-
Name
2,3,5,6-Tetrachloropyridine
- EINECS 219-283-9
- CAS No. 2402-79-1
- Article Data40
- CAS DataBase
- Density 1.662 g/cm3
- Solubility
- Melting Point 90.5 °C
- Formula C5HCl4N
- Boiling Point 253.7 °C at 760 mmHg
- Molecular Weight 216.882
- Flash Point 131.7 °C
- Transport Information
- Appearance
- Safety 26-36/37/39-60-61-36
- Risk Codes 20/21/22-36/37/38-51/53
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms NSC 2009;
- PSA 12.89000
- LogP 3.69520
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4,5,6-pentachloropyridine With manganese; ammonium acetate In ethanol; water at 40℃; for 6h; Stage #2: With ammonia In ethanol; water at 65℃; for 1h; pH=8 - 9; Concentration; Temperature; | 99.5% |
| With ammonium chloride; zinc In acetonitrile at 80℃; for 3h; Reagent/catalyst; Solvent; Temperature; Green chemistry; | 88.4% |
| With ammonium chloride; dimethyl methane phosphonate; zinc In water for 0.5h; Yield given; |

| Conditions | Yield |
|---|---|
| With hydrogen; potassium carbonate at 200℃; under 15001.5 Torr; for 2h; Autoclave; | 99.2% |
-
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
A
-
6515-09-9
2,3,6-trichloropyridine

-
B
-
16063-70-0
2,3,5-trichloropyridine

-
C
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| With ammonium chloride; dimethyl methane phosphonate; zinc In water at 85 - 90℃; Product distribution; other ammonium salts; other methylphosphonates/phosphates; | A 0.9% B 1% C 97.6% |

-
-
30332-35-5
tetrachloro-4-iodopyridine

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| In diethyl ether for 19h; Irradiation; | 87% |
| Multi-step reaction with 2 steps 1: 4 percent / copper bronze / dimethylformamide / 150 °C 2: 3 percent Chromat. / copper bronze / dimethylformamide / 20 h / 150 °C View Scheme |
-
-
41797-97-1
methyl 4-cyano-2,2,4-trichlorobutyrate

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| 79% |
-
-
27425-94-1
tetrachloro-4-pyridylcopper

-
-
98-88-4
benzoyl chloride

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
18613-89-3
4-benzoyltetrachloropyridine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 0.5h; Heating; | A 9% B 72% |

-
-
23995-94-0
4-Bromo-2,3,5,6-tetrachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
14121-36-9
2,3,4,6-Tetrachloropyridine

-
C
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
D
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With Isopropylbenzene; copper In N,N-dimethyl-formamide at 150℃; for 1h; Further byproducts given; | A 63% B 15% C 16% D 17% |

-
-
23995-94-0
4-Bromo-2,3,5,6-tetrachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
14121-36-9
2,3,4,6-Tetrachloropyridine

-
D
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With copper; benzoyl chloride In N,N-dimethyl-formamide at 150℃; for 0.5h; | A 61% B 24% C n/a D 15% |

-
-
30332-35-5
tetrachloro-4-iodopyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
C
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With copper In N,N-dimethyl-formamide at 150℃; | A 22% B 4% C 52% |

-
-
23995-94-0
4-Bromo-2,3,5,6-tetrachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
14121-36-9
2,3,4,6-Tetrachloropyridine

-
C
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
E
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With para-xylene; copper In N,N-dimethyl-formamide at 150℃; for 1h; Product distribution; Mechanism; the similar reaction without p-xylene; in presence cumene, benzoyl chloride instead p-xylene; other reagent (CuCl); other polychloroiodoarenes; tetrahalohenopyridylcopper intermediate; evidence against free radical or pyridyne intermediates; | A 42% B 5% C 15% D 4% E 18% |

-
-
23995-94-0
4-Bromo-2,3,5,6-tetrachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
D
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With para-xylene; copper In N,N-dimethyl-formamide at 150℃; for 1h; Further byproducts given; | A 42% B 15% C 4% D 18% |
| With copper In N,N-dimethyl-formamide at 150℃; for 0.5h; | A 17% B 21% C 8% D 14% |
| With copper In N,N-dimethyl-formamide at 150℃; for 0.5h; Further byproducts given; | A 10% B 10% C 9% D 8% |

-
-
23995-94-0
4-Bromo-2,3,5,6-tetrachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
74894-06-7
4-bromo-2,3,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| In diethyl ether for 71h; Irradiation; | A 17% B 6% |
-
-
27425-94-1
tetrachloro-4-pyridylcopper

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 1h; Heating; | A 13.5% B 1% |
-
-
27425-94-1
tetrachloro-4-pyridylcopper

-
-
98-88-4
benzoyl chloride

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
18613-89-3
4-benzoyltetrachloropyridine

-
C
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
D
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With para-xylene In N,N-dimethyl-formamide for 23h; Product distribution; Mechanism; Heating; the similar reaction without p-xylene; other reaction times; similar range of products to those obtained from 4-bromotetrachloropyridine; | A 12% B 13% C 1% D 2.5% |

-
-
27425-94-1
tetrachloro-4-pyridylcopper

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
18613-89-3
4-benzoyltetrachloropyridine

-
C
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
D
-
18725-60-5
octachloro-4,4'-bipyridyl

| Conditions | Yield |
|---|---|
| With para-xylene; benzoyl chloride In N,N-dimethyl-formamide for 23h; Heating; | A 12% B 13% C 1% D 2.5% |


| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 210 - 220℃; | |
| With sulfur dichloride In tetrachloromethane at 0 - 80℃; for 6h; Temperature; Solvent; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride; trichlorophosphate at 180℃; |

| Conditions | Yield |
|---|---|
| bei der Destillation; unter Kohlendioxyd-Entwicklung; |
-
-
19340-26-2
tetrachloroisonicotic acid

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| With water at 180℃; | |
| With glycerol beim Destillieren; |
-
-
76942-19-3
2,6-diamino-3,5-dichloropyridine

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite Yield given; |
-
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
14121-36-9
2,3,4,6-Tetrachloropyridine

-
C
-
2808-86-8
2,3,4,5-tetrachloro-pyridine

-
D
-
19942-44-0
tetrachloro-4-dimethylaminopyridine

| Conditions | Yield |
|---|---|
| With copper In N,N-dimethyl-formamide at 150℃; for 20h; | A 3 % Chromat. B 22 % Chromat. C 2 % Chromat. D 13 % Chromat. |

-
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
14121-36-9
2,3,4,6-Tetrachloropyridine

-
C
-
2808-86-8
2,3,4,5-tetrachloro-pyridine

-
D
-
19942-44-0
tetrachloro-4-dimethylaminopyridine

| Conditions | Yield |
|---|---|
| With copper In N,N-dimethyl-formamide at 150℃; for 20h; | A 3 % Chromat. B 22 % Chromat. C 2 % Chromat. D 13 % Chromat. |

-
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
20543-31-1
bis(tetrachloropyridyl)mercury

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide Hg cathode, -1.40 V (S.C.E.), Et4NBF4; | A 0.66 g B 0.59 g |
-
-
2176-62-7
2,3,4,5,6-pentachloropyridine

-
A
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
B
-
36277-69-7
2,3,5,6,2',3',6'-heptachloro-[4,4']bipyridinyl

| Conditions | Yield |
|---|---|
| With carbon dioxide 1.) Hg cathode, DMF, Et4NBF4, -1.30 V (S.C.E.), 2.) 150 deg C, vacuum; Multistep reaction; |

| Conditions | Yield |
|---|---|
| at 180℃; im Rohr; |
-
-
16063-70-0
2,3,5-trichloropyridine

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hexachloroethane; tetrabutylammomium bromide at 50℃; for 4h; |

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In isopropyl alcohol at 20℃; for 6h; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 60 percent / aq. HCl, H2O2 2: NaNO2-HCl View Scheme |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
16063-70-0
2,3,5-trichloropyridine

| Conditions | Yield |
|---|---|
| With 5%-palladium/activated carbon; hydrogen; potassium hydroxide In 5,5-dimethyl-1,3-cyclohexadiene; water at 60℃; under 4500.45 Torr; for 6h; Reagent/catalyst; Solvent; Pressure; Autoclave; | 99.1% |
| With sodium hydroxide In benzene |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 120℃; for 3h; | 99% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
214907-25-2
(4-methoxystyrene)boronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; di-(1-adamantyl)-n-butylphosphine; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 20h; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
851660-03-2
4-trifluoromethylstyrenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; di-(1-adamantyl)-n-butylphosphine; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 20h; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
214907-11-6
4-methylstyrenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; di-(1-adamantyl)-n-butylphosphine; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 20h; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
98-80-6
phenylboronic acid

-
-
24301-97-1
tetraphenyl-2,3,5,6 pyridine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate In toluene at 100℃; for 22h; Reagent/catalyst; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 99% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
54932-38-6
2,3,5,6-tetrakis(4-methoxyphenyl)pyridine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 50℃; for 1h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,5,6-tetrachloropyridine With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; dichloro bis(acetonitrile) palladium(II); copper(l) iodide; diisopropylamine In 1,4-dioxane at 20℃; for 0.166667h; Inert atmosphere; Stage #2: phenylacetylene In 1,4-dioxane at 90℃; for 20h; | 96% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
5720-05-8
4-methylphenylboronic acid

-
-
54932-37-5
2,3,5,6-tetra-p-tolylpyridine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 96% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
55933-94-3
3,5,6-trichloro-2-hydrazinopyridine

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In methanol at 60 - 65℃; for 2h; Temperature; Green chemistry; | 96% |
| With sodium carbonate; hydrazine hydrate; butan-1-ol In water at 90 - 95℃; |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 120℃; for 3h; | 95% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
4363-35-3, 6783-05-7, 60806-02-2
Dihydroxy-styryl-boran

| Conditions | Yield |
|---|---|
| With potassium phosphate; di-(1-adamantyl)-n-butylphosphine; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 20h; Reagent/catalyst; Solvent; Suzuki-Miyaura Coupling; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With potassium fluoride In sulfolane at 120℃; for 6h; Reagent/catalyst; Solvent; Green chemistry; | 95% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
762287-57-0
(4-chloro-2-methoxyphenyl)boronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,2-dimethoxyethane for 6h; Inert atmosphere; Reflux; | 95% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
16419-60-6
2-Methylphenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium fluoride; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; | 95% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
63139-21-9
4-ethylphenylboronic acid

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 94% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride; potassium carbonate In tetrahydrofuran for 5h; Reagent/catalyst; Solvent; Reflux; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,5,6-tetrachloropyridine With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; dichloro bis(acetonitrile) palladium(II); copper(l) iodide; diisopropylamine In 1,4-dioxane at 20℃; for 0.166667h; Inert atmosphere; Stage #2: 4-methoxyphenylacetylen In 1,4-dioxane at 90℃; for 20h; | 93% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
32316-92-0
naphthalene-2-boronic acid

| Conditions | Yield |
|---|---|
| With potassium fluoride; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; | 91% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In water; acetonitrile for 4h; Inert atmosphere; Reflux; | |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In water; acetonitrile for 4h; Inert atmosphere; Reflux; |

| Conditions | Yield |
|---|---|
| With potassium fluoride; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; | 90% |
| With potassium fluoride; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; regioselective reaction; | 77% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In water; acetonitrile for 4h; Inert atmosphere; Reflux; | |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In water; acetonitrile for 4h; Reflux; Inert atmosphere; |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
123324-71-0
4-tert-butylphenylboronic acid

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate In toluene at 100℃; for 22h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; | 90% |
-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
1428946-28-4
5'-tert-butyl-(1,1':3',1"-terphenyl)-3,3"-diol

-
-
1428946-32-0
C54H42Cl4N2O4

| Conditions | Yield |
|---|---|
| With caesium carbonate In dimethyl sulfoxide at 120℃; for 0.5h; Inert atmosphere; | 89.4% |
-
-
421-20-5
Methyl fluorosulfonate

-
-
2402-79-1
2,3,5,6-tetrachloropyridine

-
-
75348-52-6
3,5,6-trichloro-1-methyl-2-pyridone

| Conditions | Yield |
|---|---|
| for 0.5h; Heating; | 89% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diisopropylamine In 1,4-dioxane at 90℃; for 20h; Sonogashira Cross-Coupling; Inert atmosphere; Sealed tube; regioselective reaction; | 89% |
| Stage #1: 2,3,5,6-tetrachloropyridine With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane at 20℃; for 0.166667h; Sonogashira Cross-Coupling; Inert atmosphere; Stage #2: 1-ethynyl-4-fluorobenzene In 1,4-dioxane at 90℃; for 20h; Sonogashira Cross-Coupling; Inert atmosphere; | 68% |
2,3,5,6-Tetrachloropyridine Chemical Properties
Molar mass:216.88014g/mol
Structure of 2,3,5,6-Tetrachloropyridine(2402-79-1):
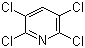
Synonyms of 2,3,5,6-Tetrachloropyridine(2402-79-1):Pyridine, 2,3,5,6-tetrachloro-;Tetrapyridine
Density:1.662 g/cm3
Flash Point:131.7 °C
Boiling Point:253.7 °C at 760 mmHg
Index of Refraction:1.588
Vapour Pressure:0.0288 mmHg at 25°C
Melting point:90.5 °C
2,3,5,6-Tetrachloropyridine Uses
2,3,5,6-Tetrachloropyridine(2402-79-1) can be used as the intermediate of medicine and pesticide.
2,3,5,6-Tetrachloropyridine(2402-79-1) can be used as the main raw material of synthetic herbicides: picloram,too .
2,3,5,6-Tetrachloropyridine Toxicity Data With Reference
| 1. | ipr-mus LD50:1150 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 11 (1967),361. |
2,3,5,6-Tetrachloropyridine Consensus Reports
2,3,5,6-Tetrachloropyridine Safety Profile
Hazard Codes:Xi

Risk Statements:
20: Harmful by inhalation
21: Harmful in contact with skin
22: Harmful if swallowed
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
51: Toxic to aquatic organisms
53: May cause long-term adverse effects in the aquatic environment
Safety Statements:
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37: Wear suitable gloves
39: Wear eye/face protection
60: This material and/or its container must be disposed of as hazardous waste
61: Avoid release to the environment. Refer to special instructions safety data sheet
2,3,5,6-Tetrachloropyridine Specification
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 2402-83-7
- 2402-95-1
- 2402-96-2
- 2402-97-3
- 2402-99-5
- 2403-22-7
- 24032-35-7
- 24032-84-6
- 24033-03-2
- 24034-73-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View