-
Name
2-Phenylimidazole
- EINECS 211-581-7
- CAS No. 670-96-2
- Article Data96
- CAS DataBase
- Density 1.141 g/cm3
- Solubility
- Melting Point 142-148 °C(lit.)
- Formula C9H8N2
- Boiling Point 340 °C at 760 mmHg
- Molecular Weight 144.176
- Flash Point 180.4 °C
- Transport Information
- Appearance colorless to beige chunks or granular powder
- Safety 26-36-36/37
- Risk Codes 36/37/38-21/22
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi,  Xn
Xn
- Synonyms 2-phenylimidazole isocyanuric acid adduct;1H-Imidazole, 2-phenyl-;Imidazole, 2-phenyl-;Curezol 2PZ;2-Phenyl-1H-imidazole;
- PSA 28.68000
- LogP 2.07670
Synthetic route

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With caesium carbonate In tetrahydrofuran; methanol at 22℃; for 0.5h; | 100% |
| With lithium hydroxide; mercaptoacetic acid In N,N-dimethyl-formamide at 20℃; for 2.5h; | 88% |
| With mercaptoacetic acid; lithium hydroxide In N,N-dimethyl-formamide at 20℃; Inert atmosphere; | 88% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 80% |
| With cetyltrimethylammonim bromide; potassium hydroxide In tetrahydrofuran; water for 7h; Reflux; Green chemistry; |

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With dimethylamine borane; potassium carbonate; triphenylphosphine; bis(triphenylphosphine)nickel(II) chloride In acetonitrile at 40℃; for 7h; Product distribution; Further Variations:; Catalysts; reaction times; | 97% |

| Conditions | Yield |
|---|---|
| With barium permanganate In dichloromethane for 24h; Heating; | 96% |
| With potassium permanganate; montmorillonite K-10 In acetonitrile at 20℃; for 1h; | 95% |
| With 1 wt percent Rh0 photodeposited TiO2 nanoparticles In isopropyl alcohol at 20℃; for 48h; Irradiation; Inert atmosphere; Sealed tube; | 94% |

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With dimethylamine borane; caesium carbonate; triphenylphosphine; bis(triphenylphosphine)nickel(II) chloride In acetonitrile at 40℃; | 96% |
-
-
37734-89-7
1-benzyl-2-phenyl-1H-imidazole

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; oxygen; dimethyl sulfoxide at 25℃; for 0.25h; | 94% |
| With triethylsilane; palladium 10% on activated carbon In tetrahydrofuran at 20℃; for 24h; Inert atmosphere; | 45% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium carbonate In water at 60℃; for 8h; Suzuki coupling; | 92% |
| With potassium carbonate In ethanol; water at 50℃; for 4h; Suzuki-Miyaura coupling reaction; Inert atmosphere; | 90% |
| With tetrabutylammomium bromide; potassium carbonate In water at 60℃; for 6h; Suzuki coupling; | 90% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 60℃; for 3h; | 68% |

| Conditions | Yield |
|---|---|
| With H6[PMo9V3O40] In acetonitrile at 90℃; for 2h; | 90% |

| Conditions | Yield |
|---|---|
| With tribromide-modified silica-coated magnetic nanoparticles (Fe3O4/SiO2/(CH2)3N+Me3Br3) In neat (no solvent) at 80℃; for 0.25h; Green chemistry; | 90% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; palladium diacetate In N,N-dimethyl-formamide at 140℃; for 48h; | 89% |
| With copper(l) iodide; magnesium oxide; triphenylphosphine; palladium diacetate In 1,4-dioxane at 150℃; for 12h; | 83% |
| With copper(l) iodide; magnesium oxide; triphenylphosphine; palladium diacetate In 1,4-dioxane at 150℃; for 12h; | 83% |
-
-
1219080-61-1
(1H-imidazol-2-yl)boronic acid

-
-
80-17-1
benzenesufonyl hydrazide

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In tert-butyl methyl ether at 60℃; for 6h; Suzuki Coupling; | 83% |
-
-
922-69-0
1,1-dimethoxyethylene

-
-
50846-98-5
2-Diazo-2H-imidazole

-
A
-
670-96-2
2-Phenylimidazole

-
B
-
105851-05-6
4,4-Dimethoxy-1,4-dihydro-imidazo[2,1-c][1,2,4]triazine

| Conditions | Yield |
|---|---|
| In benzene Ambient temperature; | A n/a B 81% |


| Conditions | Yield |
|---|---|
| With ammonium acetate; triethylammonium acetate In neat (no solvent) at 120℃; for 0.666667h; | 78% |
| With ammonium acetate; lipase In water at 45℃; for 1h; Solvent; Time; Enzymatic reaction; | 70% |
| With ammonium hydroxide In ethanol; water at 0℃; for 0.5h; | 38% |
-
-
16054-55-0
1-tosyl-2-phenyl-Δ2-imidazoline

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| Stage #1: 1-tosyl-2-phenyl-Δ2-imidazoline In tetrahydrofuran at -78℃; for 2h; Stage #2: With oxygen In tetrahydrofuran at 20℃; | 68% |
-
-
613-92-3
N-hydroxybenzenecarboximidamide

-
-
1066-54-2
trimethylsilylacetylene

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With caesium carbonate In dimethyl sulfoxide at 100℃; for 24h; Sealed tube; regioselective reaction; | 67% |

| Conditions | Yield |
|---|---|
| Irradiation; | 65% |
-
-
79866-76-5
5-phenyl-1-vinyl-1H-tetrazole

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| In ethanol; trifluoroacetic acid for 2.5h; Irradiation; | 61% |
-
-
79866-76-5
5-phenyl-1-vinyl-1H-tetrazole

-
A
-
670-96-2
2-Phenylimidazole

-
B
-
51687-24-2
5,6-dihydro-tetrazolo[5,1-a]isoquinoline

| Conditions | Yield |
|---|---|
| In ethanol; trifluoroacetic acid Irradiation; | A 61% B n/a |

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With 3-methoxypropylamine In tetrahydrofuran at 50℃; Sealed tube; | 55% |

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| In decalin at 195℃; for 2h; | 54% |

| Conditions | Yield |
|---|---|
| Stage #1: aniline With hydrogenchloride; sodium nitrite In water at 0 - 10℃; Stage #2: 1H-imidazole With sodium acetate at 5 - 20℃; | 47.07% |
| Stage #1: aniline With hydrogenchloride; sodium azide In water at 0 - 10℃; Stage #2: 1H-imidazole With sodium acetate In water at 5 - 20℃; for 51h; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In neat (no solvent) at 110℃; for 10h; | 44% |

| Conditions | Yield |
|---|---|
| With 2-methyl-propan-1-ol; methanesulfonato(2-dicyclohexylphosphino -2’,6’-di-i-propoxy-1,1‘-biphenyl)(2’-amino-1,1’-biphenyl-2-yl)palladium(II); sodium tert-pentoxide In toluene at 80℃; for 96h; Sealed tube; Inert atmosphere; | 41% |
-
-
131543-46-9
Glyoxal

-
-
56613-81-1
(S)-2-Amino-1-phenyl-1-ethanol

-
-
100-52-7
benzaldehyde

-
C
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With ammonium acetate In methanol; water for 5h; Heating; | A n/a B 40% C n/a |


-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With hydrazine In 1,4-dioxane; water for 0.166667h; Inert atmosphere; UV-irradiation; | 10% |

| Conditions | Yield |
|---|---|
| With copper(I)-thiophene-2-carboxylate; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 100℃; under 1500.15 - 2250.23 Torr; for 1h; microwave irradiation; | 8% |
| With copper(I) thiophene-2-carboxylate; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 100℃; for 2h; microwave irradiation; | 8% |
-
-
7431-45-0
2-phenylpyrimidine

-
A
-
670-96-2
2-Phenylimidazole

-
B
-
78009-13-9
2-phenylpyrimidine N-oxide

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; acetic acid at 70℃; for 6h; | A 6% B 5% C 2% |

-
-
23012-16-0
2-phenyl-1,3-oxazole-4-carboxylic acid

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 200℃; |

| Conditions | Yield |
|---|---|
| bei der Destillation; |
-
-
34039-84-4
α-(benzylideneamino)acetonitrile

-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| at 0℃; |

| Conditions | Yield |
|---|---|
| With acetic acid anschliessendes Erwaermen mit wss. HCl; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenylimidazole With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; Stage #2: ethyl bromide In tetrahydrofuran at 25℃; for 12h; | 100% |
| Stage #1: 2-Phenylimidazole With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.333333h; Inert atmosphere; Stage #2: ethyl bromide In N,N-dimethyl-formamide at 25℃; for 12h; Inert atmosphere; | 100% |
| Stage #1: 2-Phenylimidazole With sodium hydride In N,N-dimethyl acetamide for 0.25h; Green chemistry; Stage #2: ethyl bromide In N,N-dimethyl acetamide for 12h; Green chemistry; | 98% |
| With sodium ethanolate In ethanol for 4h; Heating; | 66% |
-
-
670-96-2
2-Phenylimidazole

-
-
72593-77-2
3,6,9-trioxadecyl bromide

| Conditions | Yield |
|---|---|
| With potassium hydroxide In acetonitrile at 20℃; for 24h; | 100% |
-
-
670-96-2
2-Phenylimidazole

-
-
5332-06-9
4-bromobutanenitrile

-
-
198549-12-1
4-(2-phenyl-1H-imidazol-1-yl)butanenitrile

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide | 99% |
| Stage #1: 2-Phenylimidazole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.25h; Stage #2: 4-bromobutanenitrile In N,N-dimethyl-formamide at 20℃; | 99% |
| Stage #1: 2-Phenylimidazole With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; Stage #2: 4-bromobutanenitrile In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; |
-
-
670-96-2
2-Phenylimidazole

-
-
5414-21-1
5-bromopentan-1-nitrile

-
-
791856-78-5
5-(2-phenylimidazol-1-yl)valeronitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenylimidazole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.25h; Stage #2: 5-bromopentan-1-nitrile In N,N-dimethyl-formamide at 20℃; | 99% |
-
-
670-96-2
2-Phenylimidazole

-
-
56338-00-2
4,5-dibromo-2-phenyl-1H-imidazole

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In tetrahydrofuran at 0 - 20℃; for 1h; | 99% |
| With bromine; potassium hydrogencarbonate; N,N-dimethyl-formamide In acetate buffer at 0 - 100℃; for 3h; | 76% |
| With N-Bromosuccinimide In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; Darkness; | 72% |
-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tris(triphenylphosphine)ruthenium(II) chloride; potassium deuterohydroxide; water-d2 In 1,4-dioxane at 80℃; for 16h; Inert atmosphere; Schlenk technique; chemoselective reaction; | 99% |
| With deuterium In tetrahydrofuran under 1500.15 Torr; for 12h; Glovebox; Heating; | 30% |
-
-
670-96-2
2-Phenylimidazole

-
-
292638-85-8
acrylic acid methyl ester

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 80℃; Michael Addition; Schlenk technique; Sealed tube; | 99% |
-
-
670-96-2
2-Phenylimidazole

-
-
1679-47-6
3-methyltetrahydro-2-furanone

-
-
198549-11-0
2-methyl-4-(2-phenylimidazol-1-yl)butanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenylimidazole With potassium hydride Stage #2: 3-methyltetrahydro-2-furanone at 160 - 170℃; for 3h; | 98% |
| With potassium hydride | 80% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra-(n-butyl)ammonium iodide In toluene at 20℃; for 0.25h; | 97% |
| With potassium hydroxide In 1,2-dimethoxyethane at 8℃; for 0.5h; | 90% |
| Stage #1: 2-Phenylimidazole With n-butyllithium In tetrahydrofuran at 0℃; for 0.5h; Cooling with ice; Stage #2: methyl iodide In tetrahydrofuran at 20℃; for 3h; | 69% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Phenylimidazole With tert-butyl methyl ether; nitric acid In tetrahydrofuran at 0℃; Stage #2: With sulfuric acid In dichloromethane at 0℃; for 1h; Further stages.; | 95% |
| With sodium nitrate In neat (no solvent) at 20℃; for 0.5h; Green chemistry; regiospecific reaction; | 92% |
| With sodium nitrate; sulfuric acid at 95℃; for 0.5h; | 63% |

| Conditions | Yield |
|---|---|
| With potassium hydride | 95% |
| Stage #1: 2-Phenylimidazole With potassium hydride Stage #2: 4-butanolide at 160 - 170℃; for 3h; | 95% |
-
-
670-96-2
2-Phenylimidazole

-
-
616-38-6
carbonic acid dimethyl ester

-
-
3475-07-8
N-methyl-2-phenyl-1H-imidazole

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium carbonate at 100℃; for 10h; | 94% |
| With dmap In N,N-dimethyl-formamide at 80℃; for 6h; | 70% |
| at 170℃; for 9h; | 38% |
-
-
670-96-2
2-Phenylimidazole

| Conditions | Yield |
|---|---|
| With triphenylphosphine(1,5-cyclooctadiene)[1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene]iridium(I) hexafluorophosphate; deuterium In dichloromethane at 25℃; under 760.051 Torr; for 1h; Reagent/catalyst; Sealed tube; | 94% |
| With C34H43IrN3(1+)*F6P(1-); deuterium In dichloromethane at -78℃; for 16h; Reagent/catalyst; Inert atmosphere; | 90% |
| With copper(l) iodide; tris(triphenylphosphine)ruthenium(II) chloride; water-d2; zinc In 1,4-dioxane at 80℃; for 16h; Inert atmosphere; Schlenk technique; chemoselective reaction; | 41% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In N,N-dimethyl-formamide at 20 - 140℃; for 24h; Inert atmosphere; Schlenk technique; regioselective reaction; | 94% |
| With palladium diacetate; potassium carbonate; tricyclohexylphosphine In N,N-dimethyl acetamide at 170℃; for 9h; Inert atmosphere; | 55 %Chromat. |
| With potassium phosphate; tetrakis(triphenylphosphine) palladium(0); 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In N,N-dimethyl-formamide |
-
-
670-96-2
2-Phenylimidazole

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
3475-07-8
N-methyl-2-phenyl-1H-imidazole

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 80℃; for 2h; Temperature; | 93.1% |
-
-
670-96-2
2-Phenylimidazole

-
-
683-18-1
dibutyltin chloride

-
-
220335-28-4
dichlorodibutylbis(2-phenylimidazole)tin(IV)

| Conditions | Yield |
|---|---|
| In diethyl ether; ethanol N2-atmosphere; stirring (room temp., 6 h); filtering, washing (Et2O), drying (vac., 20°C, 0.1 Torr); elem. anal.; | 93% |
-
-
670-96-2
2-Phenylimidazole

-
-
1040403-31-3
dichlorobis(2-phenyl-1H-imidazole)palladium(II)

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide to a suspn. of PdCl2 in DMF was added 2-phenylimidazole under Ar, the mixt. was stirred at 50°C for 2 h; the soln. was diluted with DMF, toluene was added, ppt. was filtered, washed with toluene and dried in air; elem. anal.; | 93% |
-
-
670-96-2
2-Phenylimidazole

-
-
501-65-5
diphenyl acetylene

-
-
38147-72-7
5,6-diphenylimidazo[2,1-a]isoquinoline

| Conditions | Yield |
|---|---|
| With {(η5-pentamethylcyclopentadienyl)cobalt(I)2}2; silver(I) acetate; sodium acetate In 2,2,2-trifluoroethanol at 60℃; for 12h; Catalytic behavior; Reagent/catalyst; Temperature; Inert atmosphere; | 93% |
| With (p-cymene)ruthenium(II) chloride; p-benzoquinone In N,N-dimethyl-formamide at 130℃; for 48h; Reagent/catalyst; Inert atmosphere; | 88% |
| With potassium tert-butylate; nickel diacetate; 4,4'-di-tert-butyl-2,2'-bipyridine In toluene at 160℃; for 20h; | 81% |
| With 1,2,3,4-tetraphenylcyclopentadiene; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper(II) acetate monohydrate In N,N-dimethyl-formamide at 80℃; for 3h; | 76% |
| With dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper diacetate; 2,3,4,5-tetraphenylcyclopenta-2,4-dienone In N,N-dimethyl-formamide at 80℃; for 20h; | 63% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; caesium carbonate In N,N-dimethyl-formamide at 150℃; Inert atmosphere; | 92% |
| With copper(I) oxide; 4,7-dimethoxy-1,10-phenanthroline; caesium carbonate; ethylene glycol; poly(ethylene glycol) In various solvent(s) at 120℃; for 72h; | 90% |
| With 4,7-dimethoxy-1,10-phenanthroline; caesium carbonate; ethylene glycol; poly(ethylene glycol); copper(I) oxide In various solvent(s) at 120℃; for 72h; | 90% |
-
-
670-96-2
2-Phenylimidazole

-
-
61698-32-6
2-phenyl-4,5-bis(hydroxymethyl)-imidazole

| Conditions | Yield |
|---|---|
| With potassium hydroxide; paraformaldehyde In ethanol | 92% |

| Conditions | Yield |
|---|---|
| In water aq. solns. of educts mixed under stirring; filtered, washed with water, EtOH and Et2O; elem. anal.; | 92% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In N,N-dimethyl-formamide at 25℃; | 90% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water | 90% |
| With potassium hydroxide In water | 86% |
| With sodium hydroxide In water | 86% |
-
-
670-96-2
2-Phenylimidazole

-
-
1124-19-2
phenyltin trichloride

-
-
220335-31-9
trichlorobutylbis(2-phenylimidazole)tin(IV)

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; stirring (room temp., 24 h); evapn., pptn. on Et2O addn., filtering, washing (Et2O), drying (vac., 20°C, 0.1 Torr); elem. anal.; | 90% |
2-Phenylimidazole Specification
The IUPAC name of 2-Phenylimidazole is 2-phenyl-1H-imidazole. With the CAS registry number 670-96-2 and EINECS 211-581-7, it is also named as 1H-Imidazole, 2-phenyl-. The product's categories are Industrial / Fine Chemicals; Imidazoles; Imidaxoles; Building Blocks; Heterocyclic Building Blocks. It is colorless to beige chunks or granular powder which should be sealed in the container and stored in the cool and dry place which must be away from oxidant.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 1.88; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.29; (4)ACD/LogD (pH 7.4): 1.69; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 10.23; (7)ACD/KOC (pH 5.5): 6.41; (8)ACD/KOC (pH 7.4): 162.43; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 17.82 Å2; (13)Index of Refraction: 1.602; (14)Molar Refractivity: 43.36 cm3; (15)Molar Volume: 126.2 cm3; (16)Polarizability: 17.19×10-24 cm3; (17)Surface Tension: 49.1 dyne/cm; (18)Density: 1.141 g/cm3; (19)Flash Point: 180.4 °C; (20)Enthalpy of Vaporization: 56.04 kJ/mol; (21)Boiling Point: 340 °C at 760 mmHg; (22)Vapour Pressure: 0.000174 mmHg at 25°C.
Preparation of 2-Phenylimidazole: It can be obtained by benzene and 2-diazo-2H-imidazole. This reaction needs irradiation. The yield is 65%.
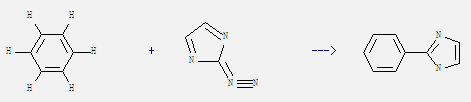
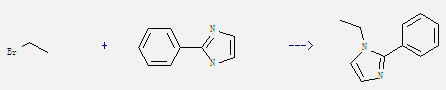
Uses of 2-Phenylimidazole: It can be used as curing agent of epoxy, polyurethane and as intermediate of pharmaceutical, pesticide and dye. What's more, it can react with bromoethane to get 1-ethyl-2-phenyl-1H-imidazole. This reaction needs reagent NaOC2H5 and solvent ethanol by heating. The reaction time is 4 hours. The yield is 66%.
When you are using this chemical, please be cautious about it as the following:
It is not only harmful in contact with skin and if swallowed, but also irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing and gloves.
People can use the following data to convert to the molecule structure.
1. SMILES:n1ccnc1c2ccccc2
2. InChI:InChI=1/C9H8N2/c1-2-4-8(5-3-1)9-10-6-7-11-9/h1-7H,(H,10,11)
3. InChIKey:ZCUJYXPAKHMBAZ-UHFFFAOYAE
Related Products
- 2-Phenylimidazole
- 670-98-4
- 67098-56-0
- 67098-97-9
- 67103-05-3
- 67104-97-6
- 67105-55-9
- 67106-22-3
- 6710-83-4
- 67108-80-9
- 67109-90-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View