-
Name
1-Phenyl-2-thiourea
- EINECS 203-151-2
- CAS No. 103-85-5
- Article Data200
- CAS DataBase
- Density 1.294 g/cm3
- Solubility Soluble in hot water and alcohol.
- Melting Point 145-150 °C(lit.)
- Formula C7H8N2S
- Boiling Point 266.7 °C at 760 mmHg
- Molecular Weight 152.22
- Flash Point 115.1 °C
- Transport Information UN 2811 6.1/PG 1
- Appearance white crystalline powder
- Safety 28-36/37-45
- Risk Codes 28-43
-
Molecular Structure
-
Hazard Symbols
 T+
T+
- Synonyms Thiourea,phenyl- (9CI);Urea, 1-phenyl-2-thio- (8CI);1-Phenyl-2-thiourea;1-Phenylthiourea;N-Phenylthiourea;NSC 5779;PTU;Phenylthiocarbamide;Phenylthiourea;U 6324;a-Phenylthiourea;
- PSA 70.14000
- LogP 2.11540
Synthetic route

| Conditions | Yield |
|---|---|
| With ammonia at -30℃; under 300.024 Torr; Addition; solid-gas reaction; | 100% |
| With ammonium hydroxide In water; acetonitrile at 20℃; for 3h; | 100% |
| With ammonia In 1,2-dimethoxyethane | 99% |

| Conditions | Yield |
|---|---|
| With hydrazine at 25℃; for 0.166667h; | 95% |
| With sodium hydroxide In water at 80℃; | 92.2% |
| With sodium hydroxide at 98℃; for 0.166667h; | 90% |

| Conditions | Yield |
|---|---|
| With ethanol for 0.0125h; Microwave irradiation; Neat (no solvent); | 94% |
| Heating; | 75% |
| With water at 100℃; Ausziehen den Salmiak durch kaltes Wasser und Umkrystallisieren das Umgeloeste aus siedendem Alkohol; |

| Conditions | Yield |
|---|---|
| Stage #1: aniline; 3-methyl-1-(methyldithiocarbonyl)imidazolium iodide In ethanol Substitution; Heating; Stage #2: With ammonia In ethanol for 3h; Substitution; | 94% |

| Conditions | Yield |
|---|---|
| With dibenzyl ether Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: ammonium thiocyanate With benzoyl chloride In acetone at 70℃; for 0.25h; Stage #2: aniline In acetone Heating; Stage #3: With sodium hydroxide at 90℃; for 0.333333h; | 82% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; trimethylbenzylammonium bromide; triethylamine In ethyl acetate for 2.5h; Heating; | 80% |
| With ethanol; ammonia at 100℃; | |
| Multi-step reaction with 2 steps 1: hydrogen chloride 2: alcohol; ammonia View Scheme |

-
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| With hydrogen sulfide In tetrahydrofuran at 90℃; for 4h; | 78% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 90℃; for 20h; | A 27% B 73% |

| Conditions | Yield |
|---|---|
| In acetone for 18h; Reflux; | 73% |

| Conditions | Yield |
|---|---|
| With ammonium thiocyanate In ethanol for 0.166667h; Heating; | 72% |

| Conditions | Yield |
|---|---|
| Stage #1: aniline; Benzoyl isothiocyanate In dichloromethane at 20℃; for 12h; Stage #2: With sodium hydroxide In tetrahydrofuran; water for 2h; Reflux; | 72% |

| Conditions | Yield |
|---|---|
| In toluene for 18h; Heating; | 71% |
-
-
74734-11-5
1H-imidazole-1-carbodithioic acid methyl ester

-
-
62-53-3
aniline

-
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| Stage #1: 1H-imidazole-1-carbodithioic acid methyl ester; aniline In ethanol Substitution; Heating; Stage #2: With ammonia In ethanol for 4.5h; Substitution; | 66% |
-
-
38012-72-5
trifluoroacetyl isothiocyanate

-
-
62-53-3
aniline

-
A
-
404-24-0
2,2,2-trifluoro-N-phenylacetamide

-
B
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | A 58.8% B 35% |
| In tetrahydrofuran |


| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; sodium ascorbate; sodium t-butanolate In dimethyl sulfoxide at 130℃; for 12h; | 55% |
-
-
1074-52-8
ammonium N-phenyldithiocarbamate

-
-
16616-43-6
1-(4-methoxy-phenyl)-3-phenyl-propynone

-
A
-
53246-45-0
(Z,Z)-3,3'-Thiodi(1-p-methoxyphenyl-3-phenyl-prop-2-ene-1-one)

-
B
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| In ethanol Ambient temperature; | A 50% B 20% |

-
-
43009-16-1
phenyldithiocarbamic acid triethylamine salt

-
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| Stage #1: phenyldithiocarbamic acid triethylamine salt With ammonia In water; acetonitrile at 0℃; for 0.5h; Stage #2: With C13H16N2(2+)*2Br3(1-) In water; acetonitrile at 0 - 20℃; for 6h; | 42% |
| With [bis(acetoxy)iodo]benzene; ammonia In water; acetonitrile for 0.5h; Cooling with ice; | |
| With 1,1'-(ethane-1,2-diyl)dipyridinium bistribromide; ammonia In water; acetonitrile at 20℃; Cooling with ice; |
-
-
16616-42-5
p-chlorobenzoylphenylacetylene

-
-
1074-52-8
ammonium N-phenyldithiocarbamate

-
A
-
53324-04-2
(E,Z)-3,3'-Thiodi(1-p-chlorophenyl-3-phenylprop-2-en-1-one)

-
B
-
53324-02-0
(Z,Z)-3,3'-Thiodi(1-p-chlorophenyl-3-phenyl-prop-2-ene-1-one)

-
C
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| In ethanol Ambient temperature; | A 20% B 30% C 15% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In water Reflux; | 30% |
| Stage #1: aniline With hydrogenchloride In water Heating; Stage #2: ammonium thiocyanate In water Reflux; |
-
-
16325-43-2
1-phenyl-4,4,6-trimethyl-1,4-dihydropyrimidine-2(3H)thione

-
A
-
58913-46-5
6,6-dimethyl-4-(phenylamino)-5,6-dihydropyridine-2(1H)-thione

-
B
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| at 140 - 160℃; for 3.5h; | A 12% B 11% |

| Conditions | Yield |
|---|---|
| With chlorobenzene |
-
-
64-17-5
ethanol

-
-
43180-42-3
1-[(E)-benzylideneamino]-2,2,2-trichloroethanol

-
-
103-72-0
phenyl isothiocyanate

-
A
-
100-52-7
benzaldehyde

-
B
-
75-87-6
chloral

-
C
-
103-85-5
monophenylthiourea


| Conditions | Yield |
|---|---|
| at 160 - 175℃; | |
| at 160 - 175℃; beim Behandeln von Anilin-hydrochlorid; | |
| at 160 - 165℃; |

-
-
64-17-5
ethanol

-
-
1594-56-5
2,4-dinitro-phenyl thiocyanate

-
-
62-53-3
aniline

-
A
-
2217-55-2
bis(2,4-dinitrophenyl) disulphide

-
B
-
961-68-2
N-phenyl-2,4-dinitroaniline

-
C
-
2253-67-0
bis-(2,4-dinitro-phenyl)-sulfide

-
D
-
103-85-5
monophenylthiourea


| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| at 40℃; |

| Conditions | Yield |
|---|---|
| at 150℃; |

-
-
99-81-0
4-Nitrophenacyl bromide

-
-
103-85-5
monophenylthiourea

-
-
102183-91-5
4-(4-nitrophenyl)-N-phenyl-1,3-thiazol-2-amine

| Conditions | Yield |
|---|---|
| With sodium hydrogen sulfate In water at 20℃; for 0.0833333h; Hantzsch reaction; regioselective reaction; | 100% |
| With (2-hydroxyethyl)trimethylazanium urea chloride In water at 20℃; for 0.333333h; Reagent/catalyst; | 97% |
| With sodium fluoride In methanol; water at 20℃; | 97% |

| Conditions | Yield |
|---|---|
| at 70℃; for 1h; | 100% |

-
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| In acetone for 0.5h; Heating; | 100% |

-
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| In acetone for 0.5h; Heating; | 100% |
-
-
2227-64-7
3'-nitro-2-bromoacetophenone

-
-
103-85-5
monophenylthiourea

-
-
325850-28-0
[4-(3-nitrophenyl)thiazol-2-yl]phenylamine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 2h; Hantzsch Thiazole Synthesis; | 100% |
| In ethanol at 80℃; | 100% |
| In water at 20℃; for 1.5h; | 90% |
| In ethanol at 20℃; for 2h; Hantzsch Thiazole Synthesis; | 5.22 g |
-
-
1415820-00-6
methyl 4-(2-chloroacetyl)-1H-pyrrole-2-carboxylate

-
-
103-85-5
monophenylthiourea

-
-
1415820-07-3
methyl 4-(2-(phenylamino)thiazol-4-yl)-1H-pyrrole-2-carboxylate

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With quinolinium monofluorochromate(VI) In acetonitrile for 1.5h; Heating; | 99% |
| With bismuth(III) nitrate In acetonitrile for 0.333333h; Heating; | 98% |
| With 3-carboxypyridinium chlorochromate In acetonitrile for 0.0333333h; Product distribution; Further Variations:; Reagents; Temperatures; without microwave irradiation; solvent-free; microwave irradiation; | 98% |
-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
103-85-5
monophenylthiourea

-
-
59819-37-3
1-<(Dimethylamino)methylene>-3-phenyl-2-thiourea

| Conditions | Yield |
|---|---|
| at 100℃; for 1h; | 99% |
| In dichloromethane | 94% |
| In methanol at 20℃; | 79% |
| In ethanol for 1.5h; Reflux; | 72% |
| (heating); |
-
-
105533-63-9
2,5-dihydroxy-3,4,6,7-tetrachloro-2,3-dihydrobenzofuran

-
-
103-85-5
monophenylthiourea

-
-
98326-98-8
2,3,5-Trichloro-6-(2-phenylamino-thiazol-5-yl)-benzene-1,4-diol

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Heating; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol at 50℃; for 0.0833333h; microwave irradiation; | 98% |
| With sodium fluoride In methanol; water at 20℃; | 98% |
| In water at 20℃; for 1h; | 96% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium hydroxide In dimethyl sulfoxide at 90℃; for 2h; | 98% |
| With polystyrne-bound selenoxide In methanol for 24h; Heating; | 85% |
| With KO2 In pyridine at 60℃; | 82% |
-
-
29310-88-1
3-bromoacetylcoumarin

-
-
103-85-5
monophenylthiourea

-
-
33856-02-9
3‑(2‑(phenylamino)thiazol‑4‑yl)‑2H‑chromen‑2‑one

| Conditions | Yield |
|---|---|
| With sodium fluoride In methanol; water at 20℃; | 98% |
| In water at 25℃; for 0.0125h; Sonication; Green chemistry; | 94% |
| at 20℃; for 0.25h; Neat (no solvent); grinding; | 85% |
-
-
619-41-0
4-(bromoacetyl)toluene

-
-
103-85-5
monophenylthiourea

-
-
93020-56-5
2-phenylamino-4(4'-methylphenyl)thiazole

| Conditions | Yield |
|---|---|
| With sodium fluoride In methanol; water at 20℃; | 98% |
| With Nafion-H In water; ethylene glycol at 50℃; for 0.166667h; | 97% |
| In water at 25℃; for 0.00555556h; Sonication; Green chemistry; | 91% |
-
-
99-73-0
para-bromophenacyl bromide

-
-
103-85-5
monophenylthiourea

-
-
42056-77-9
4-(4-bromophenyl)-N-phenyl-1,3-thiazol-2-amine

| Conditions | Yield |
|---|---|
| With sodium fluoride In methanol; water at 20℃; | 98% |
| With (2-hydroxyethyl)trimethylazanium urea chloride In water at 20℃; for 0.333333h; Reagent/catalyst; | 93% |
| In water at 25℃; for 0.00833333h; Sonication; Green chemistry; | 93% |
-
-
51605-33-5
4-methyl-5-chloromethyl-imidazole hydrochloride

-
-
103-85-5
monophenylthiourea

-
-
118796-95-5
1-phenyl-2-(4-methyl-5-imidazolylmethyl)isothiourea dihydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 0.0833333h; Ambient temperature; | 98% |
-
-
50-00-0
formaldehyd

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
103-85-5
monophenylthiourea

-
-
701277-33-0
1-phenyl-5-(2-trifluoromethyl-phenyl)-[1,3,5]triazinane-2-thione

| Conditions | Yield |
|---|---|
| In water for 0.0138889h; microwave irradiation; | 98% |

-
-
2114-00-3
2-bromo-1-phenyl-1-propanone

-
-
103-85-5
monophenylthiourea

-
-
93020-55-4
5-methyl-4-phenyl-2-(N-phenylamino)thiazole

| Conditions | Yield |
|---|---|
| In ethylene glycol at 20℃; for 0.416667h; | 98% |
| In ethanol at 50℃; for 0.0833333h; microwave irradiation; | 97% |
| In N,N-dimethyl-formamide at 80℃; for 0.0833333h; | 40% |
-
-
1484-50-0
desyl bromide

-
-
103-85-5
monophenylthiourea

-
-
29594-65-8
2-phenylamino-4,5-diphenyl-1,3-thiazole

| Conditions | Yield |
|---|---|
| In ethanol at 50℃; for 0.0833333h; microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; | 98% |
| In water at 20℃; for 2h; | 90% |
-
-
2632-13-5
2-Bromo-4'-methoxyacetophenone

-
-
103-85-5
monophenylthiourea

-
-
21344-98-9
4-(4-methoxyphenyl)-N-phenyl-1,3-thiazol-2-amine

| Conditions | Yield |
|---|---|
| In ethylene glycol at 20℃; for 0.0833333h; | 98% |
| In water at 20℃; for 1h; | 96% |
| With sodium hydrogen sulfate In water at 20℃; for 0.916667h; Hantzsch reaction; regioselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With sodium fluoride In methanol; water at 20℃; | 98% |
| In ethylene glycol at 20℃; for 0.166667h; | 96% |
| In water at 25℃; for 0.00694444h; Sonication; Green chemistry; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In PEG 400 at 80℃; for 2h; | 98% |
-
-
1395225-93-0
2-chloro-1-(6-phenylimidazo[2,1-b]thiazol-5-yl)ethanone

-
-
103-85-5
monophenylthiourea

-
-
1395225-77-0
N-phenyl-4-(6-phenylimidazo[2,1-b]thiazol-5-yl)thiazol-2-amine

| Conditions | Yield |
|---|---|
| In methanol at 90℃; under 12929 Torr; for 0.5h; Temperature; Time; Solvent; Hantzsch Thiazole Synthesis; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 20℃; for 0.416667h; Solvent; Reagent/catalyst; Temperature; Time; Milling; | 98% |
-
-
137104-38-2
methyl (2Z)-2-(bromomethyl)pent-2-enoate

-
-
103-85-5
monophenylthiourea

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; for 1h; | 98% |
-
-
103-85-5
monophenylthiourea

-
-
4115-26-8, 68743-71-5
5-imino-4-phenyl-3-phenylamino-4,5-dihydro-1,2,4-thiadiazoline

| Conditions | Yield |
|---|---|
| With polystyrene-bound diaryl telluroxide In acetic acid for 12h; Ambient temperature; | 97% |
| With benzenetellurinyl trifluoromethanesulfonate In chloroform at 50℃; for 3h; | 96% |
| With hydrogenchloride; dimethyl sulfoxide In water; acetone at 40℃; | 90% |
-
-
141-97-9
ethyl acetoacetate

-
-
100-52-7
benzaldehyde

-
-
103-85-5
monophenylthiourea

-
-
108958-75-4
ethyl 6-methyl-1,4-diphenyl-2-thioxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In N,N-dimethyl-formamide at 20℃; for 48h; | 97% |
| Stage #1: ethyl acetoacetate; benzaldehyde; monophenylthiourea In N,N-dimethyl-formamide for 1h; Biginelli Pyrimidone Synthesis; Sonication; Stage #2: With chloro-trimethyl-silane In N,N-dimethyl-formamide at 20℃; for 72h; Biginelli Pyrimidone Synthesis; | 97% |
| acid medium; | 70% |

-
-
101-02-0
triphenyl phosphite

-
-
7401-37-8
benzylsulfanylacetaldehyde

-
-
103-85-5
monophenylthiourea

-
-
79428-40-3
[2-Benzylsulfanyl-1-(3-phenyl-thioureido)-ethyl]-phosphonic acid diphenyl ester

| Conditions | Yield |
|---|---|
| In acetic acid 1.) 0.5h, room temp., 2.) 0.5h, 80 deg C; | 97% |


| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In N,N-dimethyl-formamide at 20℃; for 48h; | 97% |

1-Phenyl-2-thiourea Chemical Properties
Product Name: 1-Phenyl-2-thiourea (103-85-5)
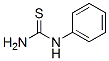
Molecular Formula: C7H8N2S
Molecular Weight: 152.22g/mol
Mol File: 103-85-5.mol
Appearance: white crystalline powder
Melting Point: 145-150 °C(lit.)
Boiling point: 266.7 °C at 760 mmHg
Storage Temperature: Poison room
Flash Point: 115.1 °C
Density: 1.294 g/cm3
Water Solubility: negligible
Stability: Stable. Incompatible with strong acids, strong oxidizing agents, strong bases.
Product Categories: Benzene derivatives
Synonyms of 1-Phenyl-2-thiourea (103-85-5): 1-phenyl-2-thio-ure ; alpha-Phenylthiourea ; Fenylthiomocovina ; NCI-C02017 ; phenyl-thioure ; phenylthiourea0 ; Rcra waste number P093 ; rcrawastenumberp093 .
1-Phenyl-2-thiourea Uses
1-Phenyl-2-thiourea (103-85-5) is used in the manufacture of rodenticides and in medical genetics.It's for the production of 2 - amino-benzothiazole, 3 - methyl-benzothiazole hydrazone such as dye intermediates. Also it's used as analytical reagent.
1-Phenyl-2-thiourea Production
From the first with sulfuric acid aniline salt, and then carry out addition reaction of sodium cyanide with sulfur derived.
1-Phenyl-2-thiourea Toxicity Data With Reference
1-Phenyl-2-thiourea (103-85-5) is classified as extremely toxic. The probable oral lethal dose is 5-50 mg/kg or between 7 drops and 1 teaspoon for a 70 kg (150 lb.) person.
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 25mg/kg (25mg/kg) | National Technical Information Service. Vol. AD277-689, | |
| mouse | LD50 | oral | 10mg/kg (10mg/kg) | Progress Report for Contract No. NIH-NCI-E-C-72-3252, Submitted to the National Cancer Institute by Litton Bionetics, Inc. Vol. NCI-E-C-72-3252, Pg. 1973, | |
| rabbit | LD50 | oral | 40mg/kg (40mg/kg) | Journal of Medicinal and Pharmaceutical Chemistry. Vol. 4, Pg. 109, 1961. | |
| rat | LD50 | intraperitoneal | 5mg/kg (5mg/kg) | ENDOCRINE: HYPERGLYCEMIA | Journal of Pharmacology and Experimental Therapeutics. Vol. 89, Pg. 186, 1947. |
| rat | LD50 | oral | 3mg/kg (3mg/kg) | Journal of Medicinal and Pharmaceutical Chemistry. Vol. 4, Pg. 109, 1961. |
1-Phenyl-2-thiourea Consensus Reports
NCI Carcinogenesis Bioassay (feed); No Evidence: mouse, rat NCITR* National Cancer Institute Carcinogenesis Technical Report Series. (Bethesda, MD 20014) No. NCI-CG-TR-148 ,1978. . EPA Extremely Hazardous Substances List. Reported in EPA TSCA Inventory.
1-Phenyl-2-thiourea Safety Profile
Dangerous disaster hazard; emits toxic fumes of oxides of sulfur and nitrogen when heated to decomposition. Avoid acids or acid fumes.
Safety Information of 1-Phenyl-2-thiourea (103-85-5):
Hazard Codes: T+![]()
Risk Statements: 28-43
28: Very Toxic if swallowed
43: May cause sensitization by skin contact
Safety Statements: 28-36/37-45
28: After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
36: Wear suitable protective clothing
37: Wear suitable gloves
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
Related Products
- 1-Phenyl-2-thiourea
- 103857-96-1
- 103858-53-3
- 103859-86-5
- 103861-77-4
- 103862-55-1
- 103862-63-1
- 10386-27-3
- 10386-28-4
- 103863-14-5
- 103863-15-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View