Hubei Lidu New Material Technology Co., Ltd
Hubei Lidu New Materials Technology Co. , Ltd. is located in No. 12,3C Industrial Park, Zhongxiang Economic Development Zone, Hubei Province. The company is committed to fine chemical raw materials, pharmaceutical intermediates and other production,
Cas:145040-37-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHubei Yuanmeng Biological Technology Co., Ltd.
Hubei Yuanmeng Biological Technology Co., Ltd., which is located in Wuhan, China. We are specializing in the exportation of APIs, and plant extracts ect. Our products has been exported to America, Australia, Brazil, the Europe, Middle East and other
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $3.0 / 5.0
Type:Trading Company
inquiryDayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySinoway Industrial Co., Ltd.
We are very compeitive on candesartan cilexetil .Our mfr is GMP certified for this item with CEP document. Candesartan cilexetil is a prodrug of the potent, long-acting, and selective angiotensin II type 1 receptor AT1 antago
Cas:145040-37-5
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:145040-37-5
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% ----------------------------------------------------------------
Cas:145040-37-5
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:145040-37-5
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages for Candesartan cilexetil Cas 145040-37-5 Guaranteed purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:White to off white crystalline and odourless power Storage:Cool dr
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $800.0 / 900.0
Type:Trading Company
inquiryHefei TNJ chemical industry co.,ltd
Our Advantages: GMP factory Full experience of large numbers containers loading in main Chinese seaports Packing with pallet as buyer's special request Best service after shipment with e-mail Cargos together with container sales s
Hangzhou Think Chemical Co. Ltd
Candesartan Cilexetil[CAS No:145040-37-5] Trityl Candesartan[CAS:139481-72-4] Trityl Candesartan Cilexetil[CAS:170791-09-0] Name Candesartan cilexetil Synonyms 1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:145040-37-5
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryEnke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api, intermedi
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryXi`an Eastling Biotech Co., Ltd.
145040-37-5 - Physico-chemical Properties Molecular Formula C33H34N6O6 Molar Mass 610.66 Density 1.37±0.1 g/cm3(Predicted) Melting Point 168-170 C Boling Point 843.3±75.0 °
Cas:145040-37-5
Min.Order:1 Gram
FOB Price: $8.0 / 9.0
Type:Lab/Research institutions
inquiryShandong Jiulong Hisince Pharmaceutical Co.,Ltd.
Assay: NLT 99% Appearance:whilt powder Storage:cool and dryer Package:durm Application:API Transportation:by sea or by air Port:shanghai
Cas:145040-37-5
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryCOLORCOM LTD.
Colorcom is a global leader in industrial chemical manufacturing and is continuously innovating and transforming to exceed client expectations and industry standards. Colorcom prides itself on superior customer and technical focus, while focusing on
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:145040-37-5
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryKono Chem Co.,Ltd
Xi'an Kono Chem Co., Ltd., founded in 2014, is a holding enterprise of Hongkong Pioneer Biotech Group. It is an export-oriented manufacturing enterprise supported by the Ministry of Commerce. Kono Chem is located in Xi'an, Shaanxi Provin
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $450.0 / 500.0
Type:Other
inquiryHubei DiBo chemical co., LTD
Candesartan cilexetil Chinese name: candesartan ester Chinese alias: 2 ethoxy - 1 - [(2 '- (1 h - four azole nitrogen - 5 - base) [1, 1' - al - 4 - base] methyl phenyl]] - 1 h - benzene and imidazole - 7 - formic acid - 1 - [[(cyclohexano
Cas:145040-37-5
Min.Order:25 Metric Ton
FOB Price: $2.0 / 3.0
Type:Other
inquiryLIDE PHARMACEUTICALS LIMITED
LIDE PHARMACEUTICALS LIMITED is a professional chemicals and APIs leading manufacturer in China. Our core business line covers APIs, Intermediates, Herb extract, etc.
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $0.9 / 1.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Candesartan cilexetil Basic information The precursor drug of Antihypertensive drug Candesartan---Candesartan cilexetil Candesartan cilexetil tabletsAppendix I Appendix II Side effects and adverse reactions Precautions App
Cas:145040-37-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:145040-37-5
Min.Order:1 Gram
FOB Price: $1000.0 / 1300.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $7.0 / 9.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Name: Candesartan cilexetil CAS NO.:145040-37-5 Molecular formula: C33H34N6O6 Molecular weight: 610.66 Appearance:White crystalline powder Storage:Store in cool and dry place, away from sun light. Package:25kg Application:Antihypertensive pharma
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Other
inquiryHubei Langyou International Trading Co., Ltd
Advantages: Hubei XinRunde Chemical Co., Ltd is a renowned pharmaceutical manufacturer. We can offer high quality products at competitive price in quick delivery with 100% custom pass guaranteed. Never stop striving to offer our best s
Cas:145040-37-5
Min.Order:10 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:145040-37-5
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHANWAYS CHEMPHARM CO.,LIMITED
Hanways Chempharm Co., Limited, the former is Hubei Hanways Pharchem CO.,Limited, set up in 2009 in Wuhan, China. We specialize in sourcing and supplying APIs, pharmaceutical intermediates, and fine chemicals for worldwide markets. The founder has d
Cas:145040-37-5
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Trading Company
inquiryShanghai Minstar Chemical Co., Ltd
Candesartan cilexetil Chemical Properties Melting point 168-170 C Boiling point 843.3±75.0 °C(Predicted) density 1.37±0.1 g/cm3(Predicted) storage temp. 2-8°C solubility DMSO: ≥15mg/mL form powder pka
Cas:145040-37-5
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Fonlynn Health Technology Co., Ltd.
Packing: According to customer requirements Delivery time: In stock or depands Port of shipment: Ningbo/Shanghai/Qingdao OEM/ODM:Welcome Sample:We can offer our existing samples at once Appearance:white powder Storage:Refer to COA / MSDS Packag
Cas:145040-37-5
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquirySynthetic route
-
-
170791-09-0
cilexetil trityl candesartan

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol; dichloromethane at -14 - -12℃; Temperature; Flow reactor; Large scale; | 98% |
| With iron(III) chloride In methanol; dichloromethane at 23℃; for 48h; Time; Reagent/catalyst; | 96% |
| With methanol; zinc(II) chloride In butanone at 20 - 40℃; for 2.5h; Product distribution / selectivity; | 95.23% |
-
-
78-09-1
orthocarbonic acid tetraethyl ester

-
-
1236156-65-2
2-(((2'-(1H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)amino)-3-aminobenzoic acid-1-(((cyclohexyloxy)carbonyl)oxy)ethyl ester

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With acetic acid In ethyl acetate at 30℃; Temperature; | 93.9% |
| With acetic acid In toluene at 60℃; for 2h; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: C30H32N6O5*C2H2O4 With potassium carbonate In ethyl acetate at 10 - 15℃; pH=6 - 7; Stage #2: orthocarbonic acid tetraethyl ester In acetic acid at 0 - 25℃; for 15h; | 92.3% |
-
-
1307853-85-5
1-{[(cyclohexyloxy)carbonyl]oxy}ethyl 1-({4-[2-(1-benzyl-1H-1,2,3,4-tetrazol-5-yl)phenyl]phenyl}methyl)-2-ethoxy-1H-1,3-benzodiazole-7-carboxylate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With 5% palladium on barium sulphate; ammonium formate In water; isopropyl alcohol at 25℃; for 14h; Temperature; Time; | 92.1% |
| With 5% palladium on barium sulphate; ammonium formate In water; isopropyl alcohol at 25℃; for 14h; | 80% |
| With ammonium formate; palladium over charcoal In water; isopropyl alcohol at 55 - 60℃; for 12 - 15h; | |
| With rosenmund catalyst; ammonium formate In water; isopropyl alcohol |

| Conditions | Yield |
|---|---|
| With sodium bicarbonate In methanol; dichloromethane; water; ethyl acetate | 75% |
-
-
139481-72-4
2-Ethoxy-1-[[2'-(N-triphenylmethyltetrazol-5-yl)-biphenyl-4-yl]methyl]benzimidazole-7-carboxylic acid

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With water In methanol; toluene for 2 - 4h; Product distribution / selectivity; Heating / reflux; | 67% |
-
-
1307853-79-7
C41H42N6O7

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With ammonium formate; 5%-palladium/activated carbon In water; isopropyl alcohol at 40 - 50℃; for 17h; | 55% |
-
-
139481-72-4
2-Ethoxy-1-[[2'-(N-triphenylmethyltetrazol-5-yl)-biphenyl-4-yl]methyl]benzimidazole-7-carboxylic acid

-
-
95789-69-8, 102672-57-1
cyclohexyl (1-iodoethyl)carbonate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol; ethanol; water; N,N-dimethyl-formamide |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol; ethanol; hexane; dichloromethane; water; acetone |

-
-
170791-09-0
cilexetil trityl candesartan

-
A
-
869631-11-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-oxo-3-[[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-2,3-dihydro-1H-benzimidazole-4-carboxylate

-
B
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With methanol for 24h; Product distribution / selectivity; Heating / reflux; | |
| With methanol; formic acid In dichloromethane at 25℃; for 5 - 23h; Product distribution / selectivity; | |
| Stage #1: cilexetil trityl candesartan With methanol; water; zinc(II) chloride for 2.5h; Heating / reflux; Stage #2: With water; sodium hydrogencarbonate In methanol at 20℃; pH=6.11; Product distribution / selectivity; |
-
-
632322-62-4
2-ethoxy-1-[(2'-cyanobiphenyl-4-yl)methyl]-1H-benzimidazole-7-carboxylicacid-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl ester

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With azido tributyltin (IV) In toluene for 52.5h; Heating / reflux; |
-
-
99464-83-2
1-chloroethyl cyclohexyl carbonate

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
139481-59-7
candesartan

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Stage #1: tert-butyldimethylsilyl chloride; candesartan With triethylamine In chloroform at 20℃; for 3h; Stage #2: 1-chloroethyl cyclohexyl carbonate With triethylamine In chloroform for 24h; |

-
-
139481-59-7
candesartan

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: triethylamine / dichloromethane / 3.75 h / 25 - 35 °C 2.1: potassium carbonate / N,N-dimethyl-formamide / 2.5 h / 60 - 65 °C 3.1: formic acid; methanol / dichloromethane / 7 h / 30 - 35 °C 3.2: 0.33 h / 15 - 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 2: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 3: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme | |
| Multi-step reaction with 3 steps 1: triethylamine / dichloromethane / 3.5 h / 15 - 23 °C / Flow reactor; Large scale 2: potassium carbonate / N,N-dimethyl-formamide / 4 h / 60 °C / Flow reactor; Alkaline conditions; Large scale 3: hydrogenchloride / dichloromethane; methanol / -14 - -12 °C / Flow reactor; Large scale View Scheme |
-
-
21606-04-2
methyl 2-carboxy-3-nitrobenzoate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: N,N-dimethyl-formamide; thionyl chloride / toluene / 2.5 h / 75 °C 2.1: sodium azide; tetrabutylammomium bromide / toluene / 4 h / -10 - -5 °C 2.2: 1 h / 80 - 85 °C 3.1: potassium carbonate; tetrabutylammomium bromide / toluene / 12 h / 80 - 85 °C 3.2: 3 h / Reflux 4.1: tin(ll) chloride / ethyl acetate / 1.5 h / Reflux 4.2: 4.5 h / -10 - 0 °C 4.3: 0.5 h / Reflux 5.1: acetic acid / toluene / 6 h / Reflux 6.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 6.2: 0 - 20 °C 7.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 7.2: 3 h / 0 °C / pH 4 8.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 9.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 10.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme | |
| Multi-step reaction with 8 steps 1.1: triethylamine; diphenyl phosphoryl azide / N,N-dimethyl-formamide / 20 - 31 °C / Inert atmosphere 1.2: 9 h / 85 - 87 °C 1.3: 3 h / 50 - 55 °C 2.1: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 3.1: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 4.1: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 5.1: acetic acid / 1 h / 90 °C 6.1: sodium hydroxide / methanol / 2 h / 90 °C 7.1: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 8.1: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
57113-90-3
methyl 2-(N-tert-butoxycarbonylamino)-3-nitrobenzoate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1.1: potassium carbonate; tetrabutylammomium bromide / toluene / 12 h / 80 - 85 °C 1.2: 3 h / Reflux 2.1: tin(ll) chloride / ethyl acetate / 1.5 h / Reflux 2.2: 4.5 h / -10 - 0 °C 2.3: 0.5 h / Reflux 3.1: acetic acid / toluene / 6 h / Reflux 4.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 4.2: 0 - 20 °C 5.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 5.2: 3 h / 0 °C / pH 4 6.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 7.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 8.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme | |
| Multi-step reaction with 7 steps 1: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 2: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 3: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 4: acetic acid / 1 h / 90 °C 5: sodium hydroxide / methanol / 2 h / 90 °C 6: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 7: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
139481-69-9
methyl 2-ethoxy-1-[[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]benzimidazole-7-carboxylate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 1.2: 3 h / 0 °C / pH 4 2.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 3.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 4.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme |
-
-
136304-78-4
methyl 3-amino-2-[[(2'-cyanobiphenyl-4-yl)methyl]amino]benzoate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: acetic acid / toluene / 6 h / Reflux 2.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 2.2: 0 - 20 °C 3.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 3.2: 3 h / 0 °C / pH 4 4.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 5.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 6.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme |
-
-
603-11-2
3-nitrophthalic acid

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: sulfuric acid / 24.75 h / 20 - 65 °C 2.1: N,N-dimethyl-formamide; thionyl chloride / toluene / 2.5 h / 75 °C 3.1: sodium azide; tetrabutylammomium bromide / toluene / 4 h / -10 - -5 °C 3.2: 1 h / 80 - 85 °C 4.1: potassium carbonate; tetrabutylammomium bromide / toluene / 12 h / 80 - 85 °C 4.2: 3 h / Reflux 5.1: tin(ll) chloride / ethyl acetate / 1.5 h / Reflux 5.2: 4.5 h / -10 - 0 °C 5.3: 0.5 h / Reflux 6.1: acetic acid / toluene / 6 h / Reflux 7.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 7.2: 0 - 20 °C 8.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 8.2: 3 h / 0 °C / pH 4 9.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 10.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 11.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme |
-
-
139481-28-0
methyl 2-<<2'-cyanobiphenyl-4-yl)methyl>amino>-3-nitrobenzoate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1.1: tin(ll) chloride / ethyl acetate / 1.5 h / Reflux 1.2: 4.5 h / -10 - 0 °C 1.3: 0.5 h / Reflux 2.1: acetic acid / toluene / 6 h / Reflux 3.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 3.2: 0 - 20 °C 4.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 4.2: 3 h / 0 °C / pH 4 5.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 6.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 7.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme |
-
-
73833-13-3
acid chloride of 1-methylhydrogen-3-nitrophthalate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 9 steps 1.1: sodium azide; tetrabutylammomium bromide / toluene / 4 h / -10 - -5 °C 1.2: 1 h / 80 - 85 °C 2.1: potassium carbonate; tetrabutylammomium bromide / toluene / 12 h / 80 - 85 °C 2.2: 3 h / Reflux 3.1: tin(ll) chloride / ethyl acetate / 1.5 h / Reflux 3.2: 4.5 h / -10 - 0 °C 3.3: 0.5 h / Reflux 4.1: acetic acid / toluene / 6 h / Reflux 5.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 5.2: 0 - 20 °C 6.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 6.2: 3 h / 0 °C / pH 4 7.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 8.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 9.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme |

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: sodium azide; tributyltin chloride / toluene / 100 h / Reflux 1.2: 0 - 20 °C 2.1: sodium hydroxide; water / ethanol / 3.5 h / Reflux 2.2: 3 h / 0 °C / pH 4 3.1: triethylamine / dichloromethane / 8 h / 0 - 20 °C 4.1: potassium carbonate; potassium iodide / N,N-dimethyl-formamide / 2 h / 75 °C 5.1: hydrogen / palladium 10% on activated carbon / toluene; methanol / 20 °C / 2280.15 Torr View Scheme |

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| With hydrogen; palladium 10% on activated carbon In methanol; toluene at 20℃; under 2280.15 Torr; Product distribution / selectivity; |
-
-
98-88-4
benzoyl chloride

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 12 steps 1.1: triethylamine / tetrahydrofuran / 7 - 16 °C 2.1: phosphorus pentachloride / dichloromethane / -11 - 19 °C 2.2: 7 h / -6 - 20 °C 3.1: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 4.1: sodium methylate / methanol / 1.5 h / 20 °C 5.1: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 6.1: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 7.1: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 8.1: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 9.1: acetic acid / 1 h / 90 °C 10.1: sodium hydroxide / methanol / 2 h / 90 °C 11.1: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 12.1: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
21388-92-1
4-bromobenzyl acetate

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 2: sodium methylate / methanol / 1.5 h / 20 °C 3: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 4: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 5: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 6: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 7: acetic acid / 1 h / 90 °C 8: sodium hydroxide / methanol / 2 h / 90 °C 9: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 10: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
873-75-6
4-bromobenzenemethanol

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1: dmap; triethylamine / tetrahydrofuran / 15 - 30 °C 2: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 3: sodium methylate / methanol / 1.5 h / 20 °C 4: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 5: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 6: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 7: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 8: acetic acid / 1 h / 90 °C 9: sodium hydroxide / methanol / 2 h / 90 °C 10: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 11: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
18039-42-4
5-Phenyl-1H-tetrazole

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: sodium carbonate; tetrabutylammomium bromide / chloroform; water / 2 h / 55 °C / Cooling with ice 1.2: 1 h 2.1: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 3.1: sodium methylate / methanol / 1.5 h / 20 °C 4.1: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 5.1: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 6.1: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 7.1: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 8.1: acetic acid / 1 h / 90 °C 9.1: sodium hydroxide / methanol / 2 h / 90 °C 10.1: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 11.1: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme | |
| Multi-step reaction with 11 steps 1: sodium carbonate; tetrabutylammomium bromide / chloroform; water / 2 h / 55 °C / Cooling with ice 2: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 3: sodium methylate / methanol / 1.5 h / 20 °C 4: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 5: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 6: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 7: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 8: acetic acid / 1 h / 90 °C 9: sodium hydroxide / methanol / 2 h / 90 °C 10: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 11: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
512182-38-6
1-(p-methoxybenzyl)-5-phenyl-1H-tetrazole

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 2: sodium methylate / methanol / 1.5 h / 20 °C 3: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 4: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 5: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 6: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 7: acetic acid / 1 h / 90 °C 8: sodium hydroxide / methanol / 2 h / 90 °C 9: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 10: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
41882-10-4
N-(4-methoxybenzyl)benzamide

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 11 steps 1.1: phosphorus pentachloride / dichloromethane / -11 - 19 °C 1.2: 7 h / -6 - 20 °C 2.1: potassium carbonate; triphenylphosphine / dichloro(1,5-cyclooctadiene)ruthenium(II) polymer / 1-methyl-pyrrolidin-2-one / 2 h / 140 °C / Inert atmosphere 3.1: sodium methylate / methanol / 1.5 h / 20 °C 4.1: phosphorus tribromide / tetrahydrofuran / 5.5 h / 0 - 20 °C 5.1: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 6.1: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 7.1: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 8.1: acetic acid / 1 h / 90 °C 9.1: sodium hydroxide / methanol / 2 h / 90 °C 10.1: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 11.1: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
1307853-40-2
5-[4'-(bromomethyl)biphenyl-2-yl]-1-(p-methoxybenzyl)-1H-tetrazole

-
-
145040-37-5
candesartan cilexetil

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: potassium carbonate / acetonitrile / 6 h / Reflux; Inert atmosphere 2: hydrogenchloride / methanol / 3.22 h / 3 - 9 °C 3: tin(II) chloride dihdyrate / methanol / 2 h / Reflux 4: acetic acid / 1 h / 90 °C 5: sodium hydroxide / methanol / 2 h / 90 °C 6: potassium carbonate / N,N-dimethyl-formamide / 4 h / 65 °C 7: ammonium formate / 5%-palladium/activated carbon / isopropyl alcohol; water / 17 h / 40 - 50 °C View Scheme |
-
-
145040-37-5
candesartan cilexetil

-
-
869631-11-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-oxo-3-[[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-2,3-dihydro-1H-benzimidazole-4-carboxylate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In acetone for 6h; Reflux; | 87.9% |

| Conditions | Yield |
|---|---|
| In ethanol at 20 - 45℃; for 5h; Concentration; Solvent; Temperature; Time; | 81% |
-
-
145040-37-5
candesartan cilexetil

-
-
74-88-4
methyl iodide

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 79% |
-
-
145040-37-5
candesartan cilexetil

-
A
-
869631-11-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-oxo-3-[[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-2,3-dihydro-1H-benzimidazole-4-carboxylate

-
B
-
914613-35-7
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2'-(1-ethyl-1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-1H-benzimidazole-7-carboxylate

-
C
-
914613-36-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2'-(2-ethyl-2H-tetrazol-5-yl)biphenyl-4-yl]methyl]-1H-benzimidazole-7-carboxylate

| Conditions | Yield |
|---|---|
| at 55℃; for 336h; Product distribution / selectivity; | |
| at 55℃; for 336h; Product distribution / selectivity; |

-
-
145040-37-5
candesartan cilexetil

-
A
-
869631-11-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-oxo-3-[[2'-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-2,3-dihydro-1H-benzimidazole-4-carboxylate

-
B
-
914613-36-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2'-(2-ethyl-2H-tetrazol-5-yl)biphenyl-4-yl]methyl]-1H-benzimidazole-7-carboxylate

| Conditions | Yield |
|---|---|
| In ethanol at 50℃; for 31h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| With triethylamine for 1h; Heating / reflux; |
-
-
75-03-6
ethyl iodide

-
-
145040-37-5
candesartan cilexetil

-
A
-
914613-35-7
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2'-(1-ethyl-1H-tetrazol-5-yl)biphenyl-4-yl]methyl]-1H-benzimidazole-7-carboxylate

-
B
-
914613-36-8
(1RS)-1-[[(cyclohexyloxy)carbonyl]oxy]ethyl 2-ethoxy-1-[[2'-(2-ethyl-2H-tetrazol-5-yl)biphenyl-4-yl]methyl]-1H-benzimidazole-7-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: candesartan cilexetil With potassium carbonate In acetone at 20℃; for 1h; Inert atmosphere; Stage #2: ethyl iodide In acetone at 20℃; for 24h; | A 2.5 g B 2.9 g |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View


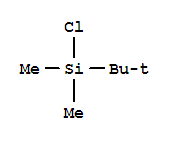




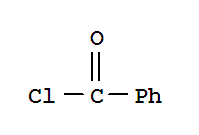

 Xn
Xn