-
Name
3-METHOXY-4-METHYLBENZALDEHYDE
- EINECS
- CAS No. 24973-22-6
- Article Data23
- CAS DataBase
- Density 1.062 g/cm3
- Solubility
- Melting Point 40.5-41.5 °C
- Formula C9H10O2
- Boiling Point 246.2 °C at 760 mmHg
- Molecular Weight 150.177
- Flash Point 105.7 °C
- Transport Information
- Appearance
- Safety 26-36/37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms m-Anisaldehyde,4-methyl- (7CI,8CI);3-Methoxy-p-tolualdehyde;
- PSA 26.30000
- LogP 1.81610
Synthetic route
-
-
4685-50-1
3-methoxy-4-methylbenzyl alcohol

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| With Dess-Martin periodane In dichloromethane at 0℃; for 0.5h; | 100% |
| With manganese(IV) oxide In tetrahydrofuran for 2h; | 95% |
| With manganese(IV) oxide In dichloromethane for 2h; Heating; | 90% |
-
-
57295-30-4
3-hydroxy-4-methylbenzaldehyde

-
-
77-78-1
dimethyl sulfate

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 45℃; | 91% |
| With potassium hydroxide | 87% |
-
-
57295-30-4
3-hydroxy-4-methylbenzaldehyde

-
-
74-88-4
methyl iodide

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate | 91% |
| Stage #1: 3-hydroxy-4-methylbenzaldehyde With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: methyl iodide In N,N-dimethyl-formamide at 20℃; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-methoxy-benzaldehyde With 1-methyl-piperazine; n-butyllithium In tetrahydrofuran; hexane at -20℃; for 0.5h; Stage #2: With sec.-butyllithium In tetrahydrofuran; hexane; cyclohexane for 0.166667h; Stage #3: methyl iodide In tetrahydrofuran; hexane; cyclohexane at -78 - 20℃; Further stages.; | 29% |
| Yield given. Multistep reaction; |
-
-
123-56-8
Succinimide

-
-
57295-33-7
(2-Bromo-5-methoxy-4-methyl-phenyl)-methanol

-
A
-
57295-32-6
2-Brom-5-methoxy-4-methylbenzaldehyd

-
B
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
C
-
90163-04-5
N-(2-Formyl-4-methoxy-5-methylphenyl)succinimid

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium hydride 1) dimethylacetamide, 2.,5-3 h, r.t., 2) 6 h, reflux; Yield given. Multistep reaction. Yields of byproduct given; | |
| With copper(l) iodide; sodium hydride 1) dimethylacetamide, 2.5-3 h, r.t., 2) 6 h, reflux; Yield given. Multistep reaction. Yields of byproduct given; | |
| With copper(l) iodide; sodium hydride 1) dimethylacetamide, 2.5-3 h, r.t., 2) 6 h, reflux; Yield given. Multistep reaction. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| (i) aq. NH2OH*HCl, NaOAc, (ii) CuSO4, Na2SO3, aq. NaOAc, (iii) /BRN= 742602/, NaNO2, aq. HCl; Multistep reaction; |
-
-
7151-68-0
3-methoxy-p-toluic acid

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| (i) SOCl2, (ii) Li; Multistep reaction; | |
| Multi-step reaction with 2 steps 1: LiAlH4 / tetrahydrofuran / 20 °C 2: MnO2 / CHCl3 / 3 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: 91 percent / acetic acid / diethyl ether 2: 88 percent / LiAlH4 / diethyl ether / 2.5 h / Heating 3: 90 percent / active MnO2 / CH2Cl2 / 2 h / Heating View Scheme |

-
-
74-88-4
methyl iodide

-
A
-
56724-03-9
3-methoxy-2-methyl-benzaldehyde

-
B
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride; N,N,N,N,-tetramethylethylenediamine; sec.-butyllithium 1) ether, cyclohexane, -72 deg C, 1 h; 2) ether, cyclohexane, -72 deg C --> rt; 3) THF, reflux, 2 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
586-30-1
3-hydroxy-4-methylbenzoic acid

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: K2CO3 / acetone / 10 h / Heating 2: LiAlH4 / tetrahydrofuran / 20 °C 3: MnO2 / CHCl3 / 3 h / Heating View Scheme |
-
-
31680-07-6
4-methyl-3-nitrobenzaldehyde

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: 91 percent / 33 percent KOH / 45 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 91 percent / KNO3 / H2SO4 3: 91 percent / 33 percent KOH / 45 °C View Scheme |
-
-
3556-83-0
methyl 4-methyl-3-methoxybenzoate

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 88 percent / LiAlH4 / diethyl ether / 2.5 h / Heating 2: 90 percent / active MnO2 / CH2Cl2 / 2 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: lithium aluminium tetrahydride / tetrahydrofuran / 1 h / 25 °C 2: manganese(IV) oxide / tetrahydrofuran / 2 h View Scheme | |
| Multi-step reaction with 2 steps 1: diisobutylaluminium hydride / tetrahydrofuran / 5.5 h / -78 - 20 °C 2: pyridinium chlorochromate / dichloromethane / 2 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: diisobutylaluminium hydride / tetrahydrofuran / 5 h / -78 - 20 °C 2: pyridinium chlorochromate / dichloromethane / 2 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: lithium aluminium tetrahydride / tetrahydrofuran / 4 h / 0 - 20 °C / Inert atmosphere 2: manganese(IV) oxide / chloroform / 18 h / 20 °C View Scheme |
-
-
108-48-5
2,6-dimethylpyridine

-
-
87808-44-4
3-methoxy-4-methylbenzoyl chloride

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| palladium In tetrahydrofuran |
-
-
141-82-2
malonic acid

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
132980-20-2
3-(3-Methoxy-4-methylphenyl)-2-propenoic acid

| Conditions | Yield |
|---|---|
| Stage #1: malonic acid; 3-methoxy-4-methylbenzaldehyde With piperidine; pyridine for 2.5h; Heating / reflux; Stage #2: With hydrogenchloride In water | 100% |
| In piperidine; pyridine for 2.5h; Heating / reflux; | 100% |
| With piperidine; pyridine for 5h; Heating; | 99.7% |

| Conditions | Yield |
|---|---|
| With piperidine; pyridine for 2.5h; Heating / reflux; Neat (no solvent); | 100% |
| With piperidine; pyridine for 2.5h; Heating / reflux; | 100% |
| With piperidine; pyridine at 100℃; Condensation; |
-
-
141-82-2
malonic acid

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: malonic acid; 3-methoxy-4-methylbenzaldehyde With piperidine; pyridine for 11h; Heating / reflux; Stage #2: With hydrogenchloride In water | 100% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| 99% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
4685-50-1
3-methoxy-4-methylbenzyl alcohol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol | 98.7% |

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In benzene for 2h; Knoevenagel Condensation; Dean-Stark; Reflux; | 92% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
108-59-8
malonic acid dimethyl ester

-
-
130921-06-1
Dimethyl 3-Methoxy-4-methylbenzylidenemalonate

| Conditions | Yield |
|---|---|
| With piperidine; benzoic acid In benzene Heating; | 91% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
126-81-8
dimedone

-
-
135-19-3
β-naphthol

-
-
1427548-18-2
C27H26O3

| Conditions | Yield |
|---|---|
| With (2,3,4,5,6-pentafluorophenyl)ammonium triflate In toluene at 50℃; for 6h; Green chemistry; | 90% |

-
-
2942-13-4
6-methoxy-1,3-benzothiazole

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
1361253-14-6
(6-methoxy-1,3-benzothiazol-2-yl)(4-methyl-3-methoxyphenyl)methanol

| Conditions | Yield |
|---|---|
| Stage #1: 6-methoxy-1,3-benzothiazole With n-butyllithium In tetrahydrofuran; hexane for 1h; Cooling with acetone-dry ice; Stage #2: 3-methoxy-4-methylbenzaldehyde In tetrahydrofuran; hexane for 2h; Cooling with acetone-dry ice; | 89% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
100-06-1
1-(4-methoxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| Stage #1: 1-(4-methoxyphenyl)ethanone With lithium hydroxide monohydrate In ethanol at 20℃; for 0.25h; Stage #2: 3-methoxy-4-methylbenzaldehyde In ethanol at 20℃; for 24h; | 88% |
-
-
79-24-3
Nitroethane

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
132980-18-8
1-(3-methoxy-4-methylphenyl)-2-nitropropene

| Conditions | Yield |
|---|---|
| With ammonium acetate for 2.5h; Heating; | 83.5% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
96-34-4
methyl chloroacetate

-
-
122333-96-4
methyl (3-methoxy-4-methylphenyl)glycidate

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol 1) -5 deg C, 2 h, 2) room temperature, 3 h; | 80% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
2065-66-9
methyl-triphenylphosphonium iodide

-
-
501371-37-5
2-methoxy-1-methyl-4-vinylbenzene

| Conditions | Yield |
|---|---|
| Stage #1: methyl-triphenylphosphonium iodide With sodium hexamethyldisilazane In tetrahydrofuran at 25℃; for 1h; Inert atmosphere; Stage #2: 3-methoxy-4-methylbenzaldehyde In tetrahydrofuran at 25℃; for 1h; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With β‐cyclodextrin In water at 80℃; for 0.633333h; | 78.5% |

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
16640-68-9
cyanomethylene triphenylphosphorane

-
-
913073-72-0
3-(3-methoxy-4-methylphenyl)acrylonitrile

| Conditions | Yield |
|---|---|
| 77% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
873-77-8
(4-chlorphenyl)magnesium bromide

-
-
1096987-59-5
(4-chlorophenyl)(3-methoxy-4-methylphenyl)methanone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 6h; Reflux; | 77% |
-
-
75-52-5
nitromethane

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
1391042-74-2
3-methoxy-4-methyl-1-[(E)-2′-nitrovinyl]benzene

| Conditions | Yield |
|---|---|
| With ammonium acetate In toluene for 18h; Inert atmosphere; Reflux; | 73% |
-
-
867-13-0
diethoxyphosphoryl-acetic acid ethyl ester

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
134058-08-5
3-(3-methoxy-4-methylphenyl)-2-propenoic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran Inert atmosphere; Stage #2: 3-methoxy-4-methylbenzaldehyde In tetrahydrofuran at 20℃; for 20h; Inert atmosphere; | 63% |
| Stage #1: diethoxyphosphoryl-acetic acid ethyl ester With sodium hydride In tetrahydrofuran; hexane for 0.416667h; Inert atmosphere; Stage #2: 3-methoxy-4-methylbenzaldehyde In tetrahydrofuran; hexane at 20℃; for 20.5h; | 63% |
| With sodium hydride 1.) toluene, 0 deg C, 1 h, 2.) toluene, THF, RT, 20 h; Yield given. Multistep reaction; |
-
-
21938-30-7
N-(5,6-Dihydro-4H-cyclopentathiazol-2-yl)-N'-isopropylidene-hydrazine

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
82814-17-3
N-(5,6-Dihydro-4H-cyclopentathiazol-2-yl)-N'-[1-(3-methoxy-4-methyl-phenyl)-meth-(E)-ylidene]-hydrazine

| Conditions | Yield |
|---|---|
| In acetic acid for 0.5h; Heating; | 61% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
113296-73-4
1,4-diacetyl-3-(2,4,5-trimethoxy-3-methylphenylmethyl)-2,5-piperazinedione

-
-
113321-18-9
(Z)-1-acetyl-3-(3-methoxy-4-methylphenylmethylene)-6-(2,4,5-trimethoxy-3-methylphenylmethyl)-2,5-piperazinedione

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In N,N-dimethyl-formamide; tert-butyl alcohol for 24h; Ambient temperature; | 58% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
6882-68-4
sophoridine

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; mineral oil at 37℃; for 13h; Reflux; | 49% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
57295-30-4
3-hydroxy-4-methylbenzaldehyde

| Conditions | Yield |
|---|---|
| With boron tribromide In dichloromethane at -10 - 20℃; for 6h; | 41.4% |
| With boron tribromide In dichloromethane at 20℃; for 5h; Cooling with ice; | 41.4% |
-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
109-77-3
malononitrile

| Conditions | Yield |
|---|---|
| With L-proline In ethanol at 20℃; for 3h; Knoevenagel Condensation; Inert atmosphere; | 32% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 75℃; for 18h; | 29% |

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
74516-54-4
2-hydroxy-4-methyl-5-methoxybenzaldehyde

| Conditions | Yield |
|---|---|
| With palladium diacetate; toluene-4-sulfonic acid; 1-fluoro-2,4,6-trimethylpyridinium trifluoromethanesulfonate; acetic acid; 2-Amino-4-chlorobenzoic acid at 90℃; for 24h; Sealed tube; | 28% |
-
-
141-84-4
2-thioxo-4-thiazolidinone

-
-
24973-22-6
3-methoxy-4-methylbenzaldehyde

-
-
58264-65-6
5-(3-methoxy-4-methyl-benzylidene)-2-thioxo-thiazolidin-4-one

| Conditions | Yield |
|---|---|
| With sodium acetate In acetic acid Heating; |
3-Methoxy-4-methylbenzaldehyde Chemical Properties
Product Name: Benzaldehyde,3-methoxy-4-methyl- (CAS NO.24973-22-6)
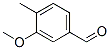
Molecular Formula: C9H10O2
Molecular Weight: 150.17g/mol
Mol File: 24973-22-6.mol
Melting Point: 40.5-41.5
Index of Refraction: 1.542
Molar Refractivity: 44.5 cm3
Molar Volume: 141.3 cm3
Surface Tension 36.1 dyne/cm
Density: 1.062 g/cm3
Flash Point: 105.7 °C
Enthalpy of Vaporization: 48.33 kJ/mol
Boiling Point: 246.2 °C at 760 mmHg
Vapour Pressure: 0.0276 mmHg at 25°C
XLogP3-AA: 2.3
H-Bond Donor: 0
H-Bond Acceptor: 2
Structure Descriptors of Benzaldehyde,3-methoxy-4-methyl- (CAS NO.24973-22-6):
IUPAC Name: 3-methoxy-4-methylbenzaldehyde
Canonical SMILES: CC1=C(C=C(C=C1)C=O)OC
InChI: InChI=1S/C9H10O2/c1-7-3-4-8(6-10)5-9(7)11-2/h3-6H,1-2H3
InChIKey: TVDHPUFLDYYBPO-UHFFFAOYSA-N
Product Categories: FINE Chemical & INTERMEDIATES; Aromatic Aldehydes & Derivatives (substituted); Aldehydes; Phenyls & Phenyl-Het; Benzaldehyde; Adehydes, Acetals & Ketones;Anisoles, Alkyloxy Compounds & Phenylacetates; Phenyls & Phenyl-Het
3-Methoxy-4-methylbenzaldehyde Safety Profile
Safety Information of Benzaldehyde,3-methoxy-4-methyl- (CAS NO.24973-22-6):
Hazard Codes: Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36/37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
3-Methoxy-4-methylbenzaldehyde Specification
Benzaldehyde,3-methoxy-4-methyl- , its CAS NO. is 24973-22-6, the synonym is 3-Methoxy-4-methylbenzaldehyde .
Related Products
- 3-Methoxy-4-methylbenzaldehyde
- 24973-49-7
- 24973-50-0
- 24974-71-8
- 24974-72-9
- 24974-73-0
- 24974-74-1
- 24974-75-2
- 2497-91-8
- 24979-70-2
- 24979-71-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View