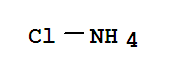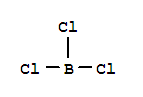Hebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:6569-51-3
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:6569-51-3
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine chemicals, ph
Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:6569-51-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
BORAZINE CAS:6569-51-3 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, st
Cas:6569-51-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Superiority We can customize and synthesize products that other suppliers may not be able to provide. Advantage cof, mof ligand manufacturer Product Detail
Cas:6569-51-3
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Hebei Quanhe Biotechnology Co. LTD
1. Timely and efficient service to ensure communication with customers 2. Produce products of different specifications and sizes according to your requirements. 3. Quality procedures and standards recognized by SGS. Advanced plant equipment ensures s
Henan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Triumph International Development Limilted
Appearance:white or yellowish white flaky crystal Storage:Keep the sun from direct sunlight ;prevention against high temperature Package:As customer request Application:Used for research and industrial manufacture. Transportatio
Wuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:6569-51-3
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
SHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
Cas:6569-51-3
Min.Order:100 Gram
FOB Price: $100.0 / 2000.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:flammable liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Used as Pharma
TaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Hebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:6569-51-3
Min.Order:1 Kilogram
FOB Price: $8.0 / 10.0
Type:Trading Company
inquiryJiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:6569-51-3
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:6569-51-3
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:6569-51-3
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Bluecrystal chem-union
We are a Union of chemistry in China, consists of chemists,engineers, laboratories,factories in China. We organize surplus capacity of R&D and production as well as custom synthesis for chemical products and chemical business project. We are supp
Cas:6569-51-3
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Trading Company
inquiryHebei Mojin Biotechnology Co.,Ltd
1, High quality with competitive price:2, Fast and safe delivery3.Excellent pre-sales and after-sales service4. Well-trained and professional technologist and sales with rich experience in the field for 5-10 yearsAppearance:see detailed specification
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:6569-51-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Synthetic route

| Conditions | Yield |
|---|---|
| 92.6% | |
| 92.6% | |
| In diethylene glycol | 46% |

| Conditions | Yield |
|---|---|
| aluminium trichloride In further solvent(s) (N2), powder of (NH4)2SO4 treated with soln. of NaBH4 in tetraglyme, stirred for 6 h at 140°C catalized by AlCl3; | 67% |
| In further solvent(s) byproducts: Na2SO4, H2; absence of air and moisture; heating in tetraglyme to 135°C (over 1 h), then 1 h at 135°C, continuous distn. off of borazine and H2 at 2-5 Torr; condensation in trap at liquid-N2 temp., fractional low-temp. distn. (collection at -78°C); | 59.9% |
| In further solvent(s) byproducts: Na2SO4, H2; absence of air and moisture; stepwise addn. of solid mixt. of educt into tetraglyme at 135°C, heating to 135°C for 2 h; condensation in trap at liquid-N2 temp., fractional low-temp. distn. (collection at -78°C); | 53.8% |
-
-
10043-11-5
ammonia borane complex

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| In further solvent(s) byproducts: H2; absence of air and moisture; slow addn. of solid H3NBH3 into tetraglymeat 140-160°C (over 3 h), distn. off of H2 and product (2-5 Torr); condensation in trap at liquid-N2 temp., fractional low-temp. distn. (collection at -78°C); | 67% |
| With nickel at 80℃; under 30 Torr; for 6h; Inert atmosphere; | 53% |
| In 1,2-dimethoxyethane |

| Conditions | Yield |
|---|---|
| 65% | |
| 65% | |
| In dibutyl ether under N2; exclusion of moisture (drybox); B-trichloroborazine was suspended in the dry solvent; soln. of LiBH4 was added to the slurry over 2 h; crude borazine was distilled into a dry-ice condenser; several line transfers; fractional distillation; identified by IR; (1)H-NMR; boiling point;; |

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| [Re(NO)(η(2)-H2)Br2(PiPr3)2] In 1,4-dioxane byproducts: H2; at 45°C for 24 h, or at 85°C for 1 h; | 65% |
| chloro(1,5-cyclooctadiene)rhodium(I) dimer In further solvent(s) byproducts: H2; at 45°C for 72 h in tetraglime; NMR; | 10% |
| chloro(1,5-cyclooctadiene)rhodium(I) dimer In diethylene glycol dimethyl ether byproducts: H2; at 45°C for 72 h; NMR; | 10% |
-
-
19287-45-7
diborane

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| With ammonia byproducts: H2; quickly heating at 250-300°C; | 47% |
| With NH3 byproducts: H2; quickly heating at 250-300°C; | 47% |
| With ammonia byproducts: H2; heating at 200°C and 11 atm; 15 min; | 41.5% |

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| heating at 200°C 5 min; | 45% |
| heating at 200°C 5 min; | 45% |
| byproducts: H2; at 200°C 5 min; | >99 |

| Conditions | Yield |
|---|---|
| 40% | |
| 40% |

| Conditions | Yield |
|---|---|
| In solid byproducts: H2; heated in Schlenk vessel up to 470 K; held at this temp. for 6 h before slowly cooled down; monitored by NMR spectroscopy; | A n/a B 40% |

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| With ammonia heating at 200°C; | 40% |
| With NH3 heating at 200°C; | 40% |
| at 200°C in sealed tube; | |
| at 200°C in sealed tube; |

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| quickly heating to 200°C; | 30% |
| quickly heating to 200°C; | 30% |

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| fast heating up to 200°C; | 30% |
| fast heating up to 200°C; | 30% |

| Conditions | Yield |
|---|---|
| In diethylene glycol 30 min, 130°C; identified by 11B NMR spectroscopy;; | A 27% B n/a |

| Conditions | Yield |
|---|---|
| 23% | |
| 23% |
-
-
16940-66-2
sodium tetrahydroborate

-
A
-
10043-11-5
ammonia borane complex

-
B
-
75885-49-3
μ-aminodiborane

-
C
-
13871-09-5
cyclotriborane

-
D
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| In further solvent(s) byproducts: H2; under Ar atm.; grinded (NH4)2SO4 and NaBH4 stirred in triglyme heated to120°C for 30 min, treated at this temp. until the cease of gases released; mixt. distd. several times under high vac., collected in liq. N2 traps, condensed at -78°C; detd. by XRD, NMR, mass-spectrometry; | A n/a B n/a C n/a D 18% |


| Conditions | Yield |
|---|---|
| Stage #1: B,B',B''-trichloroborazine With pyridine In diethyl ether at -78℃; for 0.0833333h; Inert atmosphere; Schlenk technique; Glovebox; Stage #2: lithium borohydride at 20℃; for 0.5h; Inert atmosphere; Schlenk technique; Glovebox; | A 17% B n/a |


| Conditions | Yield |
|---|---|
| byproducts: H2; decompn. at different temp. values; | A n/a B 15% |
| byproducts: H2; decompn. at different temp. values; | A n/a B 15% |

| Conditions | Yield |
|---|---|
| cobalt at 175 - 180°C, heating; | |
| cobalt at 175 - 180°C, heating; |

-
-
6569-51-3
borazine

| Conditions | Yield |
|---|---|
| 200°C; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; thermal decompn. (vac., 1 Pa, glass ampoule in stainless steel capsule, 120°C, 95 min); further products; mass spectrometry; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) Kinetics; thermal decompn. (vac., 1 Pa, glass ampoule in stainless steel capsule, 47°C, 25 min or 60°C, 50 min); further products; mass spectrometry; |

| Conditions | Yield |
|---|---|
| byproducts: H2; 60-70°C, then 3 h, 85-90°C; | |
| byproducts: H2; 60-70°C, then 3 h, 85-90°C; |


| Conditions | Yield |
|---|---|
| byproducts: H2; 60-70°C, then 3 h, 85-90°C; | |
| byproducts: H2; 60-70°C, then 3 h, 85-90°C; |


| Conditions | Yield |
|---|---|
| byproducts: H2; 60-70°C, then 3 h at 85-90°C; | |
| byproducts: H2; 60-70°C, then 3 h at 85-90°C; |

| Conditions | Yield |
|---|---|
| With NH3 NH3:B2H6 mol ratio = 9:1; | A n/a B 0% |
| With NH3 heating with NH3 excess; |

| Conditions | Yield |
|---|---|
| With NH3 with B2H6 excess; | |
| With NH3 with B2H6 excess; |

| Conditions | Yield |
|---|---|
| In diethylene glycol | |
| In diethylene glycol dimethyl ether |

| Conditions | Yield |
|---|---|
| With water | |
| With H2O |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and ethylene (7.9:1 ratio) were condensed (-196°C) to acatalytic amt. Rh-complex, mixt. allowed to warm to room temp., initialyellow soln. turned reddish brown within min and became homogeneous, mixt. stirred 1 h; flask frozen on vacuum line (-196°C) and degassed, volatile material fractionated through a -78, -110 and -196°C trap series, product found in the -78°C trap; | 100% |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and propylene (8.5 to 7.5 :1) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 2 h; vacuum line fractionation of mixt. through a -63, -110 and -196°C trap series, product found in the -63°C trap; | 98% |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and the propene (ratio 8.4:1) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 1 h; vacuum line fractionation of mixt. through a 0, -45, -110 and -196°C trap series, product found in the -45°C trap, elem. anal.; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 2℃; for 35h; | 95% |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and excess ethylene (1:3 ratio) were condensed (-196°C)to a catalytic amt. Rh-complex, mixt. allowed to warm to room temp., mixt. stirred 2 h; flask frozen on vacuum line (-196°C) and degassed, volatile material fractionated through traps at -63 and -196°C, product found in the -63°C trap; | 92% |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and 1-butene (ratio 5.8:1) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 3 h; vacuum line fractionation of mixt. through a -45, -110 and -196°C trap series, product found in the -45°C trap, elem. anal.; | 91% |
-
-
677-21-4
1,1,1-trifluoropropylene

-
-
6569-51-3
borazine

-
A
-
155862-04-7
2-(trifluoropropyl)borazine

-
B
-
155862-05-8
2,4-bis-(trifluoropropyl)borazine

-
C
-
155862-06-9
2,4,6-tris-(trifluoropropyl)borazine

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and excess of the propene (ratio 1:2.9) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 16 h, resulting product was a solid with a moist appearance; vacuum line fractionation of mixt. through a -78 and -196°C traps, 2-substd. and 2,4-products found in the -78°C trap; extn. (CH2Cl2) of solid residue, evapn. (ac.), solid sublimed at 80°C (2,4,6-product), elem. anal.; | A 14% B 84% C n/a |


| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and the styrene (ratio 4:1) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 76 h; vacuum line fractionation of mixt. through a 0, -45 and -196°C trap series, product found in the 0°C trap; | 74% |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and trans-2-butene (ratio 6:1) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 185 h; vacuum line fractionation of mixt. through a -45, -110 and -196°C trap series, product found in the -45°C trap; | 72% |

| Conditions | Yield |
|---|---|
| carbonylhydridetris(triphenylphosphine)rhodium(I) room temp.; 4 h;; | 71.8% |
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) RhH(CO9(PPh3)3 in a Fischer-Porter glass pressure vessel, evacuating, condensing B3N3H6 and C2H2 into the flask at -196°C, allowing to warm to room temp., 4 h; attaching to vac. line, freezing at -196°C, degassing, fractionating through a -30,-70,-116,-196 °C trap series; | 71.8% |
| With catalyst: Ir(I)-compound In not given 55°C; |
-
-
74-85-1
ethene

-
-
6569-51-3
borazine

-
A
-
88916-94-3
2-ethylcyclotriborazine

-
B
-
89123-50-2
B3H(C2H5)2(NH)3

-
C
-
7443-22-3
2,4,6-triethyl-borazine

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and excess ethylene (1:3.5 ratio) were condensed (-196°C) to a catalytic amt. Rh-complex, mixt. allowed to warm to room temp., mixt. turned reddish brown within min, mixt. stirred 26 h; flask frozen on vacuum line (-196°C) and degassed, volatile material fractionated through traps at -78, -110 and -196°C, productsfound in the -78°C trap, identified by GC/MS; | A 65% B 29.5% C 5.5% |


| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and excess propylene (1:3.3) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 1 h; vacuum line fractionation of mixt. through a -63, -110 and -196°C trap series, products found in the -63°C trap, products sepd. by vacuum line fractionation with condensation in -30 and 0°C traps (2,4- respectively 2,4,6-product); | A 39.5% B 60.5% |

-
-
6569-51-3
borazine

-
-
10043-11-5
boron nitride

| Conditions | Yield |
|---|---|
| In neat (no solvent) pyrolysis under pressure (100 MPa) at temperatures between 250°C and 700°C (N2), elem.anal.; | 60% |
| In neat (no solvent) High Pressure; borazine sealed under N2 in a gold capsule, pyrolyzed at 250-700°C, 25-100MPa, heating rate 10°C/min; | |
| In neat (no solvent) preparation of BN layers by induction heating of borazol; | |
| In neat (no solvent, gas phase) byproducts: H2; deposition on varius substrates; | |
| N2-carrying gas, chemical vapor deposition (graphite substrate, 1300-1800°C, 100-10000 Pa); detd. by IR spectroscopy; |

| Conditions | Yield |
|---|---|
| hydridocarbonyltris(triphenylphosphine)rhodium(I) In neat (no solvent) Kinetics; borazine and the anisole (ratio 10.5:1) reacted in the presence of a catalytic amt. Rh-complex at room temp. for 96.5 h; vacuum line fractionation of mixt. through a 0, -45 and -196°C trap series, product found in the 0°C trap; | 57% |

| Conditions | Yield |
|---|---|
| In diethylene glycol 3 h; | 39% |
| In diethyl ether 3 h; | 39% |
| In diethyl ether 3 h; | 39% |
| In diethylene glycol dimethyl ether 3 h; | 39% |
-
-
187737-37-7
propene

-
-
6569-51-3
borazine

-
A
-
110272-06-5
B-2-propenylborazine

-
B
-
110272-05-4
B-trans(1-propenyl)borazine

-
C
-
121232-06-2
B-propylborazine

| Conditions | Yield |
|---|---|
| palladium(II) bromide In neat (no solvent) stirring in vac., in a Fischer-Porter pressure reactor, room temp., 6 h; vac. line fractionation through a -65 °C trap, mixture of B-propyl- and B-propenylborazine, separation of the propenyl compds. by preparative GLC; | A n/a B n/a C 22% |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View