-
Name
1,1,1-TRICHLOROACETONE
- EINECS 213-063-6
- CAS No. 918-00-3
- Article Data31
- CAS DataBase
- Density 1.475 g/cm3
- Solubility
- Melting Point 9-11℃
- Formula C3H3 Cl3 O
- Boiling Point 134°C
- Molecular Weight 161.415
- Flash Point 64°C
- Transport Information
- Appearance colorless liquid
- Safety Mutation data reported. When heated to decomposition it emits toxic vapors of Cl−.
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
- Hazard Symbols R25:; R26:; R34:; R50/53:;
- Synonyms 1,1,1-Trichloro-2-propanone;1,1,1-Trichloroacetone; 1,1,1-Trichloropropanone; NSC 2645; Trichloromethylmethyl ketone; a,a,a-Trichloroacetone
- PSA 17.07000
- LogP 1.94560
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium dichromate; sulfuric acid In acetic acid for 0.166667h; Ambient temperature; | 82% |
| With potassium dichromate; sulfuric acid | |
| With potassium dichromate; sulfuric acid In water |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; sodium chloride In water at 23 - 25℃; Product distribution; current 11 A; | 82% |
| With sodium hydrogencarbonate; sodium chloride In water at 23 - 25℃; electrochemical (current 11 A); | 82% |
| With chlorine; potassium hydroxide In water at 15 - 20℃; for 6.6h; Reagent/catalyst; Temperature; |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite | 80% |
| With sodium hydrogencarbonate; sodium chloride Product distribution; electrochemically; | 65% |
| With sodium hydrogencarbonate; sodium chloride In water electrochemical; | 65% |
| With sodium acetate; chlorine; acetic acid; sodium chloride Reagens 4: Wasser; |
-
-
6430-25-7
acetyl-triphenylgermane

-
A
-
67-72-1
hexachloroethane

-
B
-
918-00-3
1,1,1-trichloroacetone

-
C
-
1626-24-0
chlorotriphenylgermane

-
D
-
75-36-5
acetyl chloride

| Conditions | Yield |
|---|---|
| With tetrachloromethane In tetrachloromethane Irradiation (UV/VIS); Ph3GeCOMe photolyzed in CCl4; | A 53% B 10% C 78% D 74% |


| Conditions | Yield |
|---|---|
| With perhydrodibenzo-18-crown-6 In acetonitrile for 0.16h; | 40% |
| With dibenzo-18-crown-6 In acetonitrile for 0.16h; Product distribution; other reagent; reaction without macrocyclic polyether; other reaction time.; | 25% |

| Conditions | Yield |
|---|---|
| In diethyl ether |

| Conditions | Yield |
|---|---|
| (i) EtOH, benzene, (ii) propanoic acid, H2SO4; Multistep reaction; |
-
-
56-23-5
tetrachloromethane

-
-
86310-10-3
5,5-dicyclopropyl-2-methoxy-2-methyl-Δ3-1,3,4-oxadiazoline

-
A
-
67-66-3
chloroform

-
B
-
79-20-9
acetic acid methyl ester

-
C
-
918-00-3
1,1,1-trichloroacetone

-
D
-
86310-12-5
1,4-dichloro-1-cyclopropylbut-1-ene

-
E
-
86310-11-4
1,1,1,2-tetrachloro-2,2-dicyclopropylethane

-
F
-
86310-13-6
(Z)-1,4,7-Trichloro-5-trichloromethyl-hept-3-ene

| Conditions | Yield |
|---|---|
| Mechanism; Heating; | A 10 % Chromat. B 88 % Chromat. C 3 % Chromat. D 7 % Chromat. E n/a F 26 % Chromat. |

-
-
56-23-5
tetrachloromethane

-
-
431-03-8
dimethylglyoxal

-
A
-
918-00-3
1,1,1-trichloroacetone

-
B
-
73623-81-1
butane-2,3-dione (enol form)

-
C
-
75-36-5
acetyl chloride

-
D
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| In various solvent(s) Product distribution; Irradiation; |


| Conditions | Yield |
|---|---|
| With chlorine In N,N-dimethyl-formamide at 50℃; for 2h; Product distribution; other chlorination agent; | A 95 % Chromat. B 5 % Chromat. |

-
A
-
2229-07-4
methyl radical

-
B
-
3170-80-7
trichloromethyl radical

-
C
-
918-00-3
1,1,1-trichloroacetone

-
D
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| In tetrachloromethane Rate constant; Heating; |

-
-
7782-50-5
chlorine

-
-
78-95-5
chloroacetone

-
A
-
921-03-9
1,1,3-trichloroacetone

-
B
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| at 30 - 40℃; |


-
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid Hydrolysis; |
-
-
513-88-2
1,1-Dichloroacetone

-
-
7782-50-5
chlorine

-
A
-
921-03-9
1,1,3-trichloroacetone

-
B
-
918-00-3
1,1,1-trichloroacetone

-
-
498-23-7
citraconic acid

-
-
7732-18-5
water

-
-
7782-50-5
chlorine

-
A
-
6625-00-9
β-chloromethacrylic acid

-
B
-
918-00-3
1,1,1-trichloroacetone

-
C
-
10027-62-0
α,β,β-trichloro-isobutyric acid

| Conditions | Yield |
|---|---|
| citraconsaeures Natrium reagiert; |


-
-
513-88-2
1,1-Dichloroacetone

-
A
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
B
-
918-00-3
1,1,1-trichloroacetone

-
C
-
1768-31-6
pentachloroacetone

| Conditions | Yield |
|---|---|
| With pyridine; chlorine at 70 - 80℃; for 4h; |

-
-
78-95-5
chloroacetone

-
A
-
116-16-5
1,1,1,3,3,3-hexachloro-propan-2-one

-
B
-
918-00-3
1,1,1-trichloroacetone

-
C
-
1768-31-6
pentachloroacetone

| Conditions | Yield |
|---|---|
| With pyridine; chlorine at 70 - 80℃; for 7.75h; |

-
-
67-64-1
acetone

-
A
-
513-88-2
1,1-Dichloroacetone

-
B
-
921-03-9
1,1,3-trichloroacetone

-
C
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; chlorine at 20 - 80℃; for 8.5h; |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
A
-
302-17-0
chloral hydrate

-
B
-
67-66-3
chloroform

-
C
-
513-88-2
1,1-Dichloroacetone

-
D
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With chlorine In aq. phosphate buffer pH=7; Kinetics; pH-value; UV-irradiation; |


| Conditions | Yield |
|---|---|
| With chromium dichloride; hydrogen; magnesium bromide; Pd/C (10percent) In tetrahydrofuran at 20℃; for 0.5h; | 99% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
100-52-7
benzaldehyde

-
-
53973-14-1, 148470-37-5, 119856-19-8
(Z)-2-Chloro-1-phenyl-1-buten-3-one

| Conditions | Yield |
|---|---|
| With chromium dichloride; hydrogen; magnesium bromide; Pd/C (10percent) In tetrahydrofuran at 20℃; for 0.5h; | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 8h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 97% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
39687-95-1
isocyanoacetic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine; copper(l) chloride In 1,2-dichloro-ethane at 18 - 23℃; for 0.5h; | 95% |
| With triethylamine; copper(l) chloride In 1,2-dichloro-ethane for 0.5h; Ambient temperature; | 95% |

| Conditions | Yield |
|---|---|
| With copper(l) chloride at 110℃; for 12h; | 90% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 88% |

| Conditions | Yield |
|---|---|
| With chromium dichloride; hydrogen; magnesium bromide; Pd/C (10percent) In tetrahydrofuran at 0℃; for 12h; | 87% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 86% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 85% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
1197-22-4
N-phenylmethanesulfonamide

-
-
3700-65-0
N-(methylsulfonyl)-N-phenylacetamide

| Conditions | Yield |
|---|---|
| With Aliquat 336; sodium carbonate In acetonitrile at 80℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 85% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 85% |
| With aniline In chloroform compatitive reaction with aniline; reaction condition: 1) 5 - 10 deg C, 30 min; 2) 25 deg C, 1 h; | 80% |
-
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With Noyori's catalyst In formic acid; triethylamine at 28℃; for 17h; Reagent/catalyst; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 82% |

| Conditions | Yield |
|---|---|
| With chromium dichloride; hydrogen; magnesium bromide; Pd/C (10percent) In tetrahydrofuran at 0℃; for 12h; | 82% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
1082905-88-1
1,1,1-trichloro-2-sulfanylpropan-2-ol

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydrogen sulfide In methanol at -50℃; | 81% |
-
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 25℃; for 1h; | 80% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
79894-70-5
methyl-2,2,2-trichloroacetimidate

| Conditions | Yield |
|---|---|
| With pyridine; hydroxylamine In ethanol | 80% |

| Conditions | Yield |
|---|---|
| With triethylamine In hexane Heating; | 80% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
6758-40-3
p-Nitrobenzoyl-L-(+)-glutamic acid

-
-
2387-48-6
2,4-diamino-5-nitroso-6-hydroxypyrimidine

-
-
59-30-3
folate

| Conditions | Yield |
|---|---|
| Stage #1: p-Nitrobenzoyl-L-(+)-glutamic acid; 2,4-diamino6-hydroxy-5-nitroso pyrimidine With hydrogen In water at 82℃; under 6000.6 Torr; Stage #2: 1,1,1-trichloroacetone With sodium metabisulfite; sodium hydroxide In water at 45℃; pH=4.5; pH-value; Pressure; Temperature; | 80% |

-
-
918-00-3
1,1,1-trichloroacetone

-
-
4271-30-1
N-(4-aminobenzoyl)-L-glutamic acid

-
-
35011-47-3
2,4,5-triamino-6-hydroxypyrimidine sulfate

-
-
59-30-3
folate

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1-trichloroacetone; N-(4-aminobenzoyl)-L-glutamic acid With sodium acetate; sodium sulfite In acetic acid at 43℃; for 0.5h; Stage #2: 2,4,5-triamino-6-hydroxypyrimidine sulfate In acetic acid at 38℃; for 5h; | 75.5% |


| Conditions | Yield |
|---|---|
| In hexane at 25℃; | 75% |
-
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 25℃; for 1h; | 75% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
51425-58-2
(Z)-1-(trimethylsiloxy)-1-decene

| Conditions | Yield |
|---|---|
| Stage #1: (Z)-1-(trimethylsiloxy)-1-decene With methyllithium In tetrahydrofuran at 0℃; for 0.5h; Metallation; Stage #2: With tetrabutoxytitanium In tetrahydrofuran at 0℃; for 0.5h; complexation; Stage #3: 1,1,1-trichloroacetone In tetrahydrofuran at 0℃; for 1h; Condensation; | 72% |
-
-
918-00-3
1,1,1-trichloroacetone

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
1010796-56-1
(E)-3-dimethylamino-1,1,1-trichloro-3-buten-2-one

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In toluene at 110℃; for 24h; | 68% |
1,1,1-Trichloropropanone Chemical Properties
IUPAC Name: 1,1,1-Trichloropropan-2-one
The MF of 1,1,1-Trichloropropanone (CAS NO.918-00-3) is C3H3Cl3O.
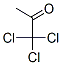
The MW of 1,1,1-Trichloropropanone (CAS NO.918-00-3) is 161.41.
Synonyms of 1,1,1-Trichloropropanone (CAS NO.918-00-3): 1,1,1-Trichloroacetone ; 2-Propanone, trichloro- ; 1,1, 1-Trichloro-2-propanone ; 2-Propanone, 1,1,1-trichloro-
Index of Refraction: 1.469
Density: 1.475 g/ml
Flash Point: 64.4 °C
Boiling Point: 134.5 °C
1,1,1-Trichloropropanone Uses
1,1,1-Trichloropropanone (CAS NO.918-00-3) is used as organic intermediates, fine chemicals, pharmaceutical research and development.
1,1,1-Trichloropropanone Toxicity Data With Reference
| 1. | mic-bac-sat 359 µg/plate | MUREAV Mutation Research. 155 (1985),53. | ||
| 2. | cyt-ham-ovr 23 mg/L | MUREAV Mutation Research. 206 (1988),431. |
1,1,1-Trichloropropanone Safety Profile
Mutation data reported. When heated to decomposition it emits toxic vapors of Cl−.Safety information of 1,1,1-Trichloropropanone (CAS NO.918-00-3):
Risk Statements
20/21/22 Harmful by inhalation, in contact with skin and if swallowed
36/37/38 Irritating to eyes, respiratory system and skin
Safety Statements
26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39 Wear suitable protective clothing, gloves and eye/face protection
RTECS UC3839000
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 918-01-4
- 918-02-5
- 91804-23-8
- 918-04-7
- 918-05-8
- 91807-59-9
- 91809-66-4
- 91809-67-5
- 918138-38-2
- 91-81-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View