Jiangsu Huaen Energy Co., Ltd.
Geographical advantages: the processing base is located in Jiangsu Coastal Chemical Industrial Park with convenient transportation, good human, economic and social environment, and huge potential for market development; Technology Advantages: The
Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:88-17-5
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:88-17-5
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:Colorless to light yellow transparent liquid Storage:Store be
Cas:88-17-5
Min.Order:250 Kilogram
FOB Price: $9.0 / 16.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Appearance:White or off-white Solid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:88-17-5
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Cas:88-17-5
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
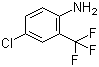
Product Name: 2-Aminobenzotrifluoride Synonyms: α,α,α-Trifluoro-o-toluidine, 2-Aminobenzotrifluoride;2-Aminobenzotrifluoride ,99%;o-Amino-α,α,α-trifluorotoluene;2-(Trifluoromethyl)aniline,α,&al
Cas:88-17-5
Min.Order:1 Gram
FOB Price: $8900.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Product name 2-Aminotrifluoromethylbenzene Synonyms 2-Aminobenzotrifluor CAS No. 88-17-5 Molecular weight 161.13 Molecular formula C7H6F3N
Cas:88-17-5
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:88-17-5
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:88-17-5
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
2-AminotrifluoromethylbenzeneAppearance:Colorless transparent liquid Storage:Keep away of light, cool place Package:according to customers' requirements Application:pharmaceutical intermediates Transportation:By air(EMS or EUB or FedEx or TNT ect...)
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediat
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Cas:88-17-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBeijing Greenchem Technology Co.,Ltd. ( Panjin Greenchem Technology Co.Ltd .)
Capability on chemical synthesis1. Beijing High-Tech Enterprises2. Strong R&D Team3. 8 years of experiences in R & D of high-tech Catalyst;4. 5000 production techniques, 69 items of national patents, and 360 kinds of products on sales;5. The producti
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
DB BIOTECH CO., LTD
Q1: Are you a manufacturer Answer: Yes, we are factory founded on 2012.Q2: How to contact with us Click "contact supplier" And then send us message the product you interest in, you will get reply within 12 hours.Q3:Which kind of payment terms do you
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:88-17-5
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hangzhou Ocean Chemical Co., Ltd.
★.Best quality according to requirement ★.Competitive price in China market ★.Mature Technical support ★.Professional logistic support
Cas:88-17-5
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hubei Langyou International Trading Co., Ltd
High purity 2-Aminobenzotrifluoride CAS 88-17-5 with factory price Application:High purity 2-Aminobenzotrifluoride CAS 88-17-5 with factory price
Cas:88-17-5
Min.Order:0
Negotiable
Type:Other
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With thionyl chloride In methanol at 75℃; for 3h; | 94% |

| Conditions | Yield |
|---|---|
| With 1-acetoxy-1,2-benziodoxol-3-one; potassium acetate In acetonitrile at 80℃; for 13h; Reagent/catalyst; Solvent; Temperature; Time; | 89% |

| Conditions | Yield |
|---|---|
| With 1,3-dimethyl-2-imidazolidinone; sodium trimethylsilanethiolate at 185℃; for 24h; | 87% |
| With sodium hydrogensulfide; sodium hydrogencarbonate In methanol; water for 0.5h; Heating; | 68% |
| In water at 31 - 35℃; for 48h; reduction with baker's yeast (Saccharomyces cerevisiae); | 66% |

-
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol | 85% |
| With hydrogenchloride In ethanol at 100℃; for 12h; | 1.13 g |
| With hydrogenchloride In ethanol Reflux; |
-
-
81290-20-2
(trifluoromethyl)trimethylsilane

-
-
615-36-1
2-bromoaniline

-
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; dicyclohexyl-(2′,4′,6′-triisopropyl-3,6-dimethoxy-[1,1′-biphenyl]-2-yl)phosphine; sodium dodecyl-sulfate; cesium fluoride In toluene at 110℃; for 12h; Inert atmosphere; | 70% |
-
-
887144-94-7
Togni's reagent II

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 75℃; for 6h; Schlenk technique; Inert atmosphere; | A 20% B 66% |
| With potassium carbonate; nickel(II) hydroxide In dimethyl sulfoxide at 35℃; for 2h; Reagent/catalyst; Solvent; Temperature; Overall yield = 89 %; | A 20% B 61% |
| With potassium carbonate; nickel(II) hydroxide In dimethyl sulfoxide at 35℃; for 2h; Reagent/catalyst; Solvent; Inert atmosphere; | A 20% B 49% |

-
-
2314-97-8
iodotrifluoromethane

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With fac-tris(2-phenylpyridinato-N,C2')iridium(III); potassium carbonate In 1,2-dichloro-ethane at 20℃; for 24h; Inert atmosphere; Schlenk technique; Irradiation; regioselective reaction; | A 34% B 54% |
| With sulfuric acid; dihydrogen peroxide; iron(II) sulfate; dimethyl sulfoxide In water at 40 - 50℃; for 0.333333h; | A 7.2 %Spectr. B 8.5 %Spectr. |
| With dihydrogen peroxide; iron(II) sulfate In water; dimethyl sulfoxide | A 13 %Spectr. B 22 %Spectr. |

-
-
1548-68-1
1-azido-2-(trifluoromethyl)benzene

-
A
-
90701-87-4
2-methoxy-3-trifluoromethyl-3H-azepine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; methanol for 15h; Irradiation; | A 49% B 5% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 30℃; for 3h; Inert atmosphere; | A 23% B 44% |

-
-
98-56-6
4-chlorobenzotrifluoride

-
-
62-53-3
aniline

-
A
-
98-16-8
3-trifluoromethylaniline

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With 2,3,7,8-tetrafluoro-S-(trifluoromethyl)dibenzothiophenium trifluoromethanesulfonate In N,N-dimethyl-formamide at 30℃; for 3h; Reagent/catalyst; Inert atmosphere; | A 23% B 44% |

-
-
129922-39-0
3,7-dinitro-Se-(trifluoromethyl)dibenzoselenophenium triflate

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 0.5h; Ambient temperature; Title compound not separated from byproducts; | A 16 % Spectr. B 39% |
| In N,N-dimethyl-formamide for 0.5h; Ambient temperature; Title compound not separated from byproducts; | A 16 % Spectr. B 39 % Spectr. |

-
-
129946-88-9
Umemoto's reagent

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 80℃; for 5h; Title compound not separated from byproducts; | A 15% B 37% |
| In N,N-dimethyl-formamide at 80℃; for 7h; | A 18% B 39 % Spectr. |
| In N,N-dimethyl-formamide at 30℃; for 3h; Inert atmosphere; | A 2% B 5% |

-
-
62-53-3
aniline

-
-
160656-66-6
S-(Trifluoromethyl)-7-nitrodibenzothiophenium-3-sulfonate

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 80℃; for 0.5h; | A 18 % Spectr. B 37% |
| In N,N-dimethyl-formamide at 80℃; for 0.5h; | A 18% B 37 % Spectr. |
| In N,N-dimethyl-formamide at 80℃; for 0.5h; Product distribution; other temperature; |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 30℃; for 3h; Inert atmosphere; | A 17% B 37% |

-
-
75-63-8
Bromotrifluoromethane

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With α-picoline; sodium metabisulfite; sulfur dioxide; zinc In N,N-dimethyl-formamide at 20℃; under 2250.2 - 3750.3 Torr; for 2h; Product distribution; other alkylating reagents, other method, other pressure, temp.; various substrates; | A 20% B 36% |
| With α-picoline; sodium metabisulfite; sulfur dioxide; zinc In N,N-dimethyl-formamide at 20℃; under 2250.2 - 3750.3 Torr; for 2h; also with NaS2O4; | A 20% B 36% |
| With sulfur dioxide; zinc In N,N-dimethyl-formamide under 2250.2 - 3750.3 Torr; for 2h; Ambient temperature; | A 20% B 36% |

-
-
75-63-8
Bromotrifluoromethane

-
-
95-51-2
2-Chloroaniline

-
A
-
433-94-3
2-chloro-6-(trifluoromethyl)benzenamine

-
B
-
39885-50-2
2-chloro-4-(trifluoromethyl)aniline

-
C
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With α-picoline; sodium metabisulfite; sulfur dioxide; zinc In N,N-dimethyl-formamide at 50℃; under 2250.2 - 3750.3 Torr; for 4h; | A 21% B 21% C 14% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 30℃; for 3h; Inert atmosphere; | A 6% B 14% |

-
-
384-22-5
2-nitrobenzotrifluoride

-
A
-
393-39-5
4-fluoro-2-(trifluoromethyl)aniline

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With bismuth; hydrogen fluoride In dichloromethane Yields of byproduct given; | A 12% B n/a |

| Conditions | Yield |
|---|---|
| With sodium hypobromide |

| Conditions | Yield |
|---|---|
| With steam; hydrazine hydrate |
-
-
77811-21-3
2-(2'-trifluoromethylphenyl)azo-4-phenyl-1,3,4-thiadiazol-5-one

-
A
-
645-48-7
1-phenyl-3-thiosemicarbazide

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With sodium hydroxide; zinc In methanol Heating; |
-
-
129922-33-4
Se-(trifluoromethyl)dibenzoselenophenium triflate

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 20h; Ambient temperature; | A 4 % Spectr. B 7 % Spectr. |
| In N,N-dimethyl-formamide for 20h; Ambient temperature; Title compound not separated from byproducts; | A 4 % Spectr. B 7 % Spectr. |

-
-
129922-35-6
3-nitro-S-(trifluoromethyl)dibenzothiophenium triflate

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 0.5h; Ambient temperature; Title compound not separated from byproducts; | A 11 % Spectr. B 18 % Spectr. |

-
-
129922-37-8
S-(trifluoromethyl)-3,7-dinitrodibenzothiophenium trifluoromethanesulfonate

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 2h; Ambient temperature; Title compound not separated from byproducts; | A 26 % Spectr. B 57 % Spectr. |

-
-
55642-42-7
bis(trifluoromethyl)tellurium

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
98-16-8
3-trifluoromethylaniline

-
C
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 140℃; for 60h; Title compound not separated from byproducts; | A 24 % Spectr. B 21 % Spectr. C 21 % Spectr. |

-
-
62-53-3
aniline

-
-
160656-62-2
5-(trifluoromethyl)-5H-dibenzo[b,d]thiophenium-3-sulfonate

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 80℃; for 7h; | A 18 % Spectr. B 36 % Spectr. |
| In N,N-dimethyl-formamide at 80℃; for 7h; Product distribution; other time; |

-
-
129946-88-9
Umemoto's reagent

-
-
62-53-3
aniline

-
A
-
132-65-0
dibenzothiophene

-
B
-
455-14-1
4-trifluoromethylphenylamine

-
C
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In d7-N,N-dimethylformamide at 62℃; Kinetics; Thermodynamic data; other temperatures and solvents; ΔH(excit.), ΔS(excit.), ΔG(excit.); |

-
-
129922-37-8
S-(trifluoromethyl)-3,7-dinitrodibenzothiophenium trifluoromethanesulfonate

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
59005-75-3
3,7-dinitro-dibenzothiophene

-
C
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In d7-N,N-dimethylformamide at -10℃; Kinetics; Thermodynamic data; other temperatures and solvents; ΔH(excit.), ΔS(excit.), ΔG(excit.); |

-
-
62-53-3
aniline

-
A
-
455-14-1
4-trifluoromethylphenylamine

-
B
-
139-66-2
diphenyl sulfide

-
C
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In d7-N,N-dimethylformamide at 81.4℃; Kinetics; Thermodynamic data; other temperatures and solvents; ΔH(excit.), ΔS(excit.), ΔG(excit.); |

-
-
13668-61-6
(cyclopent-2-eneyl)acetic acid

-
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| With dmap; N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane | 100% |
-
-
463-71-8
thiophosgene

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
1743-86-8
2-(trifluoromethyl)phenyl isothiocyanate

| Conditions | Yield |
|---|---|
| In water at 20℃; for 12h; | 100% |
| With sodium hydrogencarbonate In water at 0 - 20℃; for 24h; Inert atmosphere; | 95% |
| With triethylamine In dichloromethane at 20℃; for 1h; |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With 2C12H25O3S(1-)*Zn(2+); (meso)-1,1'-([2,2'-bipyridine]-6,6'-diyl)bis(2,2-dimethylpropan-1-ol) In water at 20℃; for 22h; enantioselective reaction; | 100% |
-
-
76-02-8
Trichloroacetyl chloride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
349416-85-9
2,2,2-trichloro-N-(2-(trifluoromethyl)phenyl)acetamide

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 0.5h; Cooling with ice; | 100% |
| With potassium carbonate In dichloromethane at 20℃; Inert atmosphere; | 93% |
-
-
1429217-23-1
(R)-N-(1-(3-bromophenyl)ethyl)-3-methoxy-1-methyl-2-oxo-1,2-dihydropyridine-4-carboxamide

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
1429217-51-5
(R)-3-methoxy-1-methyl-2-oxo-N-(1-(3-(2-(trifluoromethyl)phenylamino)phenyl)ethyl)-1,2-dihydropyridine-4-carboxamide

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In toluene at 110℃; Inert atmosphere; | 100% |
-
-
3282-30-2
pivaloyl chloride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
125686-52-4
N-<2-(trifluromethyl)phenyl>-2,2-dimethylpropanamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; for 16h; | 100% |
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 12h; Inert atmosphere; | |
| With sodium hydrogencarbonate In dichloromethane at 20℃; |
-
-
15486-96-1
3-Bromopropionyl chloride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
3945-39-9
3-bromo-propionic acid-(2-trifluoromethyl-anilide)

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane at 20℃; for 0.5h; Inert atmosphere; | 100% |
| With potassium carbonate |
-
-
532-55-8
Benzoyl isothiocyanate

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
63494-72-4
N-((2-(trifluoromethyl)phenyl)carbamothioyl)benzamide

| Conditions | Yield |
|---|---|
| In acetone for 1.5h; Reflux; | 99% |
| In benzene | 86% |
| In tetrahydrofuran at 60 - 65℃; for 0.166667h; Microwave irradiation; | 80% |
-
-
1079-66-9
chloro-diphenylphosphine

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
648930-41-0
C6H4(o-CF3)NP(C6H5)2P(C6H5)2

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 0.5h; | 99% |

| Conditions | Yield |
|---|---|
| In water for 0.0416667h; microwave irradiation; | 99% |
| In water at 20℃; for 0.416667h; Ionic liquid; Green chemistry; | 90% |
-
-
39973-76-7
ethyl 2-benzoyl-3-ethoxyacrylate

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
854777-97-2
2-benzoyl-3-(2-trifluoromethylphenylamino)acrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| In toluene at 150℃; for 1h; Microwave irradiation; | 99% |
| In toluene for 18h; Heating; | 82% |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; N-Bromosuccinimide; 1-butyl-3-methylimidazolium trifluoromethanesulfonimide In toluene at 40℃; regioselective reaction; | 99% |
| With N-Bromosuccinimide In N,N-dimethyl-formamide at 0 - 20℃; for 2h; Inert atmosphere; Schlenk technique; | 93% |
| With 1-hexyl-3-methyl-1-imidazolium bromide; copper(ll) bromide for 1h; regioselective reaction; | 92% |

| Conditions | Yield |
|---|---|
| With tetraethoxy orthosilicate; sulfuric acid at 160℃; for 16h; | 98% |
-
-
1121-60-4
pyridine-2-carbaldehyde

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
386713-04-8
N-(2-pyridylmethylidene)-2-trifluoromethylaniline

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In benzene at 80℃; for 8h; | 98% |
| In toluene for 24h; Reflux; | 94% |
| With toluene-4-sulfonic acid In toluene at 70℃; for 48h; Molecular sieve; | 91.9% |

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In benzene at 80℃; for 8h; | 98% |
-
-
50-00-0
formaldehyd

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
103-85-5
monophenylthiourea

-
-
701277-33-0
1-phenyl-5-(2-trifluoromethyl-phenyl)-[1,3,5]triazinane-2-thione

| Conditions | Yield |
|---|---|
| In water for 0.0138889h; microwave irradiation; | 98% |

-
-
98946-18-0
tert-Butyl 2,2,2-trichloroacetimidate

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
864365-12-8
tert-butyl-(2-trifluoromethyl-phenyl)-amine

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In cyclohexane at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; benzoic acid In toluene at 110℃; for 5h; | 98% |
| With 1,1'-bis-(diphenylphosphino)ferrocene; palladium diacetate; benzoic acid In toluene at 110℃; for 1h; Inert atmosphere; chemoselective reaction; | 98% |
-
-
76-05-1
trifluoroacetic acid

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
2368-64-1
2,2,2-trifluoro-N-(2-(trifluoromethyl)phenyl)acetamide

| Conditions | Yield |
|---|---|
| With pyridine; dmap In dichloromethane at 0 - 20℃; for 15.5h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With diethylzinc In hexane; toluene at 50℃; for 2h; Schlenk technique; Inert atmosphere; | 98% |
-
-
75-87-6
chloral

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
444-93-9
(E)-2-(hydroxyimino)-N-(2-(trifluoromethyl)phenyl)acetamide

| Conditions | Yield |
|---|---|
| With hydroxyammonium sulfate; sodium sulfate In ethanol at 60 - 80℃; for 6h; | 97% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetone at 90℃; for 3h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-(trifluoromethyl)benzenamine With hydrogenchloride; sodium nitrite In water at 0 - 5℃; for 0.5h; Stage #2: acetylacetone With sodium acetate In ethanol; water at 0 - 20℃; for 4h; | 97% |
-
-
98-09-9
benzenesulfonyl chloride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
86802-68-8
N-(2-(trifluoromethyl)phenyl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With pyridine In acetonitrile at 35 - 55℃; for 6.5h; Inert atmosphere; Large scale; | 96.8% |
| With pyridine at 85℃; for 8h; | 88% |
| With pyridine at 146℃; for 4h; | 43% |
-
-
108-24-7
acetic anhydride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
344-62-7
N-[2-(trifluoromethyl)phenyl]acetamide

| Conditions | Yield |
|---|---|
| With 2,3,4,5,7,8,9,10-octahydropyrimido[1,2-a]azepin-1-ium acetate In neat (no solvent) at 50℃; for 0.5h; Green chemistry; | 96% |
| at 80℃; for 5h; Large scale; | 95% |
| Stage #1: acetic anhydride; 2-(trifluoromethyl)benzenamine at 20℃; for 1h; Stage #2: With water; sodium hydrogencarbonate | 92% |
-
-
50-00-0
formaldehyd

-
-
1736-71-6
2-trifluoromethylphenyl thiourea

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
701277-32-9
1-(2-trifluoromethylphenyl)-5-(2-trifluoromethylphenyl)-2-thioxo hexahydro-1,3,5-triazine

| Conditions | Yield |
|---|---|
| In water for 0.0125h; microwave irradiation; | 96% |

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
68672-37-7
4-amino-3-(trifluoromethyl)phenyl thiocyanate

| Conditions | Yield |
|---|---|
| With bromine In methanol; dichloromethane | 96% |

| Conditions | Yield |
|---|---|
| With sodium phenoxide; C47H70BrO4PPdSi In tetrahydrofuran at 20℃; for 1h; Schlenk technique; Inert atmosphere; Sealed tube; | 96% |
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II); N,N,N′,N′-tetramethyl-N″-tert-butylguanidine; 4-phenyl-N-methylpyridinium iodide; 4,4'-di-tert-butyl-2,2'-bipyridine; zinc In dimethyl sulfoxide at 80℃; for 8h; Reagent/catalyst; Inert atmosphere; | 83% |
-
-
403-42-9
1-(4-fluorophenyl)ethanone

-
-
88-17-5
2-(trifluoromethyl)benzenamine

-
-
133671-30-4
N-<1-(4-fluorophenyl)ethylidene>-2-(trifluoromethyl)aniline

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 10h; Heating; | 95% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 T,
T,  Xi,
Xi,  Xn,
Xn,  F
F