy-3-[5-(3-nitro-phenyl)-3-oxo-1-phenyl-pent-4-enyl]-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 95% |
-
-
81-81-2
warfarin

-
-
104-88-1
4-chlorobenzaldehyde

-
-
1189759-36-1
4-hydroxy-3-[5-(4-chloro-phenyl)-3-oxo-1-phenyl-pent-4-enyl]-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 93% |
-
-
81-81-2
warfarin

-
-
89-98-5
2-chloro-benzaldehyde

-
-
1189759-37-2
4-hydroxy-3-[5-(2-chloro-phenyl)-3-oxo-1-phenyl-pent-4-enyl]-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 93% |
-
-
81-81-2
warfarin

-
-
555-16-8
4-nitrobenzaldehdye

-
-
1189759-40-7
4-hydroxy-3-[5-(4-nitro-phenyl)-3-oxo-1-phenyl-pent-4-enyl]-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 93% |
-
-
81-81-2
warfarin

-
-
910618-41-6
3-fluoro-3-(1-phenyl-3-oxobutyl)-2H-benzopyran-2,4-dione

| Conditions | Yield |
|---|---|
| With N-fluorobis<(trifluoromethyl)sulfonyl>imide In chloroform; water at 35℃; for 0.25h; | 92% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 5h; | A 85% B n/a |

-
-
98-01-1
furfural

-
-
81-81-2
warfarin

-
-
1253372-65-4
4-hydroxy-3-(5-furan-2-yl-3-oxo-1-phenyl-pent-4-enyl)-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 85% |
-
-
81-81-2
warfarin

-
-
100-52-7
benzaldehyde

-
-
1189759-35-0
4-hydroxy-3-(3-oxo-1,5-diphenyl-pent-4-enyl)-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 85% |
-
-
81-81-2
warfarin

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
1189759-38-3
4-hydroxy-3-[5-(4-methoxy-phenyl)-3-oxo-1-phenyl-pent-4-enyl]-chromen-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; | 85% |

| Conditions | Yield |
|---|---|
| With pyrographite; potassium hydroxide In water; isopropyl alcohol at 75℃; for 8h; pH=7.8 - 8; Product distribution / selectivity; Inert atmosphere; | 81% |
-
-
81-81-2
warfarin

| Conditions | Yield |
|---|---|
| With pyridine; sodium hydroxide; hydroxylamine hydrochloride In ethanol 1) 12 h, 2) reflux, 6 h; | 77% |
-
-
81-81-2
warfarin

-
-
10049-16-8
vanadium(IV) fluoride

| Conditions | Yield |
|---|---|
| In methanol methanolic soln. of VF4 added to methanolic soln. of C19H16O4 dropwise with stirring, mixt. had pH 3, refluxed for 2 h, kept overnight with stirring at room temp.; solvent removed on a rotary apparatus under reduced pressure; recrystn. (MeOH/n-hexane, 1/1); elem. anal., detd. by thermal anal.; | 77% |
-
-
81-81-2
warfarin

-
-
37149-18-1
1-oxyl-4-carboxyl-2,2,6,6-tetramethylpiperidine

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane; N,N-dimethyl-formamide at 0 - 20℃; for 26h; Inert atmosphere; | 75% |
-
-
81-81-2
warfarin

-
-
108-24-7
acetic anhydride

-
-
5979-00-0
4-acetoxy-3-(3-oxo-1-phenylbutyl)-2H-1-benzopyran-2-one

| Conditions | Yield |
|---|---|
| for 1h; Heating; | 71.4% |

| Conditions | Yield |
|---|---|
| In ethanol; water Heating; stereoselective reaction; | 68% |


| Conditions | Yield |
|---|---|
| In acetone at 45℃; for 48h; | 65% |
-
-
81-81-2
warfarin

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
929551-51-9
trifluoro-methanesulfonic acid 2-oxo-3-(3-oxo-1-phenyl-butyl)-2H-chromen-4-yl ester

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at -5 - 25℃; | 62% |

| Conditions | Yield |
|---|---|
| With pentamethylcyclopentadienyl(benzene)cobalt(III) hexafluorophosphate; potassium carbonate; silver carbonate In 2-methyltetrahydrofuran at 60℃; for 16h; Inert atmosphere; Sealed tube; | 56% |
-
-
67-56-1
methanol

-
-
81-81-2
warfarin

-
-
518-20-7, 54288-87-8, 54288-88-9, 64753-99-7, 64754-00-3, 64754-01-4, 65207-43-4, 94902-10-0
2-methoxy-2-methyl-4-phenyl-3,4-dihydropyrano[3,2-c]chromen-5(2H)-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 23h; Reflux; | A 53% B 9% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 25℃; for 24h; | 38% |
-
-
81-81-2
warfarin

-
-
24579-14-4
4'-Hydroxywarfarin

| Conditions | Yield |
|---|---|
| With culture of Cunninghamella bainieri In water for 144h; Product distribution; Ph7 phosphate buffer; metabolism of warfarin to 4'-hydroxywarfarin using the fungus Cunninghamella bainiery, possible mechanism of aromatic hydroxylation, primary isotope effect; inhibition of hydroxylation by CO; | 2 mg |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View












 T
T