Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Yixing Zeyuan Chemical Technology Co Ltd
We can supply: 1..Place of Origin China with Super quality by reasonable price. 2. sample orders are welcome 3.Customer Satisfaction is our priority and guarantee for the future. 4.Fast delivery by safe express way to all the country. 5.Has a
Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Colorless or pale yellow crystal Conforms Assay ≥99.0%
Cas:122-57-6
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Shanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:122-57-6
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:122-57-6
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name:benzylideneacetone CAS NO:122-57-6 Grade:Medical scientific research and export Molecular formula: C10H10O Molecular weight:146.19 Product Quality 12 years of chemical raw materials Mature operation of the industry System stability
Cas:122-57-6
Min.Order:25 Kilogram
FOB Price: $2.0 / 3.0
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
Benzalacetone Basic information Product Name: Benzalacetone Synonyms: (3E)-4-Phenyl-3-buten-2-one;1-Buten-3-one-1-phenyl;2-Phenylvinyl methyl ketone;4-phenyl-3-bute
Cas:122-57-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:122-57-6
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:122-57-6
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Chemical Name: Benzalacetone CAS No.: 122-57-6 Molecular Fomula:C10H10O Chemical Structure: Molecular weight: 146.19 Appearance: slightly yellow crystal solid Assay:≥99.0 Appearance:slightly yellow?crystal solid Storage:Store in cool and dry
Hangzhou Sartort Biopharma Co., Ltd
Appearance:light yellow powder Storage:Store at <= 20°C. Package:25kg/drum Application:1. It is used for the preparation of spices and galvanized brightener. 2. GB 2760--1996 allows it used as food with spices. Formulated balm, cherries, grapes, b
Cas:122-57-6
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:122-57-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: Benzalacetone CAS: 122-57-6 MF: C10H10O MW: 146.19 EINECS: 204-555-1 Mol File: 122-57-6.mol Benzalacetone Structure Benzalacetone Chemical Properties Melting point 39-42 °C(lit.) Boiling point 260-262 &de
Hangzhou Fonlynn Health Technology Co., Ltd.
Packing: According to customer requirements Delivery time: In stock or depands Port of shipment: Ningbo/Shanghai/Qingdao OEM/ODM:Welcome Sample:We can offer our existing samples at once Appearance:white powder/ Refer to COA Storage:Refer to COA
Cas:122-57-6
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Benzalacetone CAS:122-57-6 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediate
Cas:122-57-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:122-57-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Product Detail Minimum Order Qty. 10 Gram
Cas:122-57-6
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:122-57-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd,Our business covers more than 30 countries, most of the big customers come from Europe, America and other countries in the world, we can guarantee the quality and price. In recent decades, with the efforts of all em
Cas:122-57-6
Min.Order:1 Gram
FOB Price: $600.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We have s
Cas:122-57-6
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hefei Zhaobo Technology Co., Ltd.
Our Advantages Production: Advanced chemical equipment with years of experience Staffs for producing various extract products. Quality Control:A complete set of Testing Professional and Analysis Equipment ensures the Quality Requirements and Specif
Cas:122-57-6
Min.Order:1 Kilogram
FOB Price: $75.0 / 150.0
Type:Trading Company
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:light yellow crystal powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used for Pesticide
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:122-57-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Benzalacetone CAS No.: 122-57-6 Molecule Formula:C10H11O Molecule Weight:147.19 Purity: 99.0% Package: 25kg/drum Description:White crystalline powder Manufacture Standards:Enterprise Standard TESTING
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Synthetic route
-
-
17488-65-2
4-phenylbut-3-en-2-ol

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With dicarbonyl-(2,4-bis(trimethylsilyl)bicyclo[3.3.0]nona-1,4-dien-3-one)[acetonitrile]iron; acetone at 90℃; for 18h; Opppenauer oxidation; Inert atmosphere; | 99% |
| With aluminum oxide; potassium permanganate In neat (no solvent) for 0.05h; | 97% |
| With benzyltriphenylphosphonium peroxymonosulfate for 0.0833333h; Oxidation; | 97% |
-
-
29833-94-1
2-Methyl-2-((E)-styryl)-[1,3]dithiane

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; niobium pentachloride; sodium iodide In water; ethyl acetate at 20℃; for 0.25h; | 98% |
| With iron(III)-acetylacetonate; dihydrogen peroxide; sodium iodide In water; ethyl acetate at 20℃; for 0.2h; | 95% |
| With antimonypentachloride In dichloromethane at 0℃; for 0.166667h; | 75% |
| With oxygen; 2,4,6-tris(p-chlorophenyl)pyrylium perchlorate In dichloromethane for 1.5h; Irradiation; | 50% |
| With methyltriphenylphosphonium tribromide; water 1.) THF, 3 h, room temperature; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 110℃; for 20h; Heck Reaction; Green chemistry; | 90% |
| With tris-(dibenzylideneacetone)dipalladium(0); tetrabutyl-ammonium chloride; sodium hydrogencarbonate In N,N-dimethyl-formamide at 100℃; for 4h; Heck Reaction; Microwave irradiation; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water Aldol Condensation; | 96% |
| With N,N-dimethyl-cyclohexanamine In water at 70℃; for 24h; Aldol Condensation; Green chemistry; | 94% |
| With C66H78O6(6-)*6Na(1+) at 55℃; for 1h; Reagent/catalyst; | 93.5% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 110℃; for 20h; Heck Reaction; Green chemistry; | 70% |

| Conditions | Yield |
|---|---|
| With [1,3-bis(2,6-di-isopropylphenyl)imidazol-2-ylidene]copper(I) chloride; sodium t-butanolate In toluene at 20℃; for 3h; Aldol Condensation; Schlenk technique; Inert atmosphere; | 90% |
-
-
6249-79-2, 57134-72-2, 84033-96-5, 144667-51-6, 144667-52-7, 146388-50-3
1-(3-phenyloxiran-2-yl)ethanone

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With hydrogen; triethyl phosphite In isopropyl alcohol at 100℃; under 6000.6 Torr; for 24h; Glovebox; chemoselective reaction; | 89% |
| With carbon monoxide In tetrahydrofuran; water at 110℃; under 6840.46 Torr; for 12h; Autoclave; chemoselective reaction; | 86% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; [1,1'-biphenyl]-4-yl(4-([1,1'-biphenyl]-4-yl)-1H-imidazol-2-yl)methanone; palladium dichloride In water; N,N-dimethyl-formamide at 80℃; for 8h; Heck Reaction; Sealed tube; stereoselective reaction; | 94% |
| With palladium diacetate; sodium hydrogencarbonate In water; N,N-dimethyl-formamide at 120℃; under 375.038 - 2250.23 Torr; for 1h; Reagent/catalyst; Temperature; Heck Reaction; Microwave irradiation; | 81% |
| With C33H33N2(1+)*Cl(1-); palladium diacetate; potassium carbonate In water; N,N-dimethyl-formamide for 2h; Heck Reaction; Inert atmosphere; Sealed tube; Heating; | 79% |
-
-
100-52-7
benzaldehyde

-
-
67-64-1
acetone

-
A
-
538-58-9
1,5-diphenyl-1,4-pentadiene-3-one

-
B
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With diethyl ether; magnesium bromide-butylate | |
| With 1,3,4,6,7,8-hexahydro-2H-pyrimido[1,2-a]pyrimidine at 60℃; for 0.5h; Inert atmosphere; | |
| With chromium(VI) oxide at 56℃; for 10h; | |
| Aldol Condensation; | |
| With sodium dodecyl-sulfate; sodium hydroxide In water at 40℃; for 4h; Solvent; Claisen-Schmidt Condensation; Sealed tube; |

-
-
100-52-7
benzaldehyde

-
-
67-64-1
acetone

-
A
-
122-57-6
1-Phenylbut-1-en-3-one

-
B
-
5337-88-2
3-methyl-5-phenyl-2-cyclohexen-1-one

| Conditions | Yield |
|---|---|
| With tris(acetonitrile)(η5-pentamethylcyclopentadienyl)rhodium(III) hexafluoroantimonate; rac-methylbenzylamine; silver carbonate at 80℃; for 24h; Robinson Annulation; Sealed tube; | A 10% B 65% |
| With tris(acetonitrile)(η5-pentamethylcyclopentadienyl)rhodium(III) hexafluoroantimonate; rac-methylbenzylamine; silver carbonate at 80℃; for 14h; Robinson Annulation; Sealed tube; | A 42% B 40% |


| Conditions | Yield |
|---|---|
| With palladium Hydrogenation; | |
| Multi-step reaction with 2 steps 1: hydroxylamine-O-sulphonic acid / H2O / 0.58 h / 0 °C 2: H3C-COONa, K2Te / 4 h / Ambient temperature View Scheme | |
| With formic acid; [1,3-bis(2,4,6-trimethylphenyl)imidazol]-2-ylidene; triethylamine; [Pd(1,4-di-t-butyl-1,4-diazabutadiene)(maleic anhydride)] In para-xylene; acetonitrile for 24h; Heating; | |
| With hydrogen under 760.051 Torr; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In tetrahydrofuran; methanol; water for 28h; Heating; | 95% |
| With benzyltriphenylphosphonium peroxodisulfate In acetonitrile for 0.75h; Oxidation; Heating; | 90% |
| With KMnO4/alumina at 50℃; for 0.166667h; | 87% |
| With aluminium trichloride; butyltriphenylphosphonium dichromate In chloroform for 0.75h; Oxidation; Heating; | 70% |
| With gold(III) tribromide; dimethylglyoxal In ethanol; water at 60℃; for 15h; pH=7; | 91 %Spectr. |

| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxy-3-butene With oxygen; palladium diacetate In N,N-dimethyl-formamide at 90℃; under 760.051 Torr; for 4h; Green chemistry; Stage #2: phenylboronic acid With 2.9-dimethyl-1,10-phenanthroline In N,N-dimethyl-formamide at 90℃; for 18h; Heck Reaction; Green chemistry; | 85% |

| Conditions | Yield |
|---|---|
| With Arabidopsis thaliana berberine bridge enzyme-like protein 15 L182V; oxygen In dimethyl sulfoxide at 30℃; for 24h; pH=7.0; Reagent/catalyst; Enzymatic reaction; enantioselective reaction; | A 42 %Chromat. B 58 %Chromat. |
-
-
32398-66-6
1-phenylbut-2-yn-1-ol

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With KSF clay for 0.0833333h; Irradiation; microwave irradiation; | 92% |
| With [Ag{μ2-N,S-(1,3,5-triaza-7-phosphaadamantane)=NP(=S)(OEt)2}]x[SbF6]x In water at 160℃; for 1h; Microwave irradiation; | 90% |
-
-
22956-11-2
4-Phenyl-4-phenylsulfanyl-butan-2-one

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With diiodomethane; diethylzinc; trifluoroacetic acid In dichloromethane at 0℃; | 95% |
-
-
100-52-7
benzaldehyde

-
-
67-64-1
acetone

-
A
-
5381-93-1, 86734-67-0, 86734-69-2, 127707-68-0
(4S)-4-hydroxy-4-phenylbutan-2-one

-
B
-
5381-93-1, 86734-69-2, 127707-68-0, 86734-67-0
(4R)-4-hydroxy-4-phenylbutan-2-one

-
C
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With 1H-imidazole at 20℃; under 45004.5 Torr; Aldol Addition; Flow reactor; Sonication; stereoselective reaction; | A n/a B n/a C 13% |
| L-proline In various solvent(s) at 20℃; for 20h; Product distribution; Further Variations:; Solvents; catalyst amounts; | |
| With 5-[(2S)-pyrrolidine-2-yl]-1H-tetrazole In dimethyl sulfoxide at 60℃; for 0.5h; Aldol condensation; microreactor; optical yield given as %ee; enantioselective reaction; | A n/a B n/a C 18 %Spectr. |
| With Zn(2+)*C8H5NO4(2-)*2C5H9NO2 In dimethyl sulfoxide at 40℃; for 96h; Catalytic behavior; Reagent/catalyst; Temperature; Aldol Addition; Green chemistry; Optical yield = 73 %ee; enantioselective reaction; | |
| Stage #1: acetone With L-proline In chloroform; dimethyl sulfoxide at 0℃; for 0.5h; Aldol Addition; Sealed tube; Stage #2: benzaldehyde In chloroform; dimethyl sulfoxide at 0℃; for 96h; Concentration; Time; Temperature; Solvent; Aldol Addition; Sealed tube; enantioselective reaction; | A n/a B 76 %Spectr. C 8 %Spectr. |

-
-
53309-95-8
(+/-)-2-amino-4-phenylbut-3-ene

-
-
6249-79-2, 57134-72-2, 84033-96-5, 144667-51-6, 144667-52-7, 146388-50-3
trans-2-acetyl-3-phenyloxirane

-
B
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With 4-phenylnaphthalene-1,2-dione In acetonitrile at 23℃; for 60h; | A 15% B 15% |
-
-
909413-25-8
6-(tert-butyl)-12-phenyl-5,6,7,12-tetrahydrodibenzo[c,f][1,5]azastibocine

-
-
78-94-4
methyl vinyl ketone

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With palladium diacetate In 1,4-dioxane; water for 24h; Heating; Inert atmosphere; | 79% |
| With palladium diacetate In 1,4-dioxane; water at 100℃; for 1h; Mizoroki-Heck reaction; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| With caro's acid; silica gel In acetonitrile at 20℃; for 0.366667h; | 90% |
-
-
1223632-87-8, 960601-06-3
2-(trimethylsilyl)-4-phenyl-3-butyn-2-ol

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran | 100% |

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate In water; acetonitrile at 0℃; for 5h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium chloride; zinc In ethanol at 80℃; for 0.00833333h; microwave irradiation; | 95% |
| With iron(II) oxalate In N,N-dimethyl-formamide at 100℃; for 2h; | 92% |
| With sodium tetrahydroborate; nickel dichloride In methanol at 20℃; for 0.5h; | 90% |
-
-
74896-58-5
4-ethylsulfanyl-4-phenyl-butan-2-one

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In chloroform 1.) ice-bath, 2.) r.t., 15 h; | 89% |
| With aluminum oxide; potassium sulfate; potassium hydrogensulfate; potassium peroxomonosulfate 1.) 6 h, room temperature 2.) CH2Cl2, 40 min, room temperature; Yield given. Multistep reaction; |
-
-
4438-85-1
3H-chromene-2,4-dione

-
-
32398-66-6
1-phenylbut-2-yn-1-ol

-
A
-
15151-14-1
2-methyl-4-phenylpyrano[3,2-c]chromen-5(4H)-one

-
B
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With C24H20N2O4Ru; trifluoroacetic acid In toluene at 100℃; for 3h; Inert atmosphere; | A 82% B 8% |


-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With Raney-Ni In methanol at 70℃; for 12h; Reagent/catalyst; Inert atmosphere; | 84% |
-
-
130043-06-0
2-[(tert-butyldimethylsilyl)oxy]-4-phenyl-1,3-butadiene

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With (1,10-phenanthroline)(triphenylphosphine)CuBr; sodium 2,2,2-trifluoroacetate In acetonitrile at 80℃; for 3h; | 7 %Spectr. |
| With 7,8-benzoquinoline; trimethylsilyl-2,2-difluoro-2-(fluorosulphonyl)acetate In toluene at 80℃; for 1h; Reagent/catalyst; | 55 %Spectr. |

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; water; palladium diacetate at 70℃; for 12h; Temperature; Sealed tube; Schlenk technique; diastereoselective reaction; | A n/a B 6 %Spectr. |


-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With ethanol; potassium hydrogencarbonate; silver nitrate In water at 70℃; for 8h; | 72% |
-
-
6310-44-7
3,4-dibromo-4-phenylbutan-2-one

-
A
-
10541-24-9
α-bromobenzalacetone

-
B
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide at 155 - 160℃; for 1h; | A 17% B 74% |
| With N,N-dimethyl-formamide at 155 - 160℃; for 1h; | A 17% B 73% |
-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
6249-79-2, 57134-72-2, 84033-96-5, 144667-51-6, 144667-52-7, 146388-50-3
1-(3-phenyloxiran-2-yl)ethanone

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; potassium fluoride on basic alumina In decane; toluene at 25℃; for 24h; Inert atmosphere; | 100% |
| With sodium hydroxide In 1,4-dioxane; water for 20h; Reflux; | 95% |
| With Amberlyst A-26 (OH- form); dihydrogen peroxide In 1,4-dioxane at 20℃; for 6h; | 94% |

| Conditions | Yield |
|---|---|
| With diphenylsilane; zinc(II) chloride; tetrakis(triphenylphosphine) palladium(0) In chloroform for 2h; Ambient temperature; | 100% |
| With hydrogen In toluene at 70℃; under 750.075 Torr; for 0.666667h; | 100% |
| With C50H57IrN2P; hydrogen In dichloromethane at -78 - 20℃; | 100% |
-
-
75-52-5
nitromethane

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
122348-55-4, 122348-59-8, 142232-08-4, 1574-86-3
5-nitro-4-phenylpentan-2-one

| Conditions | Yield |
|---|---|
| With hydrotalcite for 0.1h; Michael addition; Microwave irradiation; | 100% |
| With potassium fluoride; 18-crown-6 ether In acetonitrile for 168h; Heating; | 93% |
| With C10H20NaO5*HO(1-); water In methanol at 25℃; for 0.833333h; Michael addition; | 90% |
-
-
7393-43-3
tetraallyl tin

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
21573-75-1
3-methyl-1-phenyl-hexa-1,5-dien-3-ol

| Conditions | Yield |
|---|---|
| 2,6-dimethylpyridine; zinc trifluoromethanesulfonate In dichloromethane at 20℃; for 24h; | 100% |
| With 4C33H36BO6(3-)*4Nd(3+)*2C3H7NO*14H2O*CO3(2-)*2C2H8N(1+); sodium dodecyl-sulfate In water at 20℃; for 48h; chemoselective reaction; | 81.4% |
| With n-butyllithium; cerium(III) chloride 1.) hexane, THF, RT, 1 h, 2.) hexane, THF, 0 deg C, 1 h; Yield given. Multistep reaction; | |
| With 1,1'-bi-2-naphthol |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In dichloromethane for 5h; | 100% |
-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
17488-65-2
4-phenylbut-3-en-2-ol

| Conditions | Yield |
|---|---|
| With diphenylsilane; cesium fluoride at 25℃; for 0.5h; | 100% |
| With potassium tert-butylate; hydrogen; potassium isopropoxide; [Ru((R,R)-cyP2N2)HCL] at 20℃; under 22801.5 Torr; for 12h; Conversion of starting material; | 100% |
| With potassium tert-butylate; hydrogen; potassium isopropoxide; Ru((R,R)-cyP2(NH)2)HCl at 20℃; under 22801.5 Torr; for 12h; Conversion of starting material; | 100% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate at 50℃; for 8.5h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With C23H24N2O6S*Li(1+) In tetrahydrofuran; dimethyl sulfoxide at 0℃; for 48h; Reagent/catalyst; Michael Addition; | 99% |
| With lipase In water at 50℃; for 168h; Temperature; Michael Addition; Enzymatic reaction; | 99.1% |
| With N-ethyl-N,N-diisopropylamine In water for 20h; Michael Addition; Reflux; | 93% |
-
-
104-47-2
p-methoxybenzylnitrile

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
102118-66-1, 102118-67-2, 114879-80-0, 114879-87-7
(methoxy-4 phenyl)-1 phenyl-2 oxo-4 pentane carbonitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol for 4h; Ambient temperature; | 99% |
| With tetrabutylammomium bromide; potassium carbonate In toluene at 50℃; for 6h; | 70% |
-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
6310-44-7, 32147-15-2, 55025-54-2
(3S*,4R*)-3,4-dibromo-3-phenylbutan-2-one

| Conditions | Yield |
|---|---|
| With 1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione; 1-(3,5-bis(trifluoromethyl)phenyl)-3-(2-(dimethylamino)ethyl)thiourea In dichloromethane at 22℃; for 2.5h; optical yield given as %de; diastereospecific reaction; | 99% |
| With 1-(2-diethyl-aminoethyl)-3-methylimidazolium hexafluorophosphate; bromine In water at 10 - 20℃; for 0.25h; stereoselective reaction; | 91% |
| With 4-dimethylaminopyridine tribromide In acetic acid for 6h; Ambient temperature; | 85% |
| With oxone; sodium bromide at 20℃; for 0.666667h; Neat (no solvent); Mechanical ball milling; | 81% |
-
-
95-20-5
2-methyl-1H-indole

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
97355-53-8
4-(2-methyl-1H-indol-3-yl)-4-phenyl-2-butanone

| Conditions | Yield |
|---|---|
| With sodium ligninsulfonate-immobilized Sc(OTf)3 In ethanol Michael Addition; | 99% |
| bismuth oxide perchlorate In acetonitrile for 0.75h; Michael addition; ultrasonic irradiation; | 93% |
| bismuth(III) nitrate at 20℃; for 0.166667h; Michael addition; grinding; | 92% |
-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
73183-34-3
bis(pinacol)diborane

| Conditions | Yield |
|---|---|
| With ethyl cinnamate; copper(l) chloride; sodium t-butanolate In tetrahydrofuran at 20℃; | 99% |
| Stage #1: 1-Phenylbut-1-en-3-one; bis(pinacol)diborane With potassium carbonate In methanol; toluene at 60℃; for 2h; Michael type reaction; Inert atmosphere; Stage #2: With water; ammonium chloride In methanol; toluene Inert atmosphere; | 99% |
| Stage #1: bis(pinacol)diborane With copper(l) chloride; sodium t-butanolate In tetrahydrofuran at 20℃; for 0.166667h; Inert atmosphere; Stage #2: 1-Phenylbut-1-en-3-one In tetrahydrofuran; water at 20℃; for 3h; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 20℃; for 5h; Product distribution / selectivity; | 99% |
| With potassium carbonate at 20℃; for 5h; Product distribution / selectivity; | 96% |
-
-
94839-07-3
3,4-(methylenedioxy)-benzeneboronic acid

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With potassium carbonate at 20℃; for 5h; | 99% |

| Conditions | Yield |
|---|---|
| With 1-((1R,2R)-2-aminocyclohexyl)-3-(3,5-bis(trifluoromethyl)phenyl)thiourea; 4-pyrrolidin-1-ylpyridine In toluene at 20℃; for 72h; Michael Addition; enantioselective reaction; | 99% |
| With C27H37N5OS In tetrahydrofuran at 20℃; for 48h; Michael condensation; optical yield given as %ee; enantioselective reaction; | 92% |
| With (R,R)-1,2-diphenylethylenediamine; benzene-1,2-dicarboxylic acid In ethanol at 20℃; for 168h; Michael Addition; enantioselective reaction; | 75% |
-
-
13195-64-7
diisopropyl malonate

-
-
122-57-6
1-Phenylbut-1-en-3-one

| Conditions | Yield |
|---|---|
| With N-[(2S)-2-amino-3,3-dimethylbutyl]-4-methylbenzenesulfonamide In toluene at 20℃; for 72h; Michael condensation; optical yield given as %ee; regioselective reaction; | 99% |
| With (S)-3-(1H-indol-3-yl)-N1-propylpropane-1,2-diamine; trifluoroacetic acid In chloroform at 20℃; for 24h; Michael condensation; optical yield given as %ee; enantioselective reaction; | 72% |

| Conditions | Yield |
|---|---|
| With (S)-3-(1H-indol-3-yl)-N1-propylpropane-1,2-diamine; trifluoroacetic acid In chloroform at 20℃; for 24h; Michael condensation; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
15014-25-2
malonic acid dibenzyl ester

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
541508-74-1
(R)-dibenzyl 2-(3-oxo-1-phenylbutyl)malonate

| Conditions | Yield |
|---|---|
| With (S)-3-(1H-indol-3-yl)-N1-propylpropane-1,2-diamine; trifluoroacetic acid In chloroform at 20℃; for 24h; Michael condensation; optical yield given as %ee; enantioselective reaction; | 99% |
| With N-[(2S)-2-amino-3,3-dimethylbutyl]-4-methylbenzenesulfonamide In toluene at 20℃; for 72h; Michael condensation; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
108-59-8
malonic acid dimethyl ester

-
-
109012-81-9
dimethyl (R)-(-)-4-oxo-2-phenyl-1,1-pentanedicarboxylate

| Conditions | Yield |
|---|---|
| With (S)-3-(1H-indol-3-yl)-N1-propylpropane-1,2-diamine; trifluoroacetic acid In chloroform at 20℃; for 24h; Michael condensation; optical yield given as %ee; enantioselective reaction; | 99% |
| With N-[(2S)-2-amino-3,3-dimethylbutyl]-4-methylbenzenesulfonamide In toluene at 20℃; for 72h; Michael condensation; optical yield given as %ee; regioselective reaction; | 98% |
| With C59H86N10O9; benzoic acid In tetrahydrofuran at 40℃; Michael Addition; enantioselective reaction; | 97% |
| With C59H86N10O9; benzoic acid In tetrahydrofuran at 40℃; for 96h; Michael Addition; Sealed tube; enantioselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With 9-epi-9-amino-9-deoxyquinine; N,N,N',N'-tetramethyl-1,8-diaminonaphthalene; benzoic acid at 40℃; for 48h; Catalytic behavior; Reagent/catalyst; Temperature; Michael Addition; enantioselective reaction; | 99% |
| Stage #1: nitromethane With C43H64N4O4; scandium tris(trifluoromethanesulfonate) at 30℃; for 0.5h; Asymmetric Michael addition; Inert atmosphere; Neat (no solvent); Stage #2: 1-Phenylbut-1-en-3-one With dmap at 30℃; for 48h; Asymmetric Michael addition; Inert atmosphere; Neat (no solvent); optical yield given as %ee; enantioselective reaction; | 97% |
| With (S)-N1-cyclohexyl-3,3-dimethylbutane-1,2-diamine; benzoic acid In ethyl acetate at 30℃; for 36h; Michael Addition; enantioselective reaction; | 89% |
-
-
848924-85-6
2-(5-methoxy-2-oxoindolin-3-ylidene)malononitrile

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
1332338-80-3
5'-methoxy-2',5-dioxo-3-phenylspiro[cyclohexane-1,3'-indoline]-2,2-dicarbonitrile

| Conditions | Yield |
|---|---|
| With C20H25N3O; (R)-1,1'-binaphthyl-2,2'-phosphoric acid In 1,2-dichloro-ethane at 80℃; for 2h; Double Michael addition; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
1332338-72-3
C11H4BrN3O

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
1332338-85-8
6'-bromo-2',5-dioxo-3-phenylspiro[cyclohexane-1,3'-indoline]-2,2-dicarbonitrile

| Conditions | Yield |
|---|---|
| With C20H25N3O; (R)-1,1'-binaphthyl-2,2'-phosphoric acid In 1,2-dichloro-ethane at 80℃; for 3h; Double Michael addition; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
1332338-74-5
2-(2-oxo-7-(trifluoromethyl)indolin-3-ylidene)malononitrile

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
1332338-87-0
2',5-dioxo-3-phenyl-7'-(trifluoromethyl)spiro[cyclohexane-1,3'-indoline]-2,2-dicarbonitrile

| Conditions | Yield |
|---|---|
| With C20H25N3O; (R)-1,1'-binaphthyl-2,2'-phosphoric acid In 1,2-dichloro-ethane at 80℃; for 1.5h; Double Michael addition; optical yield given as %ee; enantioselective reaction; | 99% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
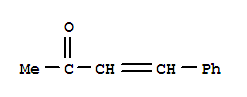



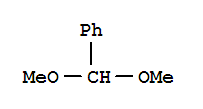
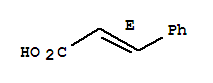

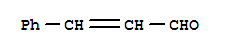









 Xn,
Xn, Xi
Xi