-
Name
Isopropyl iodide
- EINECS 200-859-3
- CAS No. 75-30-9
- Article Data106
- CAS DataBase
- Density 1.747 g/cm3
- Solubility
- Melting Point -90 °C
- Formula C3H7I
- Boiling Point 91.3 °C at 760 mmHg
- Molecular Weight 169.993
- Flash Point 42.2 °C
- Transport Information UN 2392 3/PG 3
- Appearance Colorless liquid
- Safety 36/37
- Risk Codes 10-22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 1-Methylethyliodide;2-Propyl iodide;Isopropyl iodide;sec-Propyl iodide;
- PSA 0.00000
- LogP 1.82980
Synthetic route

| Conditions | Yield |
|---|---|
| With iodoform; sodium iodide In N,N-dimethyl-formamide for 3h; Catalytic behavior; Irradiation; | 91% |
| With acetyl iodide at 80 - 90℃; for 1.5h; | 24% |
| With iodine; aluminium |
-
-
26591-86-6
(i-C3H7)3SbI2

-
A
-
75-30-9
2-iodo-propane

-
B
-
73300-45-5
tri(isopropyl)antimony

-
C
-
81492-14-0
diisopropylantimony iodide

| Conditions | Yield |
|---|---|
| under Ar, decomposed at 135°C for 30 min; distd., not separated from triisopropyl stibine; elem. anal.; | A 89% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With 2-fluoro-2-(piperidin-1-yl)-4-(2,3,5,6-tetrafluoropyridin-4-yl)-5-(2,4,6-triisopropylphenyl)-1,3-dithiole In 1,4-dioxane at 45℃; for 14h; Schlenk technique; Inert atmosphere; | 74% |

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide In chlorobenzene for 3h; Irradiation; | A 51% B 18% |

| Conditions | Yield |
|---|---|
| With CCl4*2AlI3 In dichloromethane at -20℃; for 1.5h; | 50% |
-
-
137116-57-5
5'-O-(4,4'-dimethoxytrityl)-2-N-<2-<(tert-butyldiphenylsilyloxy)methyl>benzoyl>guanosine

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
A
-
75-30-9
2-iodo-propane

-
B
-
137116-54-2
5'-O-(4,4'-dimethoxytrityl)-3'-O-(tert-butyldimethylsilyl)-2-N-<2-<(tert-butyldiphenylsilyloxy)methyl>benzoyl>guanosine

| Conditions | Yield |
|---|---|
| With pyridine; silver nitrate In tetrahydrofuran | A 42% B 16% |


| Conditions | Yield |
|---|---|
| With water; hydrogen iodide |
-
-
187737-37-7
propene

-
-
75-30-9
2-iodo-propane

| Conditions | Yield |
|---|---|
| With hydrogen iodide | |
| With ascaridole; hydrogen iodide | |
| With hydrogen iodide; diphenylamine | |
| With hydrogen iodide |

| Conditions | Yield |
|---|---|
| With hydrogen iodide; diphenylamine |

| Conditions | Yield |
|---|---|
| With hydrogen iodide |
-
-
29568-69-2
1,2-iodochloropropane

-
-
75-30-9
2-iodo-propane

| Conditions | Yield |
|---|---|
| With hydrogen iodide |

| Conditions | Yield |
|---|---|
| With phosphoric acid; potassium iodide | |
| With diiodosilane In chloroform-d1 Ambient temperature; relative rates, other reagents, var. ethers; |

| Conditions | Yield |
|---|---|
| With water; potassium iodide |

| Conditions | Yield |
|---|---|
| mit UV-Licht (253.7 nm),in fluess. Zustand;bei Abwesenheit von Sauerstoff.Irradiation; |


| Conditions | Yield |
|---|---|
| With phosphorus; water; iodine und nachfolgendes Dastillieren des Reaktionsprodukts; | |
| With water; hydrogen iodide | |
| With phosphorous; water; iodine | |
| With hydrogen iodide In water; benzene at 100℃; under 15514.9 Torr; for 16h; Inert atmosphere; Autoclave; | 100 %Chromat. |

| Conditions | Yield |
|---|---|
| With pyridine; tributyltin iodide at 125℃; Thermodynamic data; Equilibrium constant; Δ G; | |
| With sodium iodide In acetone at 55℃; Kinetics; quantitative reaction kinetics in liquid state by in situ X-ray absorption fine structure spectroscopy; | |
| With acetone; sodium iodide |
-
-
116-17-6
triisopropyl phosphite

-
-
74-88-4
methyl iodide

-
A
-
75-30-9
2-iodo-propane

-
B
-
1445-75-6
diisopropyl methanephosphonate

| Conditions | Yield |
|---|---|
| reagiert analog mit Brom-diphenyl-p-biphenylyl-methan und Homologen; |


| Conditions | Yield |
|---|---|
| With diiodosilane In chloroform-d1 at 22℃; for 4h; | 99 % Spectr. |
-
-
556-56-9
allyl iodid

-
-
593-71-5
Chloroiodomethane

-
-
1068-55-9
isopropylmagnesium chloride

-
A
-
75-30-9
2-iodo-propane

-
B
-
513-38-2
Isobutyl iodide

-
C
-
7766-51-0
4-iodobut-1-ene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether at -40℃; for 4.5h; Product distribution; | |
| In tetrahydrofuran at -40℃; for 4.5h; |

-
-
76509-66-5
diisopropyl ethylphosphonite

-
-
754-34-7
1,1,1,2,2,3,3-heptafluoro-3-iodo-propane

-
A
-
75-30-9
2-iodo-propane

-
B
-
77529-57-8
isopropyl ethyl(heptafluoropropyl)phosphinate

| Conditions | Yield |
|---|---|
| In octane Rate constant; Kinetics; Thermodynamic data; E (activ.); ΔH(excit.); ΔS(excit.); ΔG(excit.); |

-
-
187737-37-7
propene

-
-
64-19-7
acetic acid

-
A
-
75-30-9
2-iodo-propane

-
B
-
108-21-4
Isopropyl acetate

-
C
-
67-63-0
isopropyl alcohol

| Conditions | Yield |
|---|---|
| With carbon monoxide; water; hydrogen iodide; TsVK-2; palladium at 110℃; under 60800 Torr; for 2h; Product distribution; further temperature (130 deg C); |


| Conditions | Yield |
|---|---|
| With diiodosilane; iodine 1.) CDCl3, 22 deg C, 10 min, 2.) 30 min; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With diiodosilane; iodine In chloroform-d1 at 22℃; for 3h; | 100 % Spectr. |

| Conditions | Yield |
|---|---|
| With diiodosilane In chloroform-d1 for 0.166667h; Ambient temperature; other reaction time: 80 min, effect of iodine; Yield given. Further byproducts given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
187737-37-7
propene

-
-
201230-82-2
carbon monoxide

-
-
79-31-2
isobutyric Acid

-
A
-
75-30-9
2-iodo-propane

-
B
-
617-50-5
isopropyl isobutyrate

-
C
-
638-11-9
isopropyl butyrate

-
D
-
67-63-0
isopropyl alcohol

-
E
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With water; hydrogen iodide; TsVK-2; palladium at 130℃; under 60800 Torr; for 2h; Product distribution; further temperature (110, 150, 170, 200 deg C); effect of the concentration of HI; effect of the pressure of CO; |

-
-
2025-55-0
isopropyl radical

-
-
74-88-4
methyl iodide

-
A
-
75-30-9
2-iodo-propane

-
B
-
2229-07-4
methyl radical

| Conditions | Yield |
|---|---|
| Equilibrium constant; |


-
-
108-95-2
phenol

-
A
-
75-30-9
2-iodo-propane

-
B
-
56-23-5
tetrachloromethane

-
C
-
2122-46-5, 55748-05-5
phenoxy radical

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 22℃; Rate constant; multistep reaction; |


| Conditions | Yield |
|---|---|
| With trimethylsilyl iodide; bismuth(III) iodide In neat (no solvent) at 80℃; for 0.25h; | 98 % Chromat. |
-
-
626-67-5
N-methylcyclohexylamine

-
-
75-30-9
2-iodo-propane

-
-
84437-36-5
1-isopropyl-1-methyl-piperidinium; iodide

| Conditions | Yield |
|---|---|
| In chloroform at 60℃; for 8h; | 100% |
| In chloroform Heating; |
-
-
75-30-9
2-iodo-propane

-
-
5597-27-3
3-methylenebicyclo[2.2.1]heptan-2-one

-
-
108350-17-0
3-isobutylbicyclo<2.2.1>heptan-2-one

| Conditions | Yield |
|---|---|
| With copper(l) iodide; zinc In water Product distribution; further solvent: aq. ethanol, further activators: ammonium formate, ammonium chloride; ultrasonic irradiation; | 100% |
| With copper(l) iodide; zinc In water ultrasonic irradiation; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
481-39-0
5-hydroxynaphtho-1,4-quinone

-
-
97308-90-2
5-isopropoxy-1,4-naphthoquinone

| Conditions | Yield |
|---|---|
| With silver(l) oxide In dichloromethane at 20℃; for 20h; Green chemistry; regioselective reaction; | 100% |
| With silver(l) oxide In chloroform for 0.333333h; Ambient temperature; | 77% |
-
-
75-30-9
2-iodo-propane

| Conditions | Yield |
|---|---|
| In acetonitrile for 43h; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
119289-79-1
3-(4-hydroxy-2-nitrophenyl)pyridine

-
-
158461-41-7
3-(4-isopropoxy-2-nitrophenyl)pyridine

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 40℃; for 6h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium azide; [bis(acetoxy)iodo]benzene; trifluoroacetic acid In isopropyl alcohol Product distribution; various solvents; | 100% |
| With sodium azide; [bis(acetoxy)iodo]benzene; trifluoroacetic acid In isopropyl alcohol Heating; | 100% |
| With tert.-butylhydroperoxide; acetic acid ; ferriacetate; trifluoroacetic acid In acetic acid for 4h; Heating; | 92% |
-
-
75-30-9
2-iodo-propane

-
-
101-41-7
benzeneacetic acid methyl ester

-
-
72615-27-1
2-phenyl-3-methylbutyric acid methyl ester

| Conditions | Yield |
|---|---|
| With N,N,N,N,N,N-hexamethylphosphoric triamide; lithium diisopropyl amide In tetrahydrofuran at -78℃; | 100% |
| With 2-pyrrolidinon In N,N-dimethyl-formamide Flow reactor; | 16% |
| With 1) EGB.2 1) -78 deg C, 2) -78 deg C, 15 min; Yield given. Multistep reaction; |
-
-
75-30-9
2-iodo-propane

-
-
159647-63-9
3-(2,3-Dihydro-benzo[1,4]dioxin-6-yl)-2,6-diethyl-7-hydroxy-chromen-4-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 34.5h; Heating; | 100% |
| With potassium carbonate In acetone Heating; | 99.8% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
699-18-3, 32782-45-9
2-[(E)-2-nitroethenyl]furan

-
-
20992-64-7
(E)-1-(2-furyl)-3-methyl-1-butene

| Conditions | Yield |
|---|---|
| With triethylaluminum; dibenzoyl peroxide In diethyl ether at 20℃; for 2h; | 100% |
| With air; triethyl borane In tetrahydrofuran at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With air; triethyl borane In tetrahydrofuran at 20℃; | 100% |
| With triethylaluminum; dibenzoyl peroxide In diethyl ether at 20℃; for 2h; | 98% |
-
-
75-30-9
2-iodo-propane

-
-
3179-10-0, 5576-97-6
(E)-1-methoxy-4-(2-nitrovinyl)benzene

-
-
103786-20-5
(E)-1-methoxy-4-(3-methyl-but-1-en-1-yl)benzene

| Conditions | Yield |
|---|---|
| With triethyl borane; oxygen In tetrahydrofuran at 20℃; | 100% |
| With triethylaluminum; dibenzoyl peroxide In diethyl ether at 20℃; for 2h; | 99% |
| With triethyl borane In diethyl ether; water | |
| With triethyl borane In tetrahydrofuran at 20℃; |
-
-
75-30-9
2-iodo-propane

-
-
101671-01-6
1-nitro-2-(4-chlorophenyl)ethylene

| Conditions | Yield |
|---|---|
| With triethylaluminum; dibenzoyl peroxide In diethyl ether at 20℃; for 2h; | 100% |
| With air; triethyl borane In tetrahydrofuran at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile) In benzene at 80℃; for 4h; | 100% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; water; sodium chloride In acetonitrile for 20h; pH=2; Solvent; pH-value; Electrochemical reaction; Green chemistry; | 100% |
| With 2,2'-azobis(isobutyronitrile); tert-butyl(2,6-dimethoxy-1-methylcyclohexa-2,5-dien-1-yl)dimethylsilane In hexane for 14h; Giese-type addition; Heating; | 89% |
| With indium In methanol; water at 20℃; for 0.5h; | 86% |
-
-
75-30-9
2-iodo-propane

| Conditions | Yield |
|---|---|
| With triethyl borane; tri-n-butyl-tin hydride; magnesium bromide In hexane; dichloromethane at 0℃; for 1h; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
401819-62-3
4-(7-hydroxy-6-methoxy-4-quinazolinyl)-1-piperazinecarboxylic acid tert-butyl ester

-
-
205259-43-4
4-(7-isopropoxy-6-methoxy-4-quinazolinyl)-1-piperazinecarboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
63435-16-5
3-hydroxy-4-aminobenzoic acid methyl ester

-
-
681465-85-0
4-amino-3-isopropoxybenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetone at 60℃; for 2h; Inert atmosphere; | 100% |
| With caesium carbonate In acetone for 10h; Heating; | 90% |
| With caesium carbonate In acetone for 10h; Heating / reflux; | 90% |
| With caesium carbonate In acetone at 120℃; for 1h; Microwave; | 60% |
| With caesium carbonate In acetone |
-
-
75-30-9
2-iodo-propane

-
-
1439-36-7
1-triphenylphosphoranylidene-2-propanone

-
-
27653-95-8
4-methyl-1-(triphenylphosphaneylidene)pentan-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 1-triphenylphosphoranylidene-2-propanone With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: 2-iodo-propane In tetrahydrofuran; hexane at -78 - 20℃; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
49543-63-7
4-tert-butylbenzyl mercaptan

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-butylbenzyl mercaptan With tetra-(n-butyl)ammonium iodide; caesium carbonate In N,N-dimethyl-formamide at 20℃; for 1h; Stage #2: 2-iodo-propane In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 9H-fluorene With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 1.5h; Stage #2: 2-iodo-propane In tetrahydrofuran; hexane at 20℃; for 2h; Further stages.; | 100% |
| Stage #1: 9H-fluorene With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -15℃; for 0.333333h; Stage #2: 2-iodo-propane In tetrahydrofuran; hexane at -15℃; for 0.5h; Further stages.; | |
| With lithium diisopropyl amide In tetrahydrofuran at -10℃; |
-
-
75-30-9
2-iodo-propane

-
-
70768-92-2
5,6-bis(4-methoxyphenyl)-2H-pyridazin-3-one

| Conditions | Yield |
|---|---|
| 100% |
-
-
308386-35-8
1,1-dimethylethyl 4-(4'-pyridinyloxy)-1-piperidinecarboxylate

-
-
75-30-9
2-iodo-propane

-
-
845305-79-5
4-[(1-{tert-butoxycarbonyl}-4-piperidinyl)oxy]-1-(1-methylethyl)pyridinium iodide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; | 100% |
| In dichloromethane at 90℃; for 2.5 - 48h; |
-
-
75-30-9
2-iodo-propane

-
-
156001-68-2
methyl 3-cyano-4-hydroxybenzoate

-
-
213598-11-9
3-cyano-4-isopropoxybenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 50℃; for 16h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 50℃; for 16h; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; for 12h; Inert atmosphere; |
-
-
75-30-9
2-iodo-propane

-
-
123148-78-7
4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidine

-
-
213744-81-1
4-chloro-5-iodo-7-isopropyl-7H-pyrrolo[2,3-d]pyrimidine

| Conditions | Yield |
|---|---|
| Stage #1: 4-chloro-5-iodo-7H-pyrrolo[2,3-d]pyrimidine With caesium carbonate In N,N-dimethyl-formamide at 20℃; for 0.05h; Stage #2: 2-iodo-propane | 100% |
| In N,N-dimethyl-formamide |
-
-
75-30-9
2-iodo-propane

-
-
194237-29-1
5-(1-methyl-6-trifluoromethyl-2(1H)-pyridon-3-yl)-2-chloro-4-fluorobenzoic acid

-
-
194236-57-2
isopropyl 5-(1-methyl-6-trifluoromethyl-2(1H)-pyridon-3-yl)-2-chloro-4-fluorobenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 100% |
-
-
75-30-9
2-iodo-propane

-
-
344331-90-4
tert-butyl 6-bromopyridin-2-ylcarbamate

-
-
884539-05-3
tert-butyl N-(6-bromo-2-pyridyl)-N-isopropylcarbamate

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; for 25.5h; | 100% |
-
-
75-30-9
2-iodo-propane

-
-
914397-24-3
7-benzyloxy-6-bromo-2-isopropylaminoquinazoline

| Conditions | Yield |
|---|---|
| Stage #1: 7-benzyloxy-6-bromo-2-aminoquinazoline With sodium hydride In N,N-dimethyl-formamide at 5℃; for 0.5h; Stage #2: 2-iodo-propane In N,N-dimethyl-formamide at 60℃; for 1.5h; Stage #3: 2-iodo-propane With sodium hydride Product distribution / selectivity; more than 3 stages; | 100% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran-d8 byproducts: I(1-); react. in a glove box under Ar at 20°C, addn. of the organic halide via syringe to a soln. of Cp3UiPr(1-) in THF-d8, react. time: 10 min; determination by 1H NMR spectroscopy; | 100% |
2-Iodopropane Chemical Properties
The Molecular Weight of ISOPROPYL IODIDE(75-30-9): 169.99
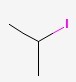
The Molecular Structure of ISOPROPYL IODIDE(75-30-9):
EINECS: 200-859-3
Melting point: -90 °C
Boiling Point: 91.3 °C at 760 mmHg
Flash Point: 42.2 °C
Index of Refraction: 1.505
Molar Refractivity: 28.87 cm3
Molar Volume: 97.2 cm3
Polarizability: 11.44 10-24 cm3
Surface Tension: 27.5 dyne/cm
Density: 1.747 g/cm3
Enthalpy of Vaporization: 30.68 kJ/mol
Vapour Pressure: 61.4 mmHg at 25°C
storage temp.: Store at RT.
Sensitive: Light Sensitive
Stability: Stable, but may discolour on exposure to light. Air sensitive. Incompatible with strong bases, strong oxidizing agents.
Appearance: Colorless liquid
Merck: 14,5214
BRN: 1098244
IUPAC Name: 2-iodopropane
Synonyms: 2-iodo-propan;2-Propyljodid;Isopropyl iodine;Isopropyliodid;i-Propyl iodide;iso-C3H7I;2-Jodpropan;2-propyliodide;
2-Iodopropane Uses
2-Iodopropane Toxicity Data With Reference
| 1. | ihl-rat LC50:320,000 mg/m3/30M | FAVUAI Fiziologicheski Aktivnye Veshchestva. Physiologically Active Substances. 7 (1975),35. | ||
| 2. | ipr-mus LD50:1300 mg/kg | 34ZIAG Toxicology of Drugs and Chemicals ,Deichmann, W.B.,New York, NY.: Academic Press, Inc.,1969,756. |
2-Iodopropane Consensus Reports
2-Iodopropane Safety Profile
The Hazard Codes of ISOPROPYL IODIDE(75-30-9):
 Xn
Xn HazardClass: 3
The Risk Statements information of ISOPROPYL IODIDE(75-30-9):
10: Flammable
22: Harmful if swallowed
The Safety Statements information of ISOPROPYL IODIDE(75-30-9):
36/37: Wear suitable protective clothing and gloves
RIDADR: UN 2392 3/PG 3
WGK Germany: 3
RTECS: TZ4200000
F: 8
PackingGroup: III
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View