-
Name
1,3-Dichloroacetone
- EINECS 208-585-6
- CAS No. 534-07-6
- Article Data91
- CAS DataBase
- Density 1.383
- Solubility water: <0.1 mm Hg ( 20 °C)
- Melting Point 43 °C
- Formula C3H4Cl2O
- Boiling Point 173 °C at 760 mmHg
- Molecular Weight 126.97
- Flash Point 52.6 °C
- Transport Information UN 3286 3/PG 2
- Appearance white or colorless crystals
- Safety 26-36/37/39-45-28A
- Risk Codes 24-26/28-34-68-36/37/38
-
Molecular Structure
-
Hazard Symbols
 T+
T+
- Synonyms 1,3-Dichloro-2-propanone;Bis(chloromethyl) ketone;NSC 8745;sym-Dichloroacetone;a,a'-Dichloroacetone;a,g-Dichloroacetone;
- PSA 17.07000
- LogP 1.03310
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium chloride In water at 20 - 60℃; for 0.333333h; | 98% |
-
-
53535-68-5
bromo-3-chloro-propanone

-
-
1001-58-7
3-mercaptopropionitrile

-
B
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| A 94% B n/a |


| Conditions | Yield |
|---|---|
| With periodic acid In acetonitrile at 20℃; for 3h; Reagent/catalyst; Solvent; Temperature; | 80% |
| With chromic acid | |
| With chromium(III) oxide; sulfuric acid |

| Conditions | Yield |
|---|---|
| With methyllithium; lithium bromide In tetrahydrofuran; diethyl ether at -78℃; for 0.333333h; | 60% |
| Yield given. Multistep reaction; |
-
-
55549-51-4
2,4-Dichlor-3-aminocrotonsaeurenitril

-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 72h; | 58% |
-
-
82313-45-9
(Z)-1,3-Dichlor-2,4,4-trimorpholino-1,3-butadien-1-carbonitril

-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 60 - 70℃; for 10h; 2) Et2O, heating; | 15% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
75-44-5
phosgene

-
-
60-29-7
diethyl ether

-
-
534-07-6
1,3-Dichloroacetone


| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With hypochloric acid |

| Conditions | Yield |
|---|---|
| beim partielle Disproportionierung; |

| Conditions | Yield |
|---|---|
| With hypochloric acid |

| Conditions | Yield |
|---|---|
| With chlorine at -20℃; im Licht; |

| Conditions | Yield |
|---|---|
| With silver(I) chloride |
-
-
20485-53-4
1-chloro-3-diazopropan-2-one

-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| With hydrogenchloride In diethyl ether for 4h; Heating; Yield given; |

| Conditions | Yield |
|---|---|
| With chlorine; nickel dichloride |

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 45℃; bei der Chlorierung; | |
| With chlorine In tetrachloromethane Product distribution; 1.) below 40 deg C, 2.) room temperature, 0.5 h; | A 67 % Chromat. B 33 % Chromat. |
| With chlorine; acetic acid | |
| With hydrogenchloride; chlorine | |
| With antimonypentachloride; chlorine |

| Conditions | Yield |
|---|---|
| With disulfur dichloride beim Siedetemperatur; | |
| With disulfur dichloride |

| Conditions | Yield |
|---|---|
| In chlorobenzene Heating; |
-
-
463-49-0
1,2-propanediene

-
A
-
60367-21-7
dichloride of 1-chloro-2-propene-2-phosphonic acid

-
B
-
40632-73-3
dichloride of 2-chloro-2-propene-1-phosphonic acid

-
C
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With oxygen; phosphorus trichloride Product distribution; |


| Conditions | Yield |
|---|---|
| With ruthenium(IV) oxide; sodium chloride In water; ethyl acetate at 5℃; Product distribution; other organic solvents; | A 1 % Chromat. B 62 % Chromat. |
-
-
82598-72-9
1,3-dichloro-2,2-propanediol

-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With dehydration at 25℃; Rate constant; Equilibrium constant; Thermodynamic data; ΔH(excit.), ΔS(excit.), ΔG(excit.); isotope effects; | |
| With water In acetonitrile Equilibrium constant; various temperatures (15-46 deg C), isotope effect; |
-
-
110-63-4
Butane-1,4-diol

-
-
67-64-1
acetone

-
A
-
513-88-2
1,1-Dichloroacetone

-
C
-
77416-10-5
2,2-Bis(chloromethyl)-1,3-dioxacycloheptane

-
D
-
146774-72-3
2-dichloromethyl-2-methyl-1,3-dioxepane

-
E
-
78-95-5
chloroacetone

-
F
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With chlorine Product distribution; 1.) 20-50 deg C, 2.) room temperature, 0.5 h; | A n/a B n/a C 28.3 % Chromat. D 13.8 % Chromat. E n/a F n/a |

-
-
110-63-4
Butane-1,4-diol

-
-
67-64-1
acetone

-
A
-
513-88-2
1,1-Dichloroacetone

-
B
-
77416-10-5
2,2-Bis(chloromethyl)-1,3-dioxacycloheptane

-
C
-
146774-72-3
2-dichloromethyl-2-methyl-1,3-dioxepane

-
D
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With chlorine 1) 20-50 deg C; 2) room temp., 0.5 h; Further byproducts given. Yields of byproduct given; | A n/a B 28.3 % Chromat. C n/a D n/a |

-
-
67-64-1
acetone

-
A
-
513-88-2
1,1-Dichloroacetone

-
B
-
78-95-5
chloroacetone

-
C
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 23 - 25℃; electrochemical; Yield given. Yields of byproduct given. Title compound not separated from byproducts; | |
| With hydrogenchloride at 23 - 25℃; electrochemical; Yield given. Yields of byproduct given; | |
| With hydrogenchloride at 22 - 25℃; Product distribution; function of current and concentration; |


-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With sulfuric acid |

-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

-
-
534-07-6
1,3-Dichloroacetone
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
534-07-6
1,3-Dichloroacetone

-
-
87595-89-9
methyl 2-[2-chloro-1-(chloromethyl)ethylidene]-1-hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| In diethyl ether for 15h; Ambient temperature; | 100% |
| In methanol at 23℃; for 4h; | 54% |
| for 3h; Ambient temperature; Yield given; | |
| In methanol at 23℃; for 4h; |
-
-
534-07-6
1,3-Dichloroacetone

-
-
215599-97-6
(E)-1,3-dichloroacetone O-(3-phenyl-2-propenyl)oxime

| Conditions | Yield |
|---|---|
| In methanol at 40℃; for 2h; | 100% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
373602-92-7
bis-(2-morpholin-4-yl-thiazol-5-yl)-methanone

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-Dichloroacetone; 1-morpholino-3-(morpholinomethylene)thiourea In acetonitrile for 2h; Heating; Stage #2: With triethylamine In acetonitrile | 100% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
682353-67-9
1,3-diazido-propan-2-one

| Conditions | Yield |
|---|---|
| With sodium azide In acetone at 20℃; | 100% |
| With sodium azide In acetone at 20℃; for 12h; | 96% |
| With sodium azide In acetone at 20℃; for 12h; | 96% |
-
-
85102-68-7
2-N-piperidino-5-methyl-1,3-dithiolium-4-thiolate

-
-
534-07-6
1,3-Dichloroacetone

| Conditions | Yield |
|---|---|
| In acetone | 100% |

| Conditions | Yield |
|---|---|
| With sodium methylate In ethanol for 1.5h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 72h; | 100% |

| Conditions | Yield |
|---|---|
| In ethanol for 3h; Heating / reflux; | 100% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
196811-66-2
4-(tert-butoxycarbonyl)piperazine-1-thiocarboxamide

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In ethyl acetate; 1,2-dichloro-ethane | 100% |
-
-
6967-89-1
naphthalene-2-carbothioamide

-
-
534-07-6
1,3-Dichloroacetone

-
-
1094428-16-6
4-(chloromethyl)-2-(naphthalen-6-yl)thiazole

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 100% |
-
-
1424835-11-9
6-fluoronaphthalene-2-carbothioamide

-
-
534-07-6
1,3-Dichloroacetone

-
-
1424835-28-8
4-(chloromethyl)-2-(2-fluoronaphthalen-6-yl)thiazole

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 100% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
122-52-1
triethyl phosphite

-
-
81431-81-4
3-chloro-2-<(diethoxyphosphoryl)oxy>-1-propene

| Conditions | Yield |
|---|---|
| at 100℃; | 99% |
| In dichloromethane for 166h; Ambient temperature; | 98% |
| at 60 - 100℃; for 3h; | 98.9% |
| at 50℃; |
-
-
534-07-6
1,3-Dichloroacetone

-
-
35433-52-4
1,3-dichloroacetone oxime

| Conditions | Yield |
|---|---|
| With sulfuric acid; hydroxylamine hydrochloride In ethanol at 20℃; for 2h; Inert atmosphere; | 99% |
| With hydroxylamine |

| Conditions | Yield |
|---|---|
| In benzene for 48h; Heating; | 99% |
-
-
617-86-7
triethylsilane

-
-
534-07-6
1,3-Dichloroacetone

-
-
7531-61-5
((1,3-dichloropropan-2-yl)oxy)triethylsilane

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-Dichloroacetone With C8H10BiCl2N; C4H6AgN2(1+)*C24H12BCl8(1-) In dichloromethane-d2 at 25℃; Stage #2: triethylsilane In dichloromethane at 25℃; | 99% |
| With C24H36AlN2O2(1+)*C24H12BCl8(1-) In benzene-d6 for 15h; Inert atmosphere; Schlenk technique; Glovebox; | 98 %Spectr. |

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; Inert atmosphere; | 99% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
2227-79-4
benzenecarbothioamide

-
-
4771-31-7
2-phenyl-4-chloromethylthiazole

| Conditions | Yield |
|---|---|
| In ethanol for 4h; Reflux; | 98% |
| In ethanol for 2h; Reflux; | 90% |
| Stage #1: 1,3-Dichloroacetone; benzenecarbothioamide In acetone Inert atmosphere; Reflux; Stage #2: With sulfuric acid for 0.5h; Inert atmosphere; | 85% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
603-35-0
triphenylphosphine

-
-
13605-65-7
(3-chloro-2-oxopropyl)triphenylphosphonium chloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 24h; Reflux; | 98% |
| In benzene at 20℃; for 6h; | 98% |
| In tetrahydrofuran at 70℃; for 5h; | 97% |
-
-
2687-43-6
O-benzylhydoxylamine hydrochloride

-
-
534-07-6
1,3-Dichloroacetone

-
-
188125-86-2
1,3-dichloro-2-propanone O-benzyloxime

| Conditions | Yield |
|---|---|
| In ethanol Ambient temperature; | 98% |
| In ethanol at 20℃; for 24h; | 2.32 g |
-
-
62-55-5
thioacetamide

-
-
534-07-6
1,3-Dichloroacetone

-
-
39238-07-8
4-(chloromethyl)-2-methyl-1,3-thiazole

| Conditions | Yield |
|---|---|
| In acetone | 97% |
| at 20 - 50℃; for 20h; | 93% |
| 92.9% |
-
-
582-25-2
Potassium benzoate

-
-
534-07-6
1,3-Dichloroacetone

-
-
38982-27-3
1,3-Dibenzoyl-1,3-dihydroxyacetone

| Conditions | Yield |
|---|---|
| 5,11,17,23,29,35-Hexa-p-tert-butyl-37,38,39,40,41,42-hexakis-(3,6,9-trioxadecyloxy)calix<6>arene In water; acetonitrile at 65℃; for 6h; | 97% |
| With ethanol |
-
-
126-30-7
2,2-Dimethyl-1,3-propanediol

-
-
534-07-6
1,3-Dichloroacetone

-
-
133961-12-3
2,2-bis-(chloromethyl)-5,5-dimethyl-1,3-dioxane

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 97% |
| With toluene-4-sulfonic acid In benzene for 19h; Heating; | 97% |
| With toluene-4-sulfonic acid In benzene for 18h; Heating; | 97% |
-
-
72505-21-6
4-trifluoromethylbenzthioamide

-
-
534-07-6
1,3-Dichloroacetone

-
-
135873-35-7
4-chloromethyl-2-[4-(trifluoromethyl)phenyl]thiazole

| Conditions | Yield |
|---|---|
| In toluene Reflux; | 97% |
| In toluene at 80℃; for 4h; | 62% |
| With thionyl chloride; sodium hydrogencarbonate 1.) 1,2-dichloroethane; Multistep reaction; | |
| In 1,2-dichloro-ethane at 130 - 140℃; for 5h; Sealed tube; |
-
-
534-07-6
1,3-Dichloroacetone

-
-
603-35-0
triphenylphosphine

-
-
13605-66-8
1-chloro-3-(triphenylphosphoranylidene)acetone

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-Dichloroacetone; triphenylphosphine In tetrahydrofuran for 4h; Heating; Stage #2: With sodium carbonate In methanol at 20℃; for 0.5h; | 97% |
| Stage #1: 1,3-Dichloroacetone; triphenylphosphine In tetrahydrofuran for 24h; Heating; Stage #2: With sodium hydrogencarbonate In water Further stages.; | 92% |
| In tetrahydrofuran for 24h; Heating; | 91% |

| Conditions | Yield |
|---|---|
| In acetone at 20 - 25℃; for 72h; | 97% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
149-73-5
trimethyl orthoformate

-
-
6626-57-9
1,3-dichloroacetone dimethyl acetal

| Conditions | Yield |
|---|---|
| With Amberlyst 15 In dichloromethane | 96% |
-
-
534-07-6
1,3-Dichloroacetone

-
-
18435-67-1
o-methoxycarbonyl-α-diazoacetophenone

| Conditions | Yield |
|---|---|
| With copper acetylacetonate In benzene for 1h; Heating; | 96% |
-
-
16982-21-1
ethyl 2-amino-2-thioxoacetate

-
-
534-07-6
1,3-Dichloroacetone

-
-
100960-16-5
4-(chloromethyl)thiazole-2-carboxylic acid-ethyl ester

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 2h; | 96% |
| In ethanol Heating / reflux; | 37% |
| In ethanol for 15h; Heating; | |
| In toluene for 2h; Inert atmosphere; Reflux; | |
| In toluene Reflux; |
-
-
591-51-5
phenyllithium

-
-
534-07-6
1,3-Dichloroacetone

-
-
87234-28-4
1,3-dichloro-2-hydroxy-2-phenylpropane

| Conditions | Yield |
|---|---|
| Stage #1: phenyllithium With cerium(III) chloride In tetrahydrofuran; diethyl ether; cyclohexane at -78℃; for 0.666667h; Stage #2: 1,3-Dichloroacetone In tetrahydrofuran; diethyl ether; cyclohexane at -78℃; for 0.166667h; | 96% |
1,3-Dichloroacetone Chemical Properties
Bis(Chloromethyl)Ketone(534-07-6) is also named as 1,3-Dichloroacetone;1,3-dichloro-2-propanon;1,3-dichloroacetona;1,3-dichloro-propan-2-one;1,3-dichloropropanone;2-Propanone, 1,3-dichloro-;2-Propanone,1,3-dichloro-,and so on.Bis(Chloromethyl)Ketone(534-07-6) is usually a crystalline solid.
The Molecular formula of BIS(CHLOROMETHYL)KETONE(534-07-6) : C3H4Cl2O
The Molecular Weight of BIS(CHLOROMETHYL)KETONE(534-07-6) : 126.97
EINECS: 208-585-6
Molecular structure: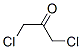
Melting point: 43 °C
Boiling point: 73 °C733.4 mm Hg(lit.)
Flash point: 193 °F
Density: 1.383 g/mL at 25 °C(lit.)
Vapor density: 4.38 (vs air)
Vapor pressure: <0.1 mm Hg ( 20 °C)
Refractive index: 1.4400
Storage temp.: Refrigerator (+4°C) + Poison room
Water Solubility: 27.9 g/L (20°C )
1,3-Dichloroacetone Uses
1,3-Dichloroacetone Toxicity Data With Reference
| 1. | mmo-sat 1250 ng/plate | MUREAV Mutation Research. 157 (1985),111. | ||
| 2. | mma-smc 5 µg/L | MUREAV Mutation Research. 155 (1985),53. | ||
| 3. | ihl-rat LC50:29 mg/m3/2H | 85GMAT Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure Izmerov, N.F., et al.,Moscow, USSR.: Centre of International Projects, GKNT,1982,44. | ||
| 4. | ihl-mus LC50:27 mg/m3/2H | 85GMAT Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure Izmerov, N.F., et al.,Moscow, USSR.: Centre of International Projects, GKNT,1982,44. |
1,3-Dichloroacetone Consensus Reports
1,3-Dichloroacetone Safety Profile
Poison by inhalation. Mutation data reported. A systemic irritant by ingestion and inhalation routes.
The Hazard Codes of BIS(CHLOROMETHYL)KETONE(534-07-6) :  T+
T+
HazardClass: 6.1
The Risk Statements information of BIS(CHLOROMETHYL)KETONE(534-07-6):
24: Toxic in contact with skin
34: Causes burns
68: Possible risk of irreversible effects
26/28: Very Toxic by inhalation and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of BIS(CHLOROMETHYL)KETONE(534-07-6) :
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
36/37/39: Wear suitable protective clothing, gloves and eye/face protection RIDADR: UN 3286 3/PG 2
WGK Germany: 3
F: 19
PackingGroup: II
HS Code: 29147090
1,3-Dichloroacetone Standards and Recommendations
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 534-08-7
- 53408-94-9
- 53408-96-1
- 53411-33-9
- 53411-70-4
- 53412-38-7
- 534-13-4
- 53413-67-5
- 53414-26-9
- 534-15-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View