-
Name
3-Acetylpyridine
- EINECS 206-496-7
- CAS No. 350-03-8
- Article Data128
- CAS DataBase
- Density 1.06 g/cm3
- Solubility soluble in hot water
- Melting Point 11-13 °C(lit.)
- Formula C7H7NO
- Boiling Point 220.8 °C at 760 mmHg
- Molecular Weight 121.139
- Flash Point 92.3 °C
- Transport Information UN 2810 6.1/PG 3
- Appearance clear colorless to yellow liquid
- Safety 45-37/39-28A-26-36
- Risk Codes 25-36/38-36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 T,
T,  Xi,
Xi,  Xn
Xn
- Synonyms beta-Acetylpyridine;1-pyridin-3-ylethanone;.beta.-Acetylpyridine;Pyridine, 3-acetyl-;Ethanone, 1-(3-pyridinyl)-;Methyl pyridyl ketone;1-(3-Pyridenyl)ethanone;Ketone, methyl 3-pyridyl;Methyl 3-pyridyl ketone;3-Pyridyl methyl ketone;FEMA No. 3424;3-acetyl pyridine;
- PSA 29.96000
- LogP 1.28420
Synthetic route
-
-
14188-94-4
3-acetylpyridine N-oxide

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With (4,4′-di-tert-butyl-2,2′-bipyridine)bis[(2-pyridinyl)phenyl]iridium(III) hexafluorophosphate; di-tert-butyl 1,4-dihydro-2,6-dimethyl-3,5-pyridine-dicarboxylate In acetonitrile at 20℃; for 0.5h; Inert atmosphere; Irradiation; chemoselective reaction; | 98% |
| With bis(tetra-n-butylammonium) tetrakis(benzenethiolato-μ3-sulfidoiron); thiophenol In methanol; acetonitrile at 20℃; for 20h; Product distribution; | 86% |
| With ammonium formate; nickel In methanol at 40℃; for 2h; | 69% |
| With ammonium formate; zinc In methanol for 6h; Heating; | 65% |
| With benzyl alcohol at 120℃; for 6h; Inert atmosphere; Schlenk technique; chemoselective reaction; | 92 %Spectr. |

| Conditions | Yield |
|---|---|
| With sulfuric acid; dihydrogen peroxide; sodium bromide In 1,4-dioxane; water at 70℃; Flow reactor; Green chemistry; | 97% |
| With copper(l) iodide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium tert-butylate; L-proline In N,N-dimethyl-formamide at 25℃; for 5h; | 90% |
| With oxygen; Azobenzene; sodium bromide In 1,4-dioxane at 80℃; for 36h; | 90% |
-
-
55676-25-0
3-(2-methyl-[1,3]dioxolan-2-yl)-pyridine

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With O-phenyl phosphorodichloridate; sodium iodide In benzene for 20h; Heating; | 96% |
-
-
52155-97-2
3-(α,α-ethyleenedithioethyl)pyridine

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With bismuth(III) chloride; benzyltriphenylphosphonium peroxymonosulfate In acetonitrile for 2h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In ethylbenzene; water at 135℃; for 24h; Concentration; Temperature; Time; Sealed tube; | 94.8% |
| With N-hydroxyphthalimide; oxygen; cobalt(II) diacetate tetrahydrate In acetic acid butyl ester at 90℃; under 760.051 Torr; for 12h; Solvent; Reagent/catalyst; Temperature; | 84% |
| With nickel-doped graphene carbon nitride nanoparticles; air In ethanol at 25℃; for 8h; Irradiation; Green chemistry; | 83% |

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In water; acetonitrile at 20℃; for 15h; Oxidation; | 94% |
-
-
32064-90-7
(1E,2E)-1,2-bis(1-(pyridin-3-yl)ethylidene)hydrazine

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With 2-phenyl-1,2-benzoisoselenazol-3(2H)-one; dihydrogen peroxide In methanol; water at 65℃; for 72h; | 93% |

| Conditions | Yield |
|---|---|
| With tetrachlorosilane; silica gel In hexane; water for 0.75h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromopyridine With diisobutylaluminium hydride; magnesium; lithium chloride In tetrahydrofuran at 25℃; for 0.5h; Stage #2: acetyl chloride With copper(l) cyanide; lithium chloride In tetrahydrofuran at 25℃; for 1h; | 90% |

| Conditions | Yield |
|---|---|
| With tetrachlorosilane; silica gel In hexane; water for 0.25h; Heating; | 85% |

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetic acid In water for 3h; Reagent/catalyst; Reflux; | 81.2% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromopyridine; -butyl vinyl ether With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In various solvent(s) at 125℃; for 30h; Regioselective Heck arylation; Stage #2: With hydrogenchloride In various solvent(s) for 1h; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromopyridine; ethylene glycol divinyl ether With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In various solvent(s) at 125℃; for 30h; Regioselective Heck arylation; Stage #2: With hydrogenchloride In various solvent(s) for 1h; | 77% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; water; mercury(II) sulfate In acetone for 2h; Heating; | 72% |
| With sulfuric acid In benzene at 80℃; |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromopyridine; ethyl vinyl ether With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In various solvent(s) at 125℃; for 30h; Regioselective Heck arylation; Stage #2: With hydrogenchloride In various solvent(s) for 1h; | 72% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water; acetonitrile at 55℃; for 12h; Wacker Oxidation; | 72% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromopyridine; ethylhexyl vinyl ether With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In various solvent(s) at 125℃; for 30h; Regioselective Heck arylation; Stage #2: With hydrogenchloride In various solvent(s) for 1h; | 71% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Chloropyridine; -butyl vinyl ether With 1,3-bis-(diphenylphosphino)propane; triethylamine; palladium diacetate In various solvent(s) at 125℃; for 30h; Regioselective Heck arylation; Stage #2: With hydrogenchloride In various solvent(s) for 1h; | 69% |
-
-
139042-59-4
1-(6-bromopyridin-3-yl)ethanone

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; water; cobalt(II) acetate; triphenylphosphine; sodium hydroxide; silicon at 100℃; for 24h; | 69% |

| Conditions | Yield |
|---|---|
| With water; oxygen; zinc(II) oxide In acetonitrile at 20℃; Irradiation; | 67% |
-
-
30510-18-0
nicotinoylacetonitrile

-
A
-
350-03-8
methyl-3-pyridylketone

-
B
-
152171-44-3
3-oxo-3-(3-pyridyl)propanamide

| Conditions | Yield |
|---|---|
| With Rhodococcus rhodochrous IFO 15564 In phosphate buffer; ethanol at 28℃; for 9.5h; pH=8.0; | A n/a B 60% |
-
-
106353-54-2
3,3-Di-morpholin-4-yl-1-pyridin-3-yl-propenone

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ethanol for 24h; Heating; | 57% |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; silver trifluoromethanesulfonate In toluene at 80℃; for 6h; Sealed tube; regioselective reaction; | 56% |
-
-
106881-77-0
(E)-1-(pyridin-3-yl)ethanone oxime

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene In dichloromethane for 0.5h; Ambient temperature; | 53% |
-
-
614-18-6
3-pyridinecarboxylic acid ethyl ester

-
-
141-78-6
ethyl acetate

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| 49% | |
| With aluminum oxide at 399.85℃; | 6 % Chromat. |
-
-
17694-68-7
3-(2-bromoacetyl)pyridine hydrobromide

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; acetic acid for 6.5h; Irradiation; Inert atmosphere; | 31% |

-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With bismuth(III) chloride; benzyltriphenylphosphonium peroxymonosulfate In acetonitrile for 5h; Heating; | 30% |
| With aluminium trichloride; benzyltriphenylphosphonium chlorochromate In acetonitrile for 1h; Heating; | 30% |

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide; sulfuric acid |

| Conditions | Yield |
|---|---|
| With diethyl ether Und anschliessenden Hydrolieseren; | |
| With diethyl ether anschliessend mit Benzol; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| Stage #1: 3-oxo-3-pyridin-3-yl-propionic acid ethyl ester With sodium ethanolate; titanium(IV) oxide In ethyl acetate at 0 - 76℃; for 5h; Stage #2: With hydrogenchloride In water at 0℃; for 5h; Temperature; Reagent/catalyst; Reflux; | 11.5 g |
-
-
110-86-1
pyridine

-
-
350-03-8
methyl-3-pyridylketone

-
-
110514-05-1
1-[2-oxo-2-(pyridin-3-yl)ethyl]pyridinium iodide

| Conditions | Yield |
|---|---|
| With iodine for 3h; Heating; | 100% |
| With iodine at 140℃; for 3h; | 100% |
| With iodine at 140℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With zirconium dioxide hydrate; isopropyl alcohol at 130℃; for 0.416667h; Meerwein-Ponndorf-Verley Reduction; | 100% |
| With C25H30ClIrN2O3; water; potassium hydroxide In isopropyl alcohol for 1h; Reflux; | 99% |
| With LaCu0.67Si1.33; hydrogen In methanol at 120℃; under 22502.3 Torr; for 10h; Autoclave; | 99% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
14188-94-4
3-acetylpyridine N-oxide

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; methyltrioxorhenium(VII) In dichloromethane; water at 15℃; for 5h; | 100% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; Inert atmosphere; | 100% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; for 72h; | 90% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
106881-77-0
(E)-1-(pyridin-3-yl)ethanone oxime

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium acetate In methanol; water Reflux; | 100% |
| With pyridine; hydroxylamine hydrochloride In ethanol for 12h; Reflux; | 94% |
| With hydroxylamine hydrochloride; sodium carbonate In ethanol; water at 55 - 62℃; for 2.66667h; optical yield given as %de; | 93% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
17694-68-7
3-(2-bromoacetyl)pyridine hydrobromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide; bromine; acetic acid In water at 40 - 75℃; for 4h; | 100% |
| With hydrogen bromide; bromine In acetic acid at 0 - 75℃; for 2 - 4h; Product distribution / selectivity; | 100% |
| With hydrogen bromide; bromine In acetic acid at 0 - 75℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; bromine In water; acetic acid at 40 - 75℃; for 2 - 4h; | 100% |

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 99.2% |
| In acetonitrile at 45℃; Kinetics; pyridinolysis; | |
| In ethyl acetate for 24h; Reflux; |
-
-
350-03-8
methyl-3-pyridylketone

-
-
5096-11-7, 10593-35-8, 4754-27-2, 7606-26-0
(R)-1-(3-pyridyl)ethanol

| Conditions | Yield |
|---|---|
| With C50H68FeN4P2(2+)*2BF4(1-); sodium t-butanolate In isopropyl alcohol at 50℃; for 1h; Reagent/catalyst; Temperature; Glovebox; Schlenk technique; enantioselective reaction; | 99.1% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; f-ampha; hydrogen; lithium tert-butoxide In isopropyl alcohol at 25 - 30℃; under 15201 Torr; for 6h; Inert atmosphere; Autoclave; enantioselective reaction; | 99% |
| With potassium tert-butylate; hydrogen; {R-4-(3,5-Xyl)2P-12-Ph2P[2.2]paracyclophane}RuCl2(S,S)-DPEN In isopropyl alcohol at 25℃; under 3750.38 - 7500.75 Torr; for 3h; | 98% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
105-36-2
ethyl bromoacetate

-
-
94795-15-0
3-acetyl-1-(2-ethoxy-2-oxoethyl)pyridin-1-ium bromide

| Conditions | Yield |
|---|---|
| In ethyl acetate Inert atmosphere; Reflux; | 99% |
| With benzene |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 48h; | 99% |
| In nitrobenzene at 35℃; Rate constant; | |
| With acetone |
-
-
872-85-5
pyridine-4-carbaldehyde

-
-
350-03-8
methyl-3-pyridylketone

-
-
13328-57-9
1-(3-Pyridyl)-3-(4-pyridyl)-2-propen-1-on

| Conditions | Yield |
|---|---|
| With base | 99% |
| Stage #1: methyl-3-pyridylketone With potassium hydroxide In methanol; water at 0℃; Claisen Schmidt condensation; Stage #2: pyridine-4-carbaldehyde In methanol; water at 0℃; for 3h; Claisen Schmidt condensation; |
-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| With hydrogen bromide; bromine In water at 55 - 60℃; for 1h; Product distribution / selectivity; | 99% |
-
-
350-03-8
methyl-3-pyridylketone

| Conditions | Yield |
|---|---|
| In chloroform ligand added to a soln. of Rh complex, stirred for 5 min at room temp.; concd. (vac.); elem. anal.; | 99% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
762-04-9
phosphonic acid diethyl ester

-
-
1370037-09-4
diethyl 1-hydroxy-1-(pyridin-3-yl)phenylethylphosphonate

| Conditions | Yield |
|---|---|
| With {(μ-η5:η1):η1-2-[(2,6-Me2C6H3)NCH2](C4H3N)YN(SiMe3)2}2 at 20℃; for 0.333333h; Inert atmosphere; neat (no solvent); | 99% |
| With n-butyllithium In neat (no solvent) at 10℃; for 0.0833333h; Catalytic behavior; Schlenk technique; Inert atmosphere; | 96% |
| With (2,6-iPr2PhNH)5SmLi2(THF)2 In hexane at 20℃; for 0.333333h; Schlenk technique; Inert atmosphere; | 94% |
| With Y[N(SiMe3)2](κ2-C6H5C(O)NC6H3(iPr)2)2(THF) In neat (no solvent) at 25℃; for 0.666667h; Pudovik Reaction; Schlenk technique; Inert atmosphere; Glovebox; Green chemistry; | 87% |
| With {[(Me3Si)2N]2Sm[(iPrN)2CN(CH2)2]}2 In neat (no solvent) at 10℃; for 0.333333h; Inert atmosphere; Schlenk technique; | 87% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
35691-93-1
3,5-Dimethyl-1H-pyrazole-4-carboxylic acid ethyl ester

-
-
1590405-71-2
ethyl 2,5-dimethylpyrazolo[1,5-a][1,8]naphthyridine-3-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 120℃; for 16h; | 99% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
20099-89-2
4-Cyanophenacyl bromide

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 98.9% |
| In ethyl acetate for 24h; Reflux; |
-
-
350-03-8
methyl-3-pyridylketone

-
-
99-73-0
para-bromophenacyl bromide

-
-
136106-09-7
3-acetyl-1-(2-(4-bromophenyl)-2-oxoethyl)pyridinium bromide

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 98.5% |
| In methanol at 40.1℃; under 750060 Torr; Rate constant; other pressures; | |
| In methanol at 40.1℃; under 750060 Torr; |

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 98.5% |
| In acetonitrile at 45℃; Kinetics; pyridinolysis; |
-
-
350-03-8
methyl-3-pyridylketone

-
-
29971-54-8, 71351-55-8
ethyl 3-(3-pyridyl)but-2-enoate

| Conditions | Yield |
|---|---|
| 98% |

| Conditions | Yield |
|---|---|
| With triiron dodecarbonyl; C52H58N4P2; hydrogen; potassium hydroxide In methanol at 45℃; under 37503.8 Torr; for 5h; enantioselective reaction; | 98% |
| With (S)-(-)-α,α-bis[3,5-bis(trifluoromethyl)phenyl]-2-pyrrolidinemethanol; Trimethyl borate; dimethylsulfide borane complex In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 96% |
| With potassium tert-butylate; hydrogen; trans-RuCl2[(R)-xylbinap][(R)-daipen] In isopropyl alcohol; tert-butyl alcohol at 25℃; under 6080.41 Torr; for 12h; Catalytic hydrogenation; | 95% |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride In methanol for 5h; Reflux; | 98% |
| Stage #1: methyl-3-pyridylketone With hydroxylamine hydrochloride In methanol for 1h; Reflux; Stage #2: With sodium acetate In methanol for 1h; Solvent; Temperature; Time; Reflux; | 95% |
| With hydroxylamine hydrochloride In ethanol; water at 80℃; Reflux; | 94% |

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 98% |
| In acetonitrile at 45℃; Kinetics; pyridinolysis; | |
| In ethyl acetate for 24h; Reflux; |

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 98% |
| In acetonitrile at 45℃; Kinetics; pyridinolysis; |
-
-
350-03-8
methyl-3-pyridylketone

-
-
18871-66-4
N,N-dimethylacetamide dimethyl acetal

-
-
287494-18-2
3-(Dimethylamino)-1-(3-pyridinyl)-2-buten-1-one

| Conditions | Yield |
|---|---|
| at 110℃; for 5h; Condensation; | 98% |
| In hexane |
-
-
350-03-8
methyl-3-pyridylketone

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
75415-01-9
3-(dimethylamino)-1-(3-pyridinyl)-2-propen-1-one

| Conditions | Yield |
|---|---|
| at 110℃; for 5h; Condensation; | 98% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
72824-04-5
2-Allyl-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With (PPh3)3CuF In tetrahydrofuran at 20℃; for 4h; | 98% |
| With indium In water at 30℃; for 24h; Inert atmosphere; | 87% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; potassium tert-butylate; boric acid In tetrahydrofuran at 23℃; for 3h; Inert atmosphere; | 51% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; potassium tert-butylate; boric acid In tetrahydrofuran at 22℃; for 3h; Inert atmosphere; | 51% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
616-38-6
carbonic acid dimethyl ester

-
-
54950-20-8
methyl nicotinoylacetate

| Conditions | Yield |
|---|---|
| With sodium hydride In toluene for 4h; Inert atmosphere; Reflux; | 98% |
| Stage #1: methyl-3-pyridylketone With potassium hexamethylsilazane In tetrahydrofuran; toluene at -78 - 20℃; for 3h; Stage #2: carbonic acid dimethyl ester In tetrahydrofuran; toluene at -78 - 20℃; Acidic aqueous solution; | 68% |
| With sodium hydride In toluene; mineral oil at 105℃; for 12h; Inert atmosphere; | 60.8% |
-
-
350-03-8
methyl-3-pyridylketone

-
-
19501-58-7
4-methoxyphenylhydrazine hydrochloride

-
-
1258795-08-2
C14H15N3O

| Conditions | Yield |
|---|---|
| With sodium acetate In ethanol at 80℃; for 0.0833333h; Microwave irradiation; air-cooling; | 98% |

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 5h; Green chemistry; | 97.9% |
3-Acetylpyridine Consensus Reports
3-Acetylpyridine Standards and Recommendations
3-Acetylpyridine Specification
The 3-Acetylpyridine, with its cas register number 350-03-8, has its IUPAC name of 1-pyridin-3-ylethanone. And this chemical is a kind of clear colorless liquid, and it is soluble in hot water, ethonal, and ethyl ether. Besides, its product categories are including acetylgroup;carbonyl compounds; heterocycles; pyridines, pyrimidines, purines and pteredines; pyridines derivates; pyridine flavor; osteoporosis. This chemical is usually used as the intermediate of risedronate sodium which could cure osteoporosis.
The characteristics of this chemical are as following: (1)XLogP3: 0.4; (2)H-Bond Donor: 0; (3)H-Bond Acceptor: 2; (4)Rotatable Bond Count: 1; (5)Tautomer Count: 2; (6)Exact Mass: 121.052764; (7)MonoIsotopic Mass: 121.052764; (8)Topological Polar Surface Area: 30; (9)Heavy Atom Count: 9; (10)Complexity: 112; (11)Covalently-Bonded Unit Count: 1.
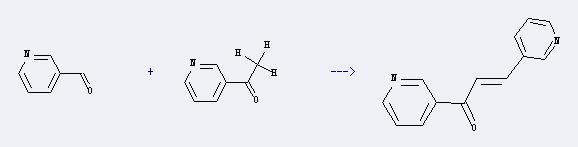
Uses of 3-Acetylpyridine: 3-Acetylpyridine could react with pyridine-3-carbaldehyde to produce 1,3-di-pyridin-3-yl-propenone. The reaction condition is below: reagent: K2CO3; solvent: diethyl ether; reaction temp.: 0-20℃; field: 90%.
Producing method of 3-Acetylpyridine: 3-Ethynyl-pyridine could react in the following condtion to produce 3-Acetylpyridine: reagent: H2O, HgSO4, conc. H2SO4; solvent: acetone; reaction time: 2 hours; other condition: heating; field: 72%.

When you are dealing with this chemical, you should be very cautious. This is a kind of toxic chemical, and it could at low levels cause damage to health. If by inhalation, in contact with skin or if swallowed, it will have harmful damage to our health. Besides, it is irritant to eyes, respiratory system and skin, and it may also may cause inflammation to the skin or other mucous membranes. So we had better take the following instructions. Wear suitable gloves and eye/face protection, and if In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And if in case of accident or if you feel unwell, seek medical advice immediately (show the label where possible). Besides, after contacting with skin, wash immediately with plenty of ... (to be specified by the manufacturer). When store it, keep it in the dry, cool, and well-ventilated storeroom.
Additionally, you could obtain the molecular structure through converting the following datas:
Canonical SMILES: CC(=O)C1=CN=CC=C1
InChI: InChI=1S/C7H7NO/c1-6(9)7-3-2-4-8-5-7/h2-5H,1H3
InChIKey: WEGYGNROSJDEIW-UHFFFAOYSA-N
Below are the toxicity information of this chemical:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| bird - wild | LD50 | oral | 178mg/kg (178mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 12, Pg. 355, 1983. | |
| mouse | LD50 | intraperitoneal | 182mg/kg (182mg/kg) | Journal of Pharmaceutical Sciences. Vol. 64, Pg. 528, 1975. | |
| quail | LD50 | oral | 422mg/kg (422mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 12, Pg. 355, 1983. | |
| rat | LD50 | oral | 46uL/kg (0.046mL/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD MUSCULOSKELETAL: OTHER CHANGES | Acute Toxicity Data. Journal of the American College of Toxicology, Part B. Vol. 1, Pg. 681, 1992. |
Related Products
- 3-Acetylpyridine
- 350038-30-1
- 35006-34-9
- 35-00-7
- 35008-50-5
- 35010-96-9
- 35011-47-3
- 35013-72-0
- 35016-63-8
- 35018-15-6
- 35018-62-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View