-
Name
2-Methyl-2-phenylpropionic acid
- EINECS 212-556-3
- CAS No. 826-55-1
- Article Data96
- CAS DataBase
- Density 1.089 g/cm3
- Solubility Insoluble in water
- Melting Point 80-82 °C
- Formula C10H12O2
- Boiling Point 270.5 °C at 760 mmHg
- Molecular Weight 164.204
- Flash Point 167.7 °C
- Transport Information
- Appearance White crystalline
- Safety 26-36/37/39
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols Xi
- Synonyms 2,2-Dimethyl-2-phenylacetic acid;2-Phenyl-2-methylpropionic acid;2-Phenylisobutyric acid;NSC 28952;NSC 29095;TH 4161;a,a-Dimethylbenzeneacetic acid;a,a-Dimethylphenylacetic acid;Hydratropicacid, a-methyl- (6CI,7CI,8CI);
- PSA 37.30000
- LogP 2.04880
Synthetic route
-
-
67174-28-1
diphenyl N-<2-methyl-2-phenyl-1-(1-pyrrolidinyl)propylidene>phosphoramidate

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethylene glycol Heating; | 100% |
-
-
57625-74-8
methyl 2-methyl-2-phenylpropionate

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In 1,4-dioxane for 24h; | 98% |
| With sodium hydroxide In methanol; water for 4h; Ambient temperature; Yield given; | |
| With sodium hydroxide; water In methanol at 80℃; for 12h; Hydrolysis; |

| Conditions | Yield |
|---|---|
| With Ag nano wire core encapsulated by a N-doped carbon shell at 700 °C In acetonitrile at 25℃; under 750.075 Torr; Electrochemical reaction; | 91% |
| With nitrogen-doped carbon nanofibers embedded with platinum nanoparticles onto flexible carbon cloth In acetonitrile at 25℃; under 760.051 Torr; Electrochemical reaction; | 92 %Chromat. |

| Conditions | Yield |
|---|---|
| With water; potassium hydroxide In ethylene glycol at 100℃; for 48h; Inert atmosphere; | 73% |
-
-
124-38-9
carbon dioxide

-
-
4148-93-0
2-phenyl-2-propyl phenyl sulfide

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
2973-86-6
lithium thiophenoxide

| Conditions | Yield |
|---|---|
| Stage #1: 2-phenyl-2-propyl phenyl sulfide With lithium In diethyl ether at 3 - 8℃; for 2h; Stage #2: carbon dioxide In diethyl ether | A 73% B n/a |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; silver nitrate for 2h; cooling; | 70% |
| With silver(l) oxide |
-
-
101-97-3
Ethyl 2-phenylethanoate

-
-
74-88-4
methyl iodide

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
2901-13-5
ethyl 2-methyl-2-phenylpropionate

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 10 - 20℃; for 16h; Alkylation; | A 28% B 52% |


| Conditions | Yield |
|---|---|
| With water; bis-triphenylphosphine-palladium(II) chloride In butanone at 115℃; under 40504.1 Torr; for 5.5h; | 50% |

| Conditions | Yield |
|---|---|
| With (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; potassium carbonate; caesium carbonate In N,N-dimethyl-formamide at 25 - 30℃; for 36h; Schlenk technique; Glovebox; Sealed tube; Irradiation; | 43% |

| Conditions | Yield |
|---|---|
| With ammonia; potassium amide anschliessend Behandeln mit CH3I; |
-
-
98-82-8
Isopropylbenzene

-
-
88982-51-8
propyl potassium

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
536-66-3
4-Isopropylbenzoic acid

-
C
-
5651-47-8
3-isopropylbenzoic acid

-
D
-
2438-04-2
2-isopropylbenzoic acid

| Conditions | Yield |
|---|---|
| at 90℃; Product distribution; anschliessend Behandeln mit festem Kohlendioxid; |

-
-
98-82-8
Isopropylbenzene

-
-
2875-37-8
pentyl potassium

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
536-66-3
4-Isopropylbenzoic acid

-
C
-
5651-47-8
3-isopropylbenzoic acid

-
D
-
2438-04-2
2-isopropylbenzoic acid

| Conditions | Yield |
|---|---|
| at 20℃; Product distribution; anschliessend Behandeln mit festem Kohlendioxid; |

-
-
98-82-8
Isopropylbenzene

-
-
4522-40-1
ethyl potassium

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
536-66-3
4-Isopropylbenzoic acid

-
C
-
5651-47-8
3-isopropylbenzoic acid

-
D
-
2438-04-2
2-isopropylbenzoic acid

| Conditions | Yield |
|---|---|
| at 20℃; Product distribution; anschliessend Behandeln mit festem Kohlendioxid; | |
| at 85℃; Product distribution; anschliessend Behandeln mit festem Kohlendioxid; |


| Conditions | Yield |
|---|---|
| With sodium anschliessendes Behandeln mit festem Kohlendioxid; |
-
-
98-82-8
Isopropylbenzene

-
-
1822-71-5
n-pentylsodium

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
536-66-3
4-Isopropylbenzoic acid

-
C
-
5651-47-8
3-isopropylbenzoic acid

-
D
-
2438-04-2
2-isopropylbenzoic acid

| Conditions | Yield |
|---|---|
| at 20℃; Product distribution; anschliessend Behandeln mit festem Kohlendioxid; |

-
-
60-29-7
diethyl ether

-
-
3003-91-6
cumyl potassium

-
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
826-55-1
2-methyl-2-phenylpropionic acid


| Conditions | Yield |
|---|---|
| With aluminum tri-bromide; benzene |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite |
-
-
10409-54-8
2-bromo-2-methyl-1-phenyl-propan-1-one

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With ethanol; silver nitrate |

| Conditions | Yield |
|---|---|
| With alkaline solution | |
| With sulfuric acid | |
| With hydrogenchloride; sodium nitrite |
-
-
76173-50-7
2-methyl-1-nitro-2-phenylpropane

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With alkaline KMNO4 |

| Conditions | Yield |
|---|---|
| With carbon dioxide |
-
-
7473-99-6
α-chloroisobutyrophenone

-
A
-
826-55-1
2-methyl-2-phenylpropionic acid

-
B
-
7473-98-5
2-hydroxy-2-methylpropiophenone

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; silver-salt | |
| With 1,4-dioxane; mercury-salt |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; aluminium trichloride | |
| With aluminium trichloride | |
| With aluminum (III) chloride Industry scale; | |
| With aluminum (III) chloride Industry scale; |

| Conditions | Yield |
|---|---|
| With aluminium trichloride |

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide at 150℃; | |
| With aluminium trichloride |
-
-
64-18-6
formic acid

-
-
617-94-7
1-methyl-1-phenylethyl alcohol

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20 - 25℃; for 2h; |

| Conditions | Yield |
|---|---|
| (i) nBuLi, heptane, (ii) /BRN= 1900390/; Multistep reaction; |
-
-
22768-22-5, 22768-23-6, 6258-73-7
4-methyl-2,4-diphenylpent-2-ene

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| (i) O3, (ii) CrO3; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 80℃; for 3h; Inert atmosphere; | 100% |
-
-
67-56-1
methanol

-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
57625-74-8
methyl 2-methyl-2-phenylpropionate

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20 - 70℃; for 21.5h; Inert atmosphere; | 99.5% |
| With sulfuric acid at 70℃; for 12h; | 94% |
| With sulfuric acid Heating; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether Reduction; | 99% |
| Stage #1: 2-methyl-2-phenylpropionic acid With 1,1'-carbonyldiimidazole In tetrahydrofuran for 0.5h; Stage #2: With sodium tetrahydroborate In tetrahydrofuran; water | 90% |
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 2h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 16h; Schlenk technique; Inert atmosphere; | 99% |
-
-
109-72-8, 29786-93-4
n-butyllithium

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| Stage #1: n-butyllithium; 2-methyl-2-phenylpropionic acid In hexane at -80℃; Schlenk technique; Inert atmosphere; Stage #2: In hexane at 20℃; for 2h; Schlenk technique; Inert atmosphere; | 99% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
100-46-9
benzylamine

-
-
889798-61-2
N-benzyl-2-methyl-2-phenylpropanamide

| Conditions | Yield |
|---|---|
| With Bromotrichloromethane; 4-(diphenylphosphino)-benzyltrimethylammonium bromide; triethylamine In tetrahydrofuran at 60℃; for 15h; Inert atmosphere; | 98% |
| With 4-(2-(1,3-dioxa-3a1,8,10-triaza-2,3a,14b-triboradibenzo[fg,op]tetracen-2-yl)phenyl)benzo[c]pyrimido[4,5-e][1,2]azaborinin-6(5H)-ol In fluorobenzene at 85℃; for 8h; Molecular sieve; | 92% |
-
-
64-17-5
ethanol

-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
2901-13-5
ethyl 2-methyl-2-phenylpropionate

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-2-phenylpropionic acid With thionyl chloride In dichloromethane for 15h; Reflux; Stage #2: ethanol In dichloromethane | 96% |
| With thionyl chloride Reflux; | 88% |
| With potassium carbonate; dibromotriphenylphosphorane In acetonitrile at 60℃; for 72h; | 66% |

| Conditions | Yield |
|---|---|
| With oxygen; palladium diacetate; potassium hydrogencarbonate; p-benzoquinone In tert-Amyl alcohol at 85℃; under 760.051 Torr; for 48h; regioselective reaction; | 96% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In tetrahydrofuran for 2h; | 96% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
16386-97-3
2-cyclohexyl-2-methylpropanoic acid

| Conditions | Yield |
|---|---|
| With Rh/Al2O3; hydrogen In acetic acid under 5688.78 Torr; | 95% |
| With hydrogen; platinum(IV) oxide |
-
-
28509-24-2
tert-butylmethyl ether

-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
169280-16-4
2-methyl-2-phenyl-propionic acid , N-methoxy-N-methylamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; thionyl chloride; potassium carbonate In water; toluene | 95% |
| With hydrogenchloride; thionyl chloride; potassium carbonate In water; toluene | 95% |

-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
169280-16-4
2-methyl-2-phenyl-propionic acid , N-methoxy-N-methylamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-2-phenylpropionic acid With thionyl chloride In toluene at 20℃; Heating / reflux; Stage #2: N,O-dimethylhydroxylamine*hydrochloride With potassium carbonate In water; toluene for 2h; | 95% |

| Conditions | Yield |
|---|---|
| With C36H24B4N2O3 In toluene at 80℃; for 4h; Catalytic behavior; Reagent/catalyst; Temperature; Molecular sieve; | 95% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
40140-16-7
ethyl (p-methoxyphenyl)oxoacetate

| Conditions | Yield |
|---|---|
| With (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; caesium carbonate In dimethyl sulfoxide at 40℃; for 1h; Glovebox; Inert atmosphere; Irradiation; | 95% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
64922-05-0
1-chloro-3-methyl-3-phenylbutan-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-2-phenylpropionic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; Stage #2: diazomethane In tetrahydrofuran; diethyl ether at 0 - 20℃; for 12h; Stage #3: With hydrogenchloride In tetrahydrofuran; diethyl ether at 0℃; for 0.0833333h; | 94% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
104-94-9
4-methoxy-aniline

-
-
862876-03-7
N-(4-methoxyphenyl)-2-methyl-2-phenylpropanamide

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; N,N,N',N'-tetramethylchloroformamidinium hexafluorophosphate In acetonitrile at 23℃; for 0.166667h; | 94% |
| With C36H24B4N2O3 In toluene at 80℃; for 14h; Molecular sieve; | 89% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
106-47-8
4-chloro-aniline

-
-
862876-04-8
N-(4-chlorophenyl)-2-methyl-2-phenylpropanamide

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; N,N,N',N'-tetramethylchloroformamidinium hexafluorophosphate In acetonitrile at 23℃; for 0.333333h; | 94% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
81927-55-1
O-benzyl 2,2,2-trichloroacetimidate

| Conditions | Yield |
|---|---|
| In toluene for 18h; Inert atmosphere; Reflux; | 94% |
-
-
911422-98-5
tert-butyl (1S,4R,5R,7S)-7-amino-5-(3-isopropoxyphenyl)-4-methyl-2-azabicyclo[3.3.1]nonane-2-carboxylate

-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
911423-23-9
tert-butyl (1S,4R,5R,7S)-5-(3-isopropoxyphenyl)-4-methyl-7-[(2-methyl-2-phenylpropanoyl)amino]-2-azabicyclo[3.3.1]nonane-2-carboxylate

| Conditions | Yield |
|---|---|
| With (benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate; triethylamine In tetrahydrofuran at 20℃; for 4h; | 93% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
873-74-5
4-Aminobenzonitrile

-
-
862876-05-9
N-(4-cyanophenyl)-2-methyl-2-phenylpropanamide

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; N,N,N',N'-tetramethylchloroformamidinium hexafluorophosphate In acetonitrile at 23℃; for 21h; Reagent/catalyst; Solvent; | 93% |
-
-
37622-90-5
4-ethoxycarbonylpyrazole

-
-
826-55-1
2-methyl-2-phenylpropionic acid

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; tert-butylammonium hexafluorophosphate(V) In dichloromethane for 2.5h; Molecular sieve; Electrochemical reaction; | 93% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-2-phenylpropionic acid With thionyl chloride In toluene at 20℃; Heating / reflux; Stage #2: pyrrolidine With potassium carbonate In water; toluene for 2h; | 92% |

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; N,N,N',N'-tetramethylchloroformamidinium hexafluorophosphate In acetonitrile at 23℃; for 5h; | 92% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
616-38-6
carbonic acid dimethyl ester

-
-
57625-74-8
methyl 2-methyl-2-phenylpropionate

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 90℃; for 16h; Inert atmosphere; Green chemistry; | 91% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
1443209-30-0
tert-butyl 4-((2-(hydrazinecarbonyl)benzo[b]thiophen-3-yl)oxy)piperidine-1-carboxylate

-
-
1581303-74-3
tert-butyl 4-((2-(2-(2-methyl-2-phenylpropanoyl)hydrazinecarbonyl)benzo[b]thiophen-3-yl)oxy)piperidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 18h; | 91% |
-
-
826-55-1
2-methyl-2-phenylpropionic acid

-
-
62-53-3
aniline

-
-
58265-36-4
2-methyl-N,2-diphenylpropanamide

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; N,N,N',N'-tetramethylchloroformamidinium hexafluorophosphate In acetonitrile at 23℃; for 0.666667h; | 91% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 91% |
| Stage #1: N-hydroxyphthalimide; 2-methyl-2-phenylpropionic acid With dmap In dichloromethane for 0.0833333h; Inert atmosphere; Stage #2: With diisopropyl-carbodiimide In dichloromethane at 20℃; Inert atmosphere; | 90% |
| With dmap; diisopropyl-carbodiimide In tetrahydrofuran at 20℃; Inert atmosphere; | 87% |
| With dmap; diisopropyl-carbodiimide In dichloromethane | |
| With diisopropyl-carbodiimide In dichloromethane |
2-Methyl-2-phenylpropionic acid Chemical Properties
Product Name: 2-Methyl-2-phenylpropionic acid (CAS NO.826-55-1)
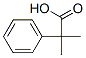
Molecular Formula: C10H12O2
Molecular Weight: 164.2g/mol
Mol File: 826-55-1.mol
EINECS: 212-556-3
Melting Point: 80-82°C
Boiling point: 270.5 °C at 760 mmHg
Flash Point: 167.7 °C
Density: 1.089 g/cm3
Surface Tension: 39.1 dyne/cm
Enthalpy of Vaporization: 53.73 kJ/mol
Vapour Pressure: 0.00336 mmHg at 25°C
XLogP3-AA: 1.6
H-Bond Donor: 1
H-Bond Acceptor: 2
Structure Descriptors of 2-Methyl-2-phenylpropionic acid (CAS NO.826-55-1):
IUPAC Name: 2-methyl-2-phenylpropanoic acid
Canonical SMILES: CC(C)(C1=CC=CC=C1)C(=O)O
InChI: InChI=1S/C10H12O2/c1-10(2,9(11)12)8-6-4-3-5-7-8/h3-7H,1-2H3,(H,11,12)
InChIKey: YYEROYLAYAVZNW-UHFFFAOYSA-N
Product Categories: Acids and Derivatives
2-Methyl-2-phenylpropionic acid Safety Profile
Safety Information of 2-Methyl-2-phenylpropionic acid (CAS NO.826-55-1):
Risk Statements: 36/37/38
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
Safety Statements: 26-36/37/39
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37: Wear suitable gloves
39: Wear eye/face protection
2-Methyl-2-phenylpropionic acid Specification
2-Methyl-2-phenylpropionic acid , its CAS NO. is 826-55-1, the synonym is 2-Phenylisobutyric acid .
Related Products
- 2-Methyl-2-phenylpropionic acid
- 82657-04-3
- 82657-72-5
- 82660-87-5
- 826-62-0
- 82-66-6
- 82668-20-0
- 82671-02-1
- 82671-03-2
- 82671-06-5
- 826-73-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View