-
Name
Urea nitrate
- EINECS 204-703-5
- CAS No. 124-47-0
- Article Data13
- CAS DataBase
- Density 1.6900
- Solubility
- Melting Point 152° with decompn
- Formula CH4N2O.HNO3
- Boiling Point 196.6 °C at 760 mmHg
- Molecular Weight 123.068
- Flash Point 72.7 °C
- Transport Information
- Appearance white granular / prill
- Safety 17-26-36/37/39-45
- Risk Codes 8-36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Acidogen nitrate;nitric acid; urea;Urea, mononitrate;Urea nitrate (wet);
- PSA 135.16000
- LogP 0.59990
Synthetic route

| Conditions | Yield |
|---|---|
| With nitric acid; acetic acid; propionic acid | |
| With nitric acid In water Heating; | |
| With nitric acid Heating; |
-
-
90802-01-0
2-amino-4,6-diureido-1,3,5-triazine

-
A
-
124-47-0
uronium nitrate

-
B
-
37640-57-6
cyanuric acid melamine

-
C
-
108-80-5
isocyanuric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride Heating; | A 34% B 34% C 70% |

-
-
90802-01-0
2-amino-4,6-diureido-1,3,5-triazine

-
A
-
645-93-2
ammelide

-
B
-
124-47-0
uronium nitrate

-
C
-
37640-57-6
cyanuric acid melamine

-
D
-
108-80-5
isocyanuric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride Heating; | A 2% B 34% C 4% D 70% |

-
-
4801-02-9
2,4,6-triureido-1,3,5-triazine

-
A
-
124-47-0
uronium nitrate

-
B
-
37640-57-6
cyanuric acid melamine

-
C
-
108-80-5
isocyanuric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 4h; Heating; | A 26% B 18% C 77% |


| Conditions | Yield |
|---|---|
| With water; nitric acid | |
| With diethyl ether; nitric acid |

| Conditions | Yield |
|---|---|
| In diethyl ether |
-
-
87-39-8
violuric acid

-
-
7722-84-1
dihydrogen peroxide

-
A
-
120-89-8
parabanic acid

-
B
-
124-47-0
uronium nitrate

-
-
124-47-0
uronium nitrate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In sulfuric acid | |
| With sodium hydroxide In sulfuric acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid cooling; | 80% |
| With sulfuric acid Giessen auf Eis und Ausziehen den Niederschlag wiederholt mit Wasser bei hoechstens 55grad; |

-
-
124-47-0
uronium nitrate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In sulfuric acid |
-
-
131921-19-2
4-(1,3-diphenylpropyl)-1H-imidazole

-
-
124-47-0
uronium nitrate

-
-
131920-91-7
4-[1,3-bis(4-nitrophenyl)propyl]-1H-imidazole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In sulfuric acid |

-
-
124-47-0
uronium nitrate

| Conditions | Yield |
|---|---|
| With KN3O4 In ethanol | 72% |
-
-
124-47-0
uronium nitrate

-
-
627-30-5
1-chloro-3-hydroxypropane

-
-
6326-12-1
carbamic acid-(3-chloro-propyl ester)

| Conditions | Yield |
|---|---|
| at 130 - 135℃; |
-
-
104054-13-9
4-(2,3-dihydro-1H-inden-2-yl)-1H-imidazole hydrochloride

-
-
7664-93-9
sulfuric acid

-
-
124-47-0
uronium nitrate

-
-
124-47-0
uronium nitrate

-
-
10268-78-7
1,3-diacetylaminobenzene

-
-
42783-40-4
1,5-bis(acetylamino)-2,4-dinitrobenzene
-
-
6282-12-8
2,4-diacetylaminotoluene

-
-
124-47-0
uronium nitrate

-
-
56850-44-3
4-methyl-2,6-dinitro-m-phenylenediamine

| Conditions | Yield |
|---|---|
| With nitric acid at 5 - 10℃; Verseifen des entstandenen Produktes mit verd.Schwefelsaeure; |

| Conditions | Yield |
|---|---|
| With sodium nitrite Darst; Ausbeute 70prozent; | |
| With sodium nitrite |

| Conditions | Yield |
|---|---|
| With cis-nitrous acid; silver nitrate |

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| at 125 - 130℃; |
-
-
124-47-0
uronium nitrate

-
-
127099-85-8, 780722-26-1
N-Cyanoguanidine

-
-
16783-51-0
N-carbamoylimidodicarbonimidic diamide

| Conditions | Yield |
|---|---|
| With ammonium hydroxide |

| Conditions | Yield |
|---|---|
| heating; |

| Conditions | Yield |
|---|---|
| (i) (heating), (ii) /BRN= 3703120/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| repeated treatment of (NH2)2CO*HNO3 with D2O; |

| Conditions | Yield |
|---|---|
| Aufbewahren des Reaktionsprodukts bei 20grad; |
-
-
124-47-0
uronium nitrate

-
A
-
108-80-5
cyanuric acid

-
C
-
124-38-9
carbon dioxide

-
D
-
57-13-6
urea

-
E
-
10024-97-2
dinitrogen monoxide

| Conditions | Yield |
|---|---|
| at 140°C; |

-
-
124-47-0
uronium nitrate

-
A
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

| Conditions | Yield |
|---|---|
| at 140℃; |


| Conditions | Yield |
|---|---|
| at 60 - 70℃; |


| Conditions | Yield |
|---|---|
| at 25℃; Equilibrium constant; |

Urea nitrate Standards and Recommendations
Urea nitrate Specification
The Urea nitrate is an organic compound with the formula CH4N2O.HNO3. The IUPAC name of this chemical is nitric acid; urea. With the CAS registry number 124-47-0, it is also named as Acidogen nitrate. Besides, it should be stored in a cool and dry place. It is a fertilizer-based high explosive that has been used in improvised explosive devices. It has a destructive power similar to better-known ammonium nitrate explosives, with a velocity of detonation between 11,155 ft/s (3,400 m/s) and 15,420 ft/s (4,700 m/s).
Physical properties about Urea nitrate are: (1)ACD/LogP: -2.11; (2)ACD/LogD (pH 5.5): -2.11; (3)ACD/LogD (pH 7.4): -2.11; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 1.69; (7)ACD/KOC (pH 7.4): 1.69; (8)#H bond acceptors: 3; (9)#H bond donors: 4; (10)Polar Surface Area: 23.55 Å2; (11)Flash Point: 72.7 °C; (12)Enthalpy of Vaporization: 43.28 kJ/mol; (13)Boiling Point: 196.6 °C at 760 mmHg; (14)Vapour Pressure: 0.395 mmHg at 25°C.
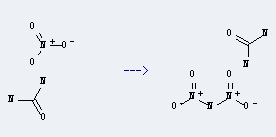
Preparation: this chemical can be prepared by N,N'',N'''-[1,3,5]triazine-2,4,6-triyl-tri-urea. This reaction will need reagent HCl. The reaction time is 4 hours by heating. The yield is about 26%.
.gif)
.gif)
.gif)
.gif)
Uses of Urea nitrate: it can be used to produce urea; compound with generic inorganic neutral component. It will need reagent KN3O4 and solvent ethanol. The yield is about 72%.
When you are using this chemical, please be cautious about it as the following:
It contacts with combustible material may cause fire. Please keep away from combustible material. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. Besides, this chemical is irritating to eyes, respiratory system and skin. When you are using it, wear suitable gloves and eye/face protection. In case of accident or if you feel unwell seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(N)N.[O-][N+](=O)O
(2)InChI: InChI=1/CH4N2O.HNO3/c2*2-1(3)4/h(H4,2,3,4);(H,2,3,4)
(3)InChIKey: AYTGUZPQPXGYFS-UHFFFAOYAL
(4)Std. InChI: InChI=1S/CH4N2O.HNO3/c2*2-1(3)4/h(H4,2,3,4);(H,2,3,4) CopyCopied
(5)Std. InChIKey: AYTGUZPQPXGYFS-UHFFFAOYSA-N
Related Products
- Urea
- Urea nitrate
- Urea, 1-(2-chloroethyl)-1-nitroso-3-phenyl- (8CI)
- Urea, 1-(2-chloroethyl)-3-(4-methylcyclohexyl)-1-nitroso-, trans- (8CI)
- Urea, 1-(fluoroacetyl)-3-phenyl-
- UREA, 1,1,3-TRIETHYL-
- Urea, 1-benzoyl-2-thio-3-p-tolyl-
- Urea, 1-ethyl-1-hydroxy-
- Urea, 1-ethyl-1-nitroso-3-phenyl-
- Urea, 2-acetyl-1-phenyl-2-thio-
- 124473-12-7
- 124474-51-7
- 1244-76-4
- 124478-60-0
- 124480-95-1
- 124-48-1
- 124485-98-9
- 124491-38-9
- 124491-42-5
- 124495-18-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View