-
Name
3-Chloroaniline
- EINECS 203-581-0
- CAS No. 108-42-9
- Article Data289
- CAS DataBase
- Density 1.216
- Solubility Water solubility: 6.8 g/L (20 °C)
- Melting Point -11--9 °C(lit.)
- Formula C6H6ClN
- Boiling Point 227.8 °C at 760 mmHg
- Molecular Weight 127.573
- Flash Point 123.9 °C
- Transport Information UN 2019 6.1/PG 2
- Appearance clear pale yellow to brown liquid
- Safety 28-36/37-45-60-61-28A
- Risk Codes 23/24/25-33-50/53
-
Molecular Structure
-
Hazard Symbols
 T,
T, N
N
- Synonyms Aniline,m-chloro- (8CI);1-Amino-3-chlorobenzene;3-Amino-1-chlorobenzene;3-Chloro-1-aminobenzene;3-Chlorobenzenamine;3-Chlorophenylamine;5-Chloroaniline;Fast Orange GC Base;NSC 17581;Orange GC base;m-Aminochlorobenzene;m-Chloroaniline;m-Chlorophenylamine;
- PSA 26.02000
- LogP 2.50340
Synthetic route

| Conditions | Yield |
|---|---|
| With 2 weight% platinum on carbon; ethanol at 100℃; under 7500.75 Torr; for 8h; Autoclave; | 100% |
| With hydrogen In ethanol at 20℃; under 7500.75 Torr; for 8h; Catalytic behavior; Sealed tube; | 100% |
| With hydrazine hydrate In ethanol; water at 80℃; for 1h; chemoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| 100% |

| Conditions | Yield |
|---|---|
| 100% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; potassium iodide | 98.2% |

| Conditions | Yield |
|---|---|
| With samarium diiodide In tetrahydrofuran for 2h; Ambient temperature; | 98% |
| Stage #1: 1-azido-3-chlorobenzene With hydrazine hydrate for 0.166667h; Inert atmosphere; Stage #2: for 10h; Irradiation; chemoselective reaction; | 90% |
| With zinc(II) tetrahydroborate In 1,2-dimethoxyethane for 3.5h; Ambient temperature; | 89% |

| Conditions | Yield |
|---|---|
| A 97.9% B n/a |
-
-
608-27-5
2,3-dichloroaniline

-
-
95-50-1
1,2-dichloro-benzene

-
-
95-76-1
m,p-dichloroaniline

-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With HI; hydrogen iodide | 96.5% |


| Conditions | Yield |
|---|---|
| With hydrogen; palladium/charcoal In tantalum; water; toluene | 96% |


| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide; C17H14N2O3; potassium hydroxide In ethanol at 60℃; for 24h; Schlenk technique; Inert atmosphere; | 96% |
| With ammonium hydroxide; potassium phosphate; 1-(5,6,7,8-tetrahydroquinolin-8-yl)-2-methylpropan-1-one; copper(I) bromide In dimethyl sulfoxide at 25℃; for 24h; Inert atmosphere; Sealed tube; | 90% |
| With copper(l) iodide; tetra(n-butyl)ammonium hydroxide; ammonia In water at 25℃; for 24h; Inert atmosphere; Sealed tube; chemoselective reaction; | 89% |

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide In 1-methyl-pyrrolidin-2-one at 80℃; for 15h; | 93% |
| With ammonium hydroxide; potassium phosphate; 1-(5,6,7,8-tetrahydroquinolin-8-yl)-2-methylpropan-1-one; copper(I) bromide In dimethyl sulfoxide at 110℃; for 24h; Inert atmosphere; Sealed tube; | 93% |
| With ammonium hydroxide; copper(l) iodide; 2,2-[μ-(N,N'-piperazindiyl)dimethyl]-bis(4,6-di-tert-butyl-phenol) In water at 120℃; for 12h; | 85% |

-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With indium; ammonium chloride In ethanol for 2.5h; Heating; | 86% |
-
-
36229-42-2
(3-chlorophenyl)magnesium bromide

-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| Stage #1: (3-chlorophenyl)magnesium bromide With zinc(II) chloride In tetrahydrofuran at -3 - 20℃; Inert atmosphere; Stage #2: With 1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone; dimethylsulfide; acetone oxime-o-p-benzenesulfonic ester; copper(ll) bromide In tetrahydrofuran at 20℃; for 1.5h; Inert atmosphere; Stage #3: With hydrogenchloride; water In tetrahydrofuran at 20℃; for 4h; Inert atmosphere; | 85% |
| Multi-step reaction with 3 steps 1: tetrahydrofuran / -3 - 20 °C / Inert atmosphere 2: copper(l) cyanide / tetrahydrofuran / 1.5 h / 20 °C / Inert atmosphere 3: hydrogenchloride / water; tetrahydrofuran / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: tetrahydrofuran / -5 - 20 °C / Inert atmosphere 2.1: acetone O-(2,4,6-trimethylphenylsulfonyl)oxime; triphenylphosphine / tetrahydrofuran / 0.08 h / 20 °C / Inert atmosphere 2.2: 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: tetrahydrofuran / 0.17 h / 10 °C / Inert atmosphere 2: copper(l) cyanide / tetrahydrofuran / 0.5 h / 20 °C / Inert atmosphere 3: water; hydrogenchloride / 24 h / 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: tetrahydrofuran / 0.17 h / 10 °C / Inert atmosphere 2: copper(l) cyanide / tetrahydrofuran / 0.5 h / 20 °C / Inert atmosphere 3: water; hydrogenchloride / 24 h / 20 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water | 84.1% |

| Conditions | Yield |
|---|---|
| With dihydridocarbonyl(1,1,1-tris(diphenylphosphinomethyl)ethane)ruthenium(II) complex; boron trifluoride diethyl etherate; hydrogen; toluene-4-sulfonic acid In tetrahydrofuran at 120℃; under 38002.6 Torr; for 16h; Glovebox; Autoclave; Inert atmosphere; | A 84% B n/a |
| With [bis(2-methylallyl)cycloocta-1,5-diene]ruthenium(II); formic acid; bis(trifluoromethanesulfonyl)amide; triethylamine; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In dibutyl ether at 130℃; for 24h; | A n/a B 76% |
| With C18H32ClIrO2P2; tris(pentafluorophenyl)borate; hydrogen; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In toluene at 120℃; under 38002.6 Torr; for 24h; Overall yield = 84 %; Overall yield = 87 %Chromat.; chemoselective reaction; |
-
-
78627-94-8
2-(trimethylsilyl)-5-chloronitrobenzene

-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin for 15h; Heating; | 83% |

| Conditions | Yield |
|---|---|
| With methanol; oxalyl dichloride at 20℃; for 2h; | 83% |
-
-
121-73-3
3-Chloronitrobenzene

-
A
-
62-53-3
aniline

-
B
-
108-42-9
3-chloro-aniline

-
C
-
15426-14-9, 106131-20-8, 106131-24-2
3,3'-dichloroazobenzene

-
D
-
139-24-2, 71297-98-8
3,3'-dichloroazoxybenzene

| Conditions | Yield |
|---|---|
| With bis(acetylacetonato)palladium(II); phosphorus; hydrogen In N,N-dimethyl-formamide; benzene at 50℃; under 1520.1 Torr; for 1.75h; Catalytic behavior; Time; chemoselective reaction; | A 9.4% B 81.4% C n/a D n/a |


| Conditions | Yield |
|---|---|
| With Rh/Al2O3; hydrogen In water; toluene at 80℃; under 30003 Torr; for 24h; Reagent/catalyst; | A 17% B 81% |
| With isopropyl alcohol; sodium hydroxide for 12h; Inert atmosphere; Reflux; chemoselective reaction; | A n/a B 76% |
| With hydrogen; nickel-chromium catalyst In isopropyl alcohol at 49.9℃; under 760 Torr; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; benzyltrimethylazanium tribroman-2-uide In water for 2h; Ambient temperature; | 79% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-chlorobenzoate With trifluorormethanesulfonic acid; trimethylsilylazide In chloroform at 90℃; for 0.055h; Schmidt Reaction; Flow reactor; Stage #2: With water In methanol; chloroform at 20℃; Schmidt Reaction; Flow reactor; | 79% |
| With thionyl chloride; hydroxylamine-O-sulfonic acid 1) 1h, reflux, 2) toluene, 2h, reflux; Yield given. Multistep reaction; |
-
-
121-73-3
3-Chloronitrobenzene

-
A
-
108-42-9
3-chloro-aniline

-
B
-
15426-14-9, 106131-20-8, 106131-24-2
3,3'-dichloroazobenzene

-
C
-
139-24-2, 71297-98-8
3,3'-dichloroazoxybenzene

| Conditions | Yield |
|---|---|
| With hydrazine hydrate; nickel(II) nitrate; zinc In tert-butyl alcohol for 0.333333h; Heating; | A 5% B 6 % Chromat. C 78% |
| With hydrazine hydrate; nickel(II) nitrate; zinc In ethanol for 3h; Heating; | A 15 % Chromat. B 12% C 43% |

-
-
120-72-9
indole

-
-
3340-78-1
2-phenyl-1,2,3,4-tetrahydroisoquinoline

-
-
121-73-3
3-Chloronitrobenzene

-
B
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With Co(dmgH)2Cl2; Eosin Y In water; acetonitrile at 20℃; for 48h; Inert atmosphere; Irradiation; Sealed tube; | A 78% B 90 %Spectr. |


| Conditions | Yield |
|---|---|
| With methanesulfonic acid; trimethylsilylazide In tetrachloromethane at 80℃; for 4h; | 76% |
-
-
63503-60-6
3-chlorophenylboronic acid

-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With copper(I) oxide; ammonium hydroxide; air In methanol at 20℃; for 18h; | 74% |
| With copper(I) oxide; ammonium hydroxide; oxygen; sodium hydroxide In water at 25℃; for 15h; | 70% |

| Conditions | Yield |
|---|---|
| With tributylgermanium hydride; thiophenol In toluene for 0.5h; Heating; | A 7% B 5% C 73% |


-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 140℃; for 6h; Green chemistry; | 71% |

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; potassium phosphate tribasic heptahydrate; water; Sucrose In water at 90℃; for 15h; Catalytic behavior; Green chemistry; | 70% |
-
-
25419-60-7
1-(3-Chlor-anilino)-3-(3-chlor-phenylimino)-propen

-
A
-
51217-97-1
β-(m-Chloranilino)acrolein

-
B
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| With water; sodium acetate; acetic acid In ethanol at 70℃; for 32.5h; | A 52% B 68% |
-
-
541-41-3
chloroformic acid ethyl ester

-
-
108-42-9
3-chloro-aniline

-
-
2150-89-2
(3-chlorophenyl) carbamic acid ethyl ester

| Conditions | Yield |
|---|---|
| With pyridine at 0℃; for 1h; | 100% |
| With sodium carbonate; benzene | |
| With potassium carbonate In 1,4-dioxane for 2h; Heating; | |
| With pyridine at 0 - 20℃; |
-
-
79-04-9
chloroacetyl chloride

-
-
108-42-9
3-chloro-aniline

-
-
2564-05-8
2-chloro-N-(3-chlorophenyl)acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 25℃; | 100% |
| With triethylamine In dichloromethane at 0 - 25℃; | 100% |
| In tetrahydrofuran at 20℃; for 18h; | 100% |
-
-
108-42-9
3-chloro-aniline

-
-
87-13-8
diethyl 2-ethoxymethylenemalonate

-
-
3412-99-5
2-(3-chlorophenylamino)methylenemalonic acid diethyl ester

| Conditions | Yield |
|---|---|
| In toluene at 110℃; for 24h; | 100% |
| In neat (no solvent) at 120℃; for 0.75h; | 98% |
| In ethanol for 3h; Reflux; | 91% |
-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
108-42-9
3-chloro-aniline

-
-
82363-26-6
(3-Chloro-phenyl)-[1-(3,4-dimethoxy-phenyl)-meth-(E)-ylidene]-amine

| Conditions | Yield |
|---|---|
| In toluene Heating; | 100% |
-
-
109768-45-8
2-isothiocyanatobenzylpyridinium bromide

-
-
108-42-9
3-chloro-aniline

-
-
109768-52-7
1-{2-[3-(3-Chloro-phenyl)-thioureido]-benzyl}-pyridinium; bromide

| Conditions | Yield |
|---|---|
| In methanol for 5h; | 100% |
-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| Stage #1: trifluoromethyl dihydro-1,4-dioxin-3-carbonyl chloride With pyridine; polystyrene-bound 4-hydroxy-3-nitrobenzophenone In dichloromethane at 20℃; for 24h; Condensation; Stage #2: 3-chloro-aniline With triethylamine In acetonitrile for 14h; Condensation; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With C15H29IrN4(2+)*2I(1-) at 120℃; for 17h; Inert atmosphere; Schlenk technique; Sealed tube; | 100% |
| With tetrachloromethane; copper(ll) bromide at 180℃; for 6h; Inert atmosphere; Sealed tube; | 99% |
| With sulfuric acid at 200℃; for 16h; | 77% |
| With TiO2 supported nano-Pd(0.3) catalyst In water at 20℃; for 5h; Inert atmosphere; Irradiation; Green chemistry; | 65 %Chromat. |
| With zinc oxide-supported iridium catalyst at 150℃; under 3750.38 Torr; for 5h; Inert atmosphere; | 99 %Chromat. |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 100℃; for 1h; | 100% |
| With triethylamine In butan-1-ol at 100℃; | |
| Stage #1: 1,4-dichlorophthalazine; 3-chloro-aniline In ethanol for 0.5h; Reflux; Stage #2: With sodium hydroxide In ethanol; water at 20℃; |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
108-42-9
3-chloro-aniline

-
-
5330-63-2
t-butyl 3-chlorophenylcarbamate

| Conditions | Yield |
|---|---|
| In methanol at 100℃; for 6h; | 100% |
| Stage #1: 3-chloro-aniline With triethylamine In tetrahydrofuran; water at 20℃; for 0.0833333h; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran; water at 0 - 20℃; for 6h; | 100% |
| With 1,1'-hexane-1,6-diylbis(3-methylpyridinium)tetrachlorocobaltate(II) In neat (no solvent) at 20℃; for 1h; | 97% |

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium (0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In toluene at 90 - 110℃; for 11h; Buchwald-Hartwig reaction; | 100% |
| With pectin-stabilized copper nanoparticles In dimethyl sulfoxide at 110℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite; trimethylsilylazide In acetonitrile at 0 - 20℃; for 1h; | 100% |
| Stage #1: 3-chloro-aniline With hydrogenchloride; sodium nitrite In ethyl acetate at 0℃; for 0.5h; Stage #2: With sodium azide In ethyl acetate at 20℃; for 2h; | 91% |
| Stage #1: 3-chloro-aniline With hydrogenchloride In water; ethyl acetate at 0℃; for 0.166667h; Stage #2: With sodium nitrite In water; ethyl acetate for 0.5h; Stage #3: With sodium azide In water; ethyl acetate for 0.5h; | 88% |
-
-
598-21-0
2-Bromoacetyl bromide

-
-
108-42-9
3-chloro-aniline

-
-
41964-65-2
2-bromo-N-(3-chlorophenyl)acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 1.25h; Inert atmosphere; | 100% |
| With triethylamine In dichloromethane at 0℃; for 1.25h; Inert atmosphere; | 100% |
| With triethylamine at 0℃; for 1h; Inert atmosphere; | 100% |
-
-
108-42-9
3-chloro-aniline

-
-
407-25-0
trifluoroacetic anhydride

-
-
40410-54-6
N-(3-chlorophenyl)-2,2,2-trifluoro-acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 0.75h; | 100% |
| With triethylamine In dichloromethane at 0 - 25℃; for 12h; Inert atmosphere; | 91% |
| With triethylamine In dichloromethane at -78 - 26℃; for 1h; | 2.29 g |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
79099-07-3
N-tert-butyloxycarbonylpiperidin-4-one

-
-
108-42-9
3-chloro-aniline

| Conditions | Yield |
|---|---|
| In acetic acid at 0℃; | 100% |
| In acetic acid at 0 - 20℃; |

-
-
108-42-9
3-chloro-aniline

-
-
160893-07-2
2-chloro-5-methoxyquinoline

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 160℃; | 100% |
| In neat (no solvent) at 160℃; |

| Conditions | Yield |
|---|---|
| With triethylamine In acetone at 90℃; for 3h; Inert atmosphere; | 100% |
-
-
108-42-9
3-chloro-aniline

-
-
89284-57-1
2,4,6-tribromo-3-chloroaniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; dihydrogen peroxide; sodium bromide In water at 15℃; for 2h; | 99.8% |
| With bromine In tetrachloromethane at 20 - 40℃; | 91% |
| With methanol; P-benzyltriphenylphosphonium tribromide; sodium hydrogencarbonate In dichloromethane for 0.166667h; Ambient temperature; | 86% |
-
-
21286-54-4
Camphorsulfonyl chloride

-
-
108-42-9
3-chloro-aniline

-
-
132886-02-3, 132957-82-5
(1S)-2-oxo-bornane-10-sulfonic acid-(3-chloro-anilide)

| Conditions | Yield |
|---|---|
| With dmap In acetonitrile at 0℃; for 1h; Inert atmosphere; | 99% |
-
-
108-42-9
3-chloro-aniline

-
-
135050-44-1
3-chloro-4-iodoaniline

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; N,N,N-trimethylbenzenemethanaminium dichloroiodate In methanol; dichloromethane at 20℃; for 0.416667h; | 99% |
| With tetramethylammonium dibromoiodate(I) at 20℃; for 0.116667h; | 97% |
| With iodine at 30℃; for 12h; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With hydrogen; triethylamine In water at 120 - 140℃; under 22502.3 - 37503.8 Torr; | 99% |
| With lithium at 230℃; Zersetzen der Reaktionsprodukte mit Wasser; | |
| With potassium fluoride; polymethylhydrosiloxane; palladium diacetate In tetrahydrofuran at 20℃; for 0.75h; | 94 % Spectr. |

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; acetone at 20℃; | 99% |
| With ammonia; N,N-dimethyl-aniline In benzene |
-
-
108-42-9
3-chloro-aniline

-
-
118-91-2
ortho-chlorobenzoic acid

-
-
13278-36-9
N-(3-chlorophenyl)anthranilic acid

| Conditions | Yield |
|---|---|
| With copper(I) oxide; copper; potassium carbonate In 2-ethoxy-ethanol at 130℃; for 24h; | 99% |
| With pyridine; copper; potassium carbonate In water for 1h; Heating; | 60% |
| With copper; potassium carbonate for 5h; Heating; | 44% |

| Conditions | Yield |
|---|---|
| In toluene Heating; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-chloro-aniline With boron trifluoride diethyl etherate In acetonitrile Cooling with ice; Stage #2: With isopentyl nitrite In acetonitrile at 0℃; for 0.5h; | 99% |
| With tetrafluoroboric acid; sodium nitrite In water Diazotization; | 95% |
| Stage #1: 3-chloro-aniline With hydrogenchloride; sodium nitrite In water for 0.166667h; Cooling with ice; Stage #2: With sodium tetrafluoroborate for 0.5h; Cooling with ice; | 74% |
-
-
4270-27-3
6-chlorouracil

-
-
108-42-9
3-chloro-aniline

-
-
7269-03-6
6-(3-chlorophenylamino)pyrimidine-2,4(1H,3H)-dione

| Conditions | Yield |
|---|---|
| at 170℃; for 0.333333h; | 99% |
| for 2h; Reflux; | 95% |
| In melt at 180 - 200℃; |
3-Chloroaniline Chemical Properties
Molecular Formula: C6H6ClN
Formula Weight: 127.57
EINECS: 203-581-0
Index of Refraction: 1.598
Molar Refractivity: 35.38 cm3
Molar Volume: 103.6 cm3
Surface Tension: 45.3 dyne/cm
Density: 1.23 g/cm3
Flash Point: 123.9 °C
Enthalpy of Vaporization: 46.43 kJ/mol
Boiling Point: 227.8 °C at 760 mmHg
Vapour Pressure: 0.0762 mmHg at 25 °C
Melting point: -11--9 °C(lit.)
Water solubility: 6.8 g/L (20 °C)
Appearance: Clear pale yellow to brown liquid
Structure of 3-Chloroaniline (CAS NO.108-42-9):
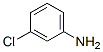
IUPAC Name: 3-Chloroaniline
Canonical SMILES: C1=CC(=CC(=C1)Cl)N
InChI: InChI=1S/C6H6ClN/c7-5-2-1-3-6(8)4-5/h1-4H,8H2
InChIKey: PNPCRKVUWYDDST-UHFFFAOYSA-N
Product Category of 3-Chloroaniline (CAS NO.108-42-9): Chlorobenzene Series;Amines;Nitrogen Compounds;CAlphabetic;Chemical Class;European Community: ISO and DIN
3-Chloroaniline Uses
3-Chloroaniline (CAS NO.108-42-9) is used as an intermediate in the production of a number of products, including agricultural chemicals, azo dyes and pigments, bactericide or biocide and pharmaceuticals.
3-Chloroaniline Production
In the presence of ferrous sulfate, 3-CHLOROANILINE (108-42-9) is synthesized from m-nitrochlorobenzne by condensation of iron powder.
3-Chloroaniline Toxicity Data With Reference
| 1. | mmo-asn 200 mg/L | CJMIAZ Canadian Journal of Microbiology. 16 (1970),369. | ||
| 2. | orl-rat LD50:256 mg/kg | GISAAA Gigiena i Sanitariya. 31 (12)(1966),6. | ||
| 3. | skn-rat LD50:250 mg/kg | 85GMAT Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure Izmerov, N.F., et al.,Moscow.: USSR, Centre of International Projects, GKNT,1982,34. | ||
| 4. | orl-mus LD50:334 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 13 (5)(1969),29. | ||
| 5. | ihl-mus LC50:550 mg/m3/4H | 85GMAT Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure Izmerov, N.F., et al.,Moscow.: USSR, Centre of International Projects, GKNT,1982,34. | ||
| 6. | ivn-dog LDLo:50 mg/kg | AEPPAE Naunyn-Schmiedebergs Archiv fuer Experimentelle Pathologie und Pharmakologie. 244 (1963),387. | ||
| 7. | skn-cat LD50:223 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 13 (5)(1969),29. | ||
| 8. | scu-cat LDLo:125 mg/kg | AHBAAM Archiv fuer Hygiene und Bakteriologie. 110 (1933),12. | ||
| 9. | orl-gpg LD50:250 mg/kg | GISAAA Gigiena i Sanitariya. 31 (12)(1966),6. |
3-Chloroaniline (CAS NO.108-42-9) hasn't been listed as a carcinogen by NTP, IARC, ACGIH, or CA Prop 65. And the toxicological properties have not been fully investigated. You can see actual entry in RTECS for complete information.
3-Chloroaniline Consensus Reports
EPA Genetic Toxicology Program. Reported in EPA TSCA Inventory.
3-Chloroaniline Safety Profile
Safety: DANGER: POISON, irritates skin, eyes, lungs
Hazard Codes:  T,
T, N
N
Risk Statements: 23/24/25-33-50/53 (Toxic by inhalation, in contact with skin and if swallowed; Danger of cumulative effects; Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment)
Safety Statements: 28-36/37-45-60-61-28A (After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer);
Poison by ingestion, skin contact, subcutaneous, and intravenous routes. Mutation data reported. When heated to decomposition it emits toxic fumes of Cl− and NOx. See also ANILINE DYES.
3-Chloroaniline Standards and Recommendations
ASSAY: 99.0% min
WATER: 0.3% max
3-Chloroaniline Specification
3-Chloroaniline , its cas register number is 108-42-9. It also can be called 1-Amino-3-chlorobenzene ; m-Aminochlorobenzene ; 3-Chlorophenylamine ; Fast Orange GC base ; and 3-Chlorobenzenamine . It should be stored in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances. It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, 3-Chloroaniline (CAS NO.108-42-9) darkens on exposure to light and air. It is not compatible with strong oxidizing agents, acids, acid chlorides, acid anhydrides, chloroformates, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: Hydrogen chloride, nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
Related Products
- 3-Chloroaniline
- 3-CHLOROANILINE HYDROCHLORIDE
- 108-43-0
- 1084334-28-0
- 108433-95-0
- 108433-99-4
- 108436-80-2
- 108437-64-5
- 108437-87-2
- 108437-88-3
- 108437-89-4
- 108-44-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View