-
Name
2-Thiopheneacetyl chloride
- EINECS 254-290-0
- CAS No. 39098-97-0
- Article Data43
- CAS DataBase
- Density 1.335 g/cm3
- Solubility Reacts with water.
- Melting Point
- Formula C6H5ClOS
- Boiling Point 202.4 °C at 760 mmHg
- Molecular Weight 160.624
- Flash Point 88.7 °C
- Transport Information UN 3265 8/PG 2
- Appearance clear brown liquid
- Safety 26-28-36/37/39-45-8-30-23
- Risk Codes 14-34-37-29
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms 2-Thienylacetyl chloride;2-thiophen-2-ylacetyl chloride;Thiophene-2-acetyl chloride;2-Thiopheneacetyl chloride (TAC);2-Thiopheneacetylchloride;2-Thiophene acetyl chloride;
- PSA 45.31000
- LogP 2.05600
Synthetic route
-
-
1918-77-0
Thiophene-2-acetic acid

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| With bis(trichloromethyl) carbonate; N,N-dimethyl-formamide In dichloromethane at -10 - 0℃; for 5h; Temperature; Reagent/catalyst; | 94.7% |
| With phosgene; 4-cyclopentylaminopyridine hydrobromide In toluene | 92% |
| With 1-butyl-3-methylimidazolium hydroxide; thionyl chloride In chloroform at 10℃; for 8h; Reagent/catalyst; Temperature; | 89.8% |

| Conditions | Yield |
|---|---|
| With thionyl chloride; triethylamine In chloroform; water; ethyl acetate | A n/a B 84% |

-
-
5271-67-0
2-Thiophenecarbonyl chloride

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: diethyl ether / 0 °C 2: aq. Ag2O / dioxane 3: (COCl)2; DMF / CH2Cl2 View Scheme |
-
-
527-72-0
2-thiophenylcarboxylic acid

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: (COCl)2; DMF / CH2Cl2 2: diethyl ether / 0 °C 3: aq. Ag2O / dioxane 4: (COCl)2; DMF / CH2Cl2 View Scheme |
-
-
72676-21-2
2-diazo-1-(thiophen-2-yl)ethanone

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. Ag2O / dioxane 2: (COCl)2; DMF / CH2Cl2 View Scheme |
-
-
765-50-4
2-Chloromethylthiophene

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: acetone; water 2: aq.-ethanolic KOH-solution 3: thionyl chloride View Scheme | |
| Multi-step reaction with 3 steps 1: acetone; water 2: aq.-ethanolic KOH-solution 3: thionyl chloride View Scheme |
-
-
20893-30-5
2-cyanomethylthiophene

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq.-ethanolic KOH-solution 2: thionyl chloride View Scheme |

| Conditions | Yield |
|---|---|
| With thionyl chloride; triethylamine In dichloromethane | |
| With thionyl chloride; triethylamine In dichloromethane |
-
-
68100-13-0
isopropyl 2-(thiophen-2-yl)acetate

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogenchloride / water; acetonitrile / 10 h / Reflux 2: pyridine; thionyl chloride / dichloromethane / 1 h / Reflux View Scheme |
-
-
57382-97-5
ethyl thiophen-2-ylacetate

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogenchloride / ethanol; water / 8 h / Reflux 2: pyridine; thionyl chloride / dichloromethane / 1 h / Reflux View Scheme |
-
-
19432-68-9
methyl 2-thiopheneacetate

-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogenchloride / water; acetonitrile / 8 h / Reflux 2: pyridine; thionyl chloride / dichloromethane / 1 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| at 20℃; for 4h; | 100% |
| for 0.5h; | 82% |
| at 20℃; for 3h; |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
121-45-9
phosphorous acid trimethyl ester

-
-
87600-60-0
(2-Thiophen-2-yl-acetyl)-phosphonic acid dimethyl ester

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
| at 0 - 20℃; for 3h; | 88% |
| In tetrahydrofuran Heating; | |
| at 0℃; for 1h; Reflux; |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
88-17-5
2-(trifluoromethyl)benzenamine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 100% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
99-09-2
3-nitro-aniline

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 100% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
660831-35-6
2-{[(2-biphenyl-4-yl-(1S)-methoxycarbonyl-ethyl)-(4'-trifluoromethyl-biphenyl-4-carbonyl)-amino]-methyl}-(2R)-pyrrolidinium chloride

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 1h; | 100% |

-
-
39098-97-0
2-thienylacetic acid chloride

-
-
1358786-71-6
C16H14N4OS

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane | 100% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
310454-69-4
ethyl (S)-1-(2-thiopheneacetyl)-3-piperidinecarboxylate

| Conditions | Yield |
|---|---|
| With sodium carbonate In water; ethyl acetate at 20℃; for 0.5h; Schotten-Baumann Reaction; Green chemistry; | 100% |

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 0.5h; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 98% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
895635-70-8
{3-[3-(2-chloro-4-pyrimidinyl)pyrazolo[1,5-a]pyridin-2-yl]phenyl}amine

-
-
941318-38-3
N-{3-[3-(2-chloro-4-pyrimidinyl)pyrazolo[1,5-a]pyridin-2-yl]phenyl}-2-(2-thienyl)acetamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-thienylacetic acid chloride; {3-[3-(2-chloro-4-pyrimidinyl)pyrazolo[1,5-a]pyridin-2-yl]phenyl}amine With triethylamine In tetrahydrofuran Stage #2: With PS-Trisamine In tetrahydrofuran for 16h; | 98% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
107-15-3
ethylenediamine

-
-
1160003-21-3
N,N'-di(2-thiopheneacetyl)ethylenediamine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 97% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
941319-32-0
5-[3-(2-chloro-4-pyrimidinyl)pyrazolo[1,5-a]pyridin-2-yl]-2-methylaniline

-
-
941319-33-1
N-{5-[3-(2-chloro-4-pyrimidinyl)pyrazolo[1,5-a]pyridin-2-yl]-2-methylphenyl}-2-(2-thienyl)acetamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-thienylacetic acid chloride; 5-[3-(2-chloro-4-pyrimidinyl)pyrazolo[1,5-a]pyridin-2-yl]-2-methylaniline With PS-Trisamine In tetrahydrofuran for 16h; Stage #2: With triethylamine In tetrahydrofuran | 97% |
-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide; hydroquinone; ammonium perrhenate In ethyl acetate for 0.5h; Ambient temperature; | 96% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 96% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
62-53-3
aniline

-
-
69047-40-1
N-phenyl-2-(thiophen-2-yl)acetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; | 96% |
-
-
39098-97-0
2-thienylacetic acid chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 23℃; for 2h; Inert atmosphere; | 96% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
19369-53-0
methyl 2-amino-5-methylthiophene-3-carboxylate

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 98℃; | 95.5% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
117362-43-3
9-Methyl-5-thia-6,13-diaza-benzo[4,5]cyclohepta[1,2-b]naphthalene

-
-
132087-68-4
C23H16N2OS2

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 1h; Heating; | 95% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
895637-18-0
3-[3-(2-chloro-4-pyrimidinyl)-4-fluoropyrazolo[1,5-a]pyridin-2-yl]aniline

-
-
941318-80-5
N-{3-[3-(2-chloro-4-pyrimidinyl)-4-fluoropyrazolo[1,5-a]pyridin-2-yl]phenyl}-2-(2-thienyl)acetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.5h; | 95% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
3399-73-3
2-(1-cyclohexenyl)ethylamine

-
-
89413-09-2
N-<2-(cyclohexen-1-yl)ethyl>thiophene-2-acetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In benzene for 1h; Ambient temperature; | 94% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
706791-83-5
3-amino-5-bromobenzoic acid methyl ester

-
-
1188322-86-2
methyl 3-bromo-5-[2-(thiophen-2-yl)acetamido]benzoate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl acetamide at 20℃; Inert atmosphere; Cooling with ice; | 94% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
15401-69-1
berberrubine chloride

-
-
1297302-78-3
C25H20NO5S(1+)*Cl(1-)

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 0.5h; | 94% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
93601-86-6
(S)-(-)-(5-methoxy-1,2,3,4-tetrahydronaphthalen-2-yl)propylamine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: (S)-(-)-(5-methoxy-1,2,3,4-tetrahydronaphthalen-2-yl)propylamine hydrochloride With sodium carbonate In dichloromethane; water at 27℃; for 1h; Stage #2: 2-thienylacetic acid chloride In dichloromethane; water at 27℃; for 1h; | 94% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
112697-91-3
quino<2,3-b><1,5>benzoxazepine

-
-
132087-58-2
C22H14N2O2S

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 1h; Heating; | 93% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
117362-42-2
quino<2,3-b><1,5>benzothiazepine

-
-
132087-65-1
C22H14N2OS2

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 1h; Heating; | 93% |
2-Thiopheneacetyl Chloride Chemical Properties
Molecular Weight:160.62
EINECS:254-290-0
Density:1.303 g/mL at 25 °C(lit.)
Boiling Point:130-135 °C (90 mmHg)
Flash Point:101 °C
Appearance:Clear brown liquid
2-Thiopheneacetyl chloride's(39098-97-0) Synonyms: TIMTEC-BB SBB006705;THIOPHENE-2-ACETYL CHLORIDE;2-Thiopheneacethylchloride;2-(2-THIENYL)ACETYL CHLORIDE;2-THIOPHENEACETYL CHLORIDE;2-THIENYLACETYL CHLORIDE;AKOS BBS-00003919;2-Thienylacetic acid chloride
2-Thiopheneacetyl chloride's(39098-97-0) Molecular Structure:
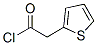
2-Thiopheneacetyl Chloride Uses
2-Thiopheneacetyl Chloride Safety Profile
Hazard Codes C
Risk Statements 14-34-37-29
Safety Statements 26-28-36/37/39-45-8-30-23
RIDADR UN 3265 8/PG 2
WGK Germany 3
F 8-10-21
TSCA T
HazardClass 8
PackingGroup III
HS Code 29349990
2-Thiopheneacetyl Chloride Specification
Accidental Release Measures of 2-Thiopheneacetyl chloride(39098-97-0)
【Small spills/leaks】Clean up spills immediately, using the appropriate protective equipment. Absorb spill using an absorbent, non-combustible material such as earth, sand, or vermiculite. Provide ventilation. Do not get water inside containers.
Stability and Reactivity of 2-Thiopheneacetyl chloride(39098-97-0)
【Stability】Stable under normal temperatures and pressures. May decompose on exposure to moist air or water. Moisture sensitive.
【Incompatibilities】Strong oxidizing agents, strong bases, moisture.
【Decomposition】Hydrogen chloride, carbon monoxide, oxides of sulfur, carbon dioxide.
【Combustion Products】Fire may produce irritating, corrosive and/or toxic gases.
Related Products
- 2-Thiopheneacetyl Chloride
- 39101-54-7
- 3910-35-8
- 39104-30-8
- 39108-47-9
- 39110-92-4
- 391-12-8
- 39115-95-2
- 39115-96-3
- 39118-50-8
- 39120-56-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View