-
Name
3-Fluorobenzaldehyde
- EINECS 207-266-9
- CAS No. 456-48-4
- Article Data144
- CAS DataBase
- Density 1.178 g/cm3
- Solubility
- Melting Point 173 °C
- Formula C7H5FO
- Boiling Point 173.2 °C at 760 mmHg
- Molecular Weight 124.115
- Flash Point 56.7 °C
- Transport Information UN 1989 3/PG 3
- Appearance clear colorless to yellow liquid
- Safety 24/25-37/39-26-36/37/39-27
- Risk Codes 37/38-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, F
F
- Synonyms Benzaldehyde,m-fluoro- (6CI,7CI,8CI);3-Fluorobenzenecarboxaldehyde;3-Fluorophenylcarboxaldehyde;NSC 66830;m-Fluorobenzaldehyde;
- PSA 17.07000
- LogP 1.63820
Synthetic route

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; zircornium(IV) n-propoxide In dichloromethane at 20℃; for 5h; molecular sieves; | 98% |
| With ammonium nitrate; hydrogenchloride; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen In water; acetonitrile at 60℃; for 6h; Green chemistry; | 95% |
| With dmap; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; copper(I) triflate; 5-[(2S)-pyrrolidine-2-yl]-1H-tetrazole In acetonitrile at 25℃; for 1.5h; Catalytic behavior; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide at 100℃; for 10h; | 93% |
| With dimethyl amine at 60℃; for 1h; | 80% |
| With calcium carbonate |

| Conditions | Yield |
|---|---|
| With iron(III) trifluoromethanesulfonate; 2-((4R,5R)-1-((4-(tert-butyl)phenyl)sulfonyl)-4,5-diphenylimidazolidin-2-yl)-6-((4R,5R)-1-((4-(tert-butyl)phenyl)sulfonyl)-4,5-diphenylimidazolidin-2-yl)pyridine; oxygen In 1,2-dichloro-ethane at 70℃; under 760.051 Torr; for 6h; Green chemistry; chemoselective reaction; | 93% |
| With tetrafluoroboric acid; iodobenzene; 3-chloro-benzenecarboperoxoic acid In water; toluene at 35℃; | 71 %Spectr. |

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide at 120℃; for 12h; | 92% |
| With N,N-dimethyl-formamide at 120℃; for 7h; | 90% |

| Conditions | Yield |
|---|---|
| at 100 - 120℃; for 10 - 12h; | 92% |

| Conditions | Yield |
|---|---|
| With triethylsilane; palladium diacetate; sodium hydrogencarbonate; sodium carbonate at 20℃; under 760.051 Torr; for 48h; | 92% |
| With rhodium(III) chloride trihydrate; hydrogen; triethylamine; triphenylphosphine In N,N-dimethyl acetamide at 90℃; under 7500.75 Torr; for 12h; Autoclave; | 82% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole; C17H16ClMnN2O2; tetrabutylammonium periodite In chloroform at 20℃; for 0.333333h; | 91% |
| With potassium 12-tungstocobaltate(III) In water; acetonitrile for 0.3h; Microwave irradiation; | 90% |
| With oxygen; copper diacetate In dimethyl sulfoxide at 120℃; for 18h; Sealed tube; | 65% |
| With 1H-imidazole; [bis(acetoxy)iodo]benzene; Co(AAOPD) In acetonitrile at 20℃; |

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide at 20℃; for 0.0833333h; Microwave irradiation; | 87% |
| With (NH4)4[ZnMo6O18(OH)6]; oxygen In water; acetonitrile at 60℃; under 760.051 Torr; for 12h; | 84% |
| With 4Na(1+)*6H(1+)*NiMo6O24(10-)=Na4H6NiMo6O24; oxygen In water; acetonitrile at 20℃; under 760.051 Torr; for 12h; Irradiation; | 81% |
| With ethanol; carbon dioxide; hexamethylenetetramine |
-
-
1073-06-9
3-fluorobromobenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 3-fluorobromobenzene With n-butyllithium; isopropylmagnesium chloride In tetrahydrofuran; hexane at 0 - 5℃; for 1h; Stage #2: N,N-dimethyl-formamide In tetrahydrofuran at 0℃; for 1h; Further stages.; | 81% |

| Conditions | Yield |
|---|---|
| With rhodium(III) iodide; hydrogen; acetic anhydride; triethylamine; triphenylphosphine In N,N-dimethyl acetamide at 100℃; for 24h; Autoclave; | 80% |

| Conditions | Yield |
|---|---|
| With rhodium(III) iodide; dichloro [1,1'-bis(diphenylphosphino)propane]palladium(II); hydrogen; acetic anhydride; triethylamine; triphenylphosphine In N,N-dimethyl acetamide at 100℃; for 24h; Autoclave; | 80% |
-
-
40096-23-9
(3-fluorophenyl)methanethiol

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; tetrakis(pyridine)silver(II) peroxodisulfate; oxygen In N,N-dimethyl-formamide at 23℃; under 760.051 Torr; Irradiation; | 78% |
-
-
456-47-3
3-fluoro-benzenemethanol

-
-
98-95-3
nitrobenzene

-
A
-
58606-65-8
N-[(3-fluorophenyl)methylidene]aniline

-
B
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With α,α,α-trifluorotoluene; titanium(IV) oxide In dodecane under 750.075 Torr; for 3h; Darkness; Inert atmosphere; Irradiation; | A 72.67% B n/a |

-
-
51608-60-7
N-(benzylidene)-p-methylbenzenesulfonamide

-
-
1418179-06-2
C20H16F3NO

-
B
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With [Cp*Rh(CH3CN)3][SbF6]2 In tert-butyl alcohol at 90℃; for 3.5h; Schlenk technique; Inert atmosphere; | A 71% B n/a |

-
-
24133-57-1
1,2-bis(3-fluorophenyl)-1,2-ethanediol

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; cetyltrimethylammonim bromide In water at 20℃; under 760.051 Torr; for 5h; Irradiation; Green chemistry; | 67% |
-
-
32222-47-2, 52923-24-7, 52950-14-8, 395-05-1
2-(3-fluorophenyl)-2-hydroxyacetic acid

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; copper diacetate In dimethyl sulfoxide at 120℃; for 10h; Sealed tube; | 58% |

| Conditions | Yield |
|---|---|
| With hydrogen; quinoline; 5% palladium on barium sulfate; sulfur at 80 - 90℃; for 9.5h; | 57% |
| With Pd-BaSO4; xylene |

| Conditions | Yield |
|---|---|
| With sodium molybdate; dihydrogen peroxide; cobalt(II) acetate; acetic acid; sodium bromide at 140℃; for 0.5h; Temperature; Concentration; | 56.1% |
| With oxygen In water at 150℃; under 30002.4 Torr; for 2h; | 48% |
| With carbon disulfide; chromyl chloride Behandeln des Reaktionsprodukts mit kalter wss. SO2-Loesung; |

| Conditions | Yield |
|---|---|
| With picoline; tert.-butylhydroperoxide; chlorophyllin coppered trisodium salt In water at 60℃; for 12h; Temperature; chemoselective reaction; | A 35% B 56% |
-
-
127598-78-1
N-(2-cyanoethyl) N-(3-fluorobenzyl)-4-methylaniline

-
A
-
118072-35-8
3-(2-Dimethylaminomethyl-4-methyl-phenylamino)-propionitrile

-
B
-
127598-86-1
N-(2-cyanoethyl) N-(3-fluorobenzyl)-2-amino-5-methylbenzaldehyde

-
C
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With water; trichlorophosphate In N,N-dimethyl-formamide Vilsmeier reaction; | A 49% B 13% C 40% |
| With water; trichlorophosphate In N,N-dimethyl-formamide Vilsmeier reaction; | A n/a B 13% C 40% |

-
-
64-19-7
acetic acid

-
-
123-38-6
propionaldehyde

-
-
215320-36-8
(+/-)-1-(3'-fluorophenyl)but-3-en-1-ol

-
C
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With trimethylsilyl acetate; boron trifluoride diethyl etherate In cyclohexane at 20℃; for 3h; Prins cyclization; | A 9% B 46% C 9% |


| Conditions | Yield |
|---|---|
| With sodium carbonate; ethylene glycol |

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol |

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide In water at 24.9℃; Kinetics; Thermodynamic data; pH 10.6; ΔH(excit.), ΔS(excit.); | |
| With N-bromoacetamide In water at 14.9 - 34.9℃; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.); |
-
-
20595-30-6
3-Fluorocinnamic acid

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With oxalic acid; pyridinium chlorochromate In water at 35℃; Rate constant; Kinetics; Thermodynamic data; further temperatures, ΔH(act.), ΔS(act.); | |
| With chloroamine-T; oxalic acid In water at 35℃; Rate constant; Kinetics; Thermodynamic data; furhter temperatures, ΔH(act.), ΔS(act.); |

| Conditions | Yield |
|---|---|
| With potassium fluoride; tetraphenylphosphonium bromide; Phthaloyl dichloride In sulfolane; toluene at 210℃; for 5h; | 7 % Chromat. |

| Conditions | Yield |
|---|---|
| With fluorine In trichlorofluoromethane at -78℃; Product distribution; addition of BCl3; |
-
-
108133-60-4
4-{[1-(3-Fluoro-phenyl)-meth-(E)-ylidene]-amino}-benzoic acid

-
A
-
150-13-0
4-amino-benzoic acid

-
B
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With borate buffer; water In methanol at 25℃; Rate constant; Kinetics; Mechanism; ΔG(activ.), ΔH(activ.), ΔS(activ.); |

| Conditions | Yield |
|---|---|
| With perchloric acid; water; chloramine-B In N,N-dimethyl-formamide at 30℃; Rate constant; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.); var. temperature; | |
| With perchloric acid; N-bromobenzenesulphonamide; acetic acid at 30℃; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.); other temp.; |

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 100% |
| In benzene Heating; |

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| at 80℃; for 1h; | |
| at 20℃; for 1h; | |
| In methanol; water at 20℃; for 0.333333h; | |
| at 20℃; for 1h; |
-
-
22483-09-6
2,2-dimethoxyethylamine

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
73261-94-6
N-<(3-fluorophenyl)methylene>-2,2-dimethoxyethanamine

| Conditions | Yield |
|---|---|
| In toluene for 1.5h; Heating; | 100% |
| for 0.333333h; Heating; | |
| In benzene Reflux; |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
100948-28-5, 82128-93-6
2-(3-fluorophenyl)-2-((trimethylsilyl)oxy)acetonitrile

| Conditions | Yield |
|---|---|
| With triethylamine at 20℃; for 24h; | 100% |
| With C32H39Br2MgN2(1-)*C16H32LiO4(1+) In chloroform-d1 at 20℃; for 1h; Inert atmosphere; Glovebox; | 99% |
| With zinc(II) iodide at 5 - 20℃; for 0.333333h; Yield given; |

| Conditions | Yield |
|---|---|
| With hydrogen; Et4N In 1,2-dimethoxyethane at 100℃; under 38000 Torr; for 13h; | 100% |
| With triethylamine; isopropyl alcohol; lithium bromide at 20℃; for 48h; Meerwein-Ponndorf-Verley reaction; | 98% |
| With Candida boidinii formate dehydrogenase; Geobacillus stearothermophilus ε‐deaminating L‐lysine dehydrogenase variant 24; nicotinamide adenine dinucleotide In aq. buffer at 30℃; for 24h; pH=7; Reagent/catalyst; Enzymatic reaction; | 94% |
-
-
4276-09-9
(R)-2-amino-3-methylbutanol

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
860805-86-3
2-[(3-fluoro-benzylidene)-amino]-3-methyl-butan-1-ol

| Conditions | Yield |
|---|---|
| With magnesium sulfate In dichloromethane at 0 - 20℃; for 16h; | 100% |
| In dichloromethane at 0 - 20℃; for 16h; drying with MgSO4; | 100% |
| With magnesium sulfate In dichloromethane at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| With magnesium sulfate In dichloromethane at 0 - 20℃; for 16h; | 100% |
| With magnesium sulfate In dichloromethane at 0 - 20℃; for 16h; | 100% |
| In dichloromethane |
-
-
773071-71-9
3-(2,3-dihydroxy-propylsulfanylmethyl)-N-[2-hydrazinocarbonyl-4,5-bis-(2-methoxy-ethoxy)-phenyl]-benzamide

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 1h; | 100% |
-
-
558-13-4
carbon tetrabromide

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
221148-37-4
1-(2,2-Dibromo-vinyl)-3-fluoro-benzene

| Conditions | Yield |
|---|---|
| With triphenylphosphine In dichloromethane | 100% |
| Stage #1: carbon tetrabromide With triphenylphosphine In dichloromethane at 0 - 10℃; for 1h; Stage #2: 3-Fluorobenzaldehyde In dichloromethane at 0℃; for 1h; | 100% |
| Stage #1: carbon tetrabromide With triphenylphosphine In dichloromethane at 0 - 10℃; for 1.05h; Stage #2: 3-Fluorobenzaldehyde In dichloromethane at 0℃; for 1h; | 100% |
-
-
2579-20-6
cis, trans-1,3-dimethylaminocyclohexane

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1217527-05-3
C22H24F2N2

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Molecular sieve; | 100% |
| In methanol at 20℃; Molecular sieve; |
-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1201652-24-5
(E)-2-(3-fluorobenzylideneamino)-N-phenylbenzamide

| Conditions | Yield |
|---|---|
| 100% |

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1332928-88-7
tert-butyl (E)-2-deuterio-3-(3'-fluorophenyl)propenoate

| Conditions | Yield |
|---|---|
| With water-d2; potassium carbonate at 50℃; for 30h; Horner-Wadsworth-Emmons olefination; Inert atmosphere; optical yield given as %de; diastereoselective reaction; | 100% |

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With iron(III) chloride In dichloromethane at 60℃; for 16h; Pictet-Spengler Synthesis; | 100% |

| Conditions | Yield |
|---|---|
| In methanol | 100% |
-
-
86302-43-4
(tert-Butoxycarbonylmethylene)triphenylphosphorane

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
277745-38-7
(E)-3-(3-fluorophenyl)acrylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 16h; | 100% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 60℃; for 33h; | 100% |
-
-
118-93-4
o-hydroxyacetophenone

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1840-00-2
(2E)-3-(3-fluorophenyl)-1-(2-hydroxyphenyl)prop-2-en-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 20℃; Aldol Condensation; | 99.2% |
| Stage #1: o-hydroxyacetophenone With potassium hydroxide In ethanol at 0℃; for 0.166667h; Claisen-Schmidt Condensation; Stage #2: 3-Fluorobenzaldehyde In ethanol at 0 - 20℃; for 8h; Claisen-Schmidt Condensation; | 76% |
| With sodium hydroxide In ethanol; water at 20℃; | 53% |
| With sodium hydroxide In ethanol; water at 20℃; for 24h; | 32% |
| With sodium hydroxide In ethanol; water at 20℃; |

| Conditions | Yield |
|---|---|
| Stage #1: malonic acid; 3-Fluorobenzaldehyde With piperidine; pyridine Knoevenagel-Doebner reaction; Reflux; Stage #2: With hydrogenchloride In water Cooling with ice; optical yield given as %de; | 99% |
| With piperidine In pyridine Heating; | 80% |
| With ammonia Erhitzen des Reaktionsprodukts auf 180-190grad; |
-
-
456-48-4
3-Fluorobenzaldehyde

-
-
66659-65-2
(rac)-1,2-bis(3-fluorophenyl)-2-hydroxyethan-1-one

| Conditions | Yield |
|---|---|
| With 1,3-dimethylbenzimidazolium Iodide; 1,8-diazabicyclo[5.4.0]undec-7-ene In tetrahydrofuran for 1h; Heating; | 99% |
| With Thiamine hydrochloride; sodium hydroxide In ethanol at 55℃; for 16h; Schlenk technique; Inert atmosphere; | 81% |
| With sodium cyanide In ethanol; water for 4h; Reflux; | 68.4% |
-
-
16676-29-2
naltrexone Hydrochloride

-
-
456-48-4
3-Fluorobenzaldehyde

| Conditions | Yield |
|---|---|
| With piperidine In methanol at 120℃; Sealed tube; | 99% |
| With sodium hydroxide for 24h; Ambient temperature; | 76% |
-
-
556-56-9
allyl iodid

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
215320-36-8
(+/-)-1-(3'-fluorophenyl)but-3-en-1-ol

| Conditions | Yield |
|---|---|
| With indium In water for 48h; | 99% |
-
-
292638-85-8
acrylic acid methyl ester

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
784144-71-4
methyl 2-((3-fluorophenyl)(hydroxy)methyl)acrylate

| Conditions | Yield |
|---|---|
| 1,4-diaza-bicyclo[2.2.2]octane; tributyl(ethyl)phosphonium tosylate at 20℃; for 24h; Baylis-Hillman reaction; | 99% |
| With 1,4-diaza-bicyclo[2.2.2]octane at 20℃; for 6h; Baylis-Hillman Reaction; Inert atmosphere; | 98% |
| With 1,4-diaza-bicyclo[2.2.2]octane at 20℃; for 3h; Morita-Baylis-Hillman Alkylation; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: With (1S,2S,5S)-4,6,6-trimethyl-2-[2-{(S)-6-phenylpyridin-2-yl}phenyl]bicyclo-[3.1.1]hept-3-en-2-ol; dimethyl zinc(II) In toluene at 0 - 20℃; for 0.5h; Inert atmosphere; Stage #2: phenylacetylene In toluene at 20℃; for 0.75h; Inert atmosphere; Stage #3: 3-Fluorobenzaldehyde In toluene at 0℃; for 72h; Inert atmosphere; | 99% |
| Stage #1: phenylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78 - 21℃; for 1h; Inert atmosphere; Stage #2: 3-Fluorobenzaldehyde In tetrahydrofuran; hexane at -78 - 21℃; for 1h; Inert atmosphere; | 96% |
| Stage #1: phenylacetylene With dimethyl zinc(II) In toluene at 20℃; for 0.5h; Stage #2: 3-Fluorobenzaldehyde In toluene at 20℃; for 24h; | 92% |
| Stage #1: phenylacetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: 3-Fluorobenzaldehyde In tetrahydrofuran; hexane at -78 - 20℃; for 1h; Inert atmosphere; | |
| Stage #1: phenylacetylene With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Stage #2: 3-Fluorobenzaldehyde In tetrahydrofuran at 20℃; |
-
-
135-19-3
β-naphthol

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
37096-98-3
14-(3-fluorophenyl)-14H-dibenzo[a,j]xanthene

| Conditions | Yield |
|---|---|
| With para-dodecylbenzenesulfonic acid at 125℃; under 5171.62 Torr; for 0.0666667h; Microwave irradiation; Neat (no solvent); | 99% |
| With phospho sulphonic acid In neat (no solvent) at 110℃; for 0.75h; Green chemistry; | 96% |
| With aminosulfonic acid for 0.0416667h; microwave irradiation; | 95% |
-
-
108-94-1
cyclohexanone

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
565416-89-9
2,6-bis(3-fluorobenzylidene)cyclohexanone

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 20℃; | 99% |
| With 1-methyl-3-(2-(sulfooxy)ethyl)-1H-imidazol-3-ium chloride at 20℃; for 2h; | 91% |
| With potassium hydroxide In ethanol at 20℃; for 4h; | 90.5% |
| With sodium hydroxide In ethanol at 5 - 20℃; Claisen Schmidt condensation; | |
| With potassium hydroxide In ethanol at 20℃; Claisen-Schmidt Condensation; |
-
-
120-92-3
cyclopentanone

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
892253-13-3
2,5-bis(3-fluorobenzylidene)cyclopentanone

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 20℃; | 99% |
| With 1-methyl-3-(2-(sulfooxy)ethyl)-1H-imidazol-3-ium chloride at 20℃; for 2h; | 91% |
| With sodium hydroxide In ethanol at 5 - 20℃; Claisen Schmidt condensation; |
-
-
40137-12-0
diethyl<<(p-chlorophenyl)sulfonyl>methyl>phosphonate

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1554271-57-6
(E)-1-(2-((4-chlorophenyl)sulfonyl)vinyl)-3-fluorobenzene

| Conditions | Yield |
|---|---|
| Stage #1: diethyl<<(p-chlorophenyl)sulfonyl>methyl>phosphonate With n-butyllithium In tetrahydrofuran; cyclohexane at -78℃; for 1h; Stage #2: 3-Fluorobenzaldehyde In tetrahydrofuran; cyclohexane at -78 - 20℃; for 1h; | 99% |
-
-
3949-36-8
3-acetylcoumarin

-
-
109-77-3
malononitrile

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
1421234-48-1
2-amino-6-(2-oxo-2H-chromen-3-yl)-4-(3-fluorophenyl)nicotinonitrile

| Conditions | Yield |
|---|---|
| With ammonium acetate In neat (no solvent) at 100℃; for 0.25h; | 99% |
| With perchloric acid adsorbed on silica gel; ammonium acetate In neat (no solvent) at 60℃; for 1.2h; Green chemistry; | 94% |


| Conditions | Yield |
|---|---|
| With scandium tris(trifluoromethanesulfonate) In nitromethane at 80℃; for 0.166667h; Microwave irradiation; Sealed tube; regioselective reaction; | 99% |
-
-
100-63-0
phenylhydrazine

-
-
456-48-4
3-Fluorobenzaldehyde

-
-
351-24-6
1-(3-fluorobenzylidene)-2-phenylhydrazine

| Conditions | Yield |
|---|---|
| In ethanol for 0.00833333h; Sonication; | 99% |
| With Fe(+3)-montmorillonite K10 |
3-Fluorobenzaldehyde Specification
The m-Fluorobenzaldehyde, with the CAS registry number 456-48-4, is also known as Benzaldehyde, 3-fluoro-. Its molecular formula is C7H5FO and its IUPAC name is 3-fluorobenzaldehyde. This chemical is clear colorless to yellow liquid which should be kept cool and dry. Keep it away from the oxides. Additionally, its product categories are Aromatic Aldehydes & Derivatives (substituted); Benzaldehyde; Fluorobenzene; Adehydes, Acetals & Ketones; Fluorine Compounds; Aldehydes; C7; Carbonyl Compounds.
Other characteristics of the m-Fluorobenzaldehyde can be summarised as followings: (1)ACD/LogP: 1.89; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.89; (4)ACD/LogD (pH 7.4): 1.89; (5)#H bond acceptors: 1; (6)#H bond donors: 0; (7)#Freely Rotating Bonds: 1; (8)Polar Surface Area: 17.07 Å2; (9)Index of Refraction: 1.539; (10)Molar Refractivity: 32.99 cm3; (11)Molar Volume: 105.3 cm3; (12)Polarizability: 13.08×10-24cm3; (13)Surface Tension: 36.8 dyne/cm; (14)Density: 1.178 g/cm3; (15)Flash Point: 56.7 °C; (16)Enthalpy of Vaporization: 40.96 kJ/mol; (17)Boiling Point: 173.2 °C at 760 mmHg; (18)Vapour Pressure: 1.28 mmHg at 25°C.
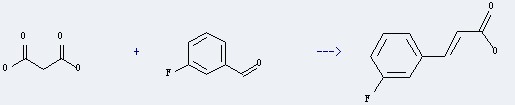
Uses of the m-Fluorobenzaldehyde: It could react with malonic acid to obtain the 3-fluoro-trans-cinnamic acid. This reaction needs the reagent of piperidine, and the solvent of pyridine. The yield is 80 %. In addition, this reaction needs the condition of heating.
When you are using this chemical, please be cautious about it as the following: This chemical is irritating to eyes, respiratory system and skin. You should wear suitable protective clothing, gloves and eye/face protection to avoid contact with skin and eyes. In case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
1.SMILES: O=Cc1cc(F)ccc1
2.InChI: InChI=1/C7H5FO/c8-7-3-1-2-6(4-7)5-9/h1-5H
3.InChIKey: PIKNVEVCWAAOMJ-UHFFFAOYAY
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View