 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; triethylamine In tert-butyl alcohol at 60℃; under 6000.6 Torr; | 96.5% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; sodium nitrite In water at 30℃; for 3h; Reagent/catalyst; Temperature; | 84% |
| With sodium nitrite In water at 60℃; | 75% |
| With water; sodium carbonate; sodium nitrite zuletzt bei 120grad; |

| Conditions | Yield |
|---|---|
| With nitronium tetrafluoborate In sulfolane at 80℃; for 2h; Product distribution; Mechanism; other alkyl- and allylsilanes; other temperature, var. solvents; | A 80% B n/a |
-
-
2009-83-8
6-chloro-1-hexanol

-
-
2483-57-0
methyl 2-nitroacetate

-
A
-
75-52-5
nitromethane

-
B
-
124-38-9
carbon dioxide

| Conditions | Yield |
|---|---|
| With di(n-butyl)tin oxide at 100℃; for 1.5h; Sealed tube; Inert atmosphere; | A n/a B n/a C 76% |


| Conditions | Yield |
|---|---|
| With di(n-butyl)tin oxide at 100℃; for 1.5h; Catalytic behavior; Reagent/catalyst; Temperature; Sealed tube; Inert atmosphere; | A n/a B n/a C 69% |


| Conditions | Yield |
|---|---|
| With tetrabutylammonium nitrite In chloroform-d1 at 20℃; | A 67% B 28 % Spectr. |
| With silver(I) nitrite | |
| With silver nitrate |
-
-
6032-29-7
(+/-)-2-pentanol

-
-
2483-57-0
methyl 2-nitroacetate

-
A
-
75-52-5
nitromethane

-
B
-
124-38-9
carbon dioxide

| Conditions | Yield |
|---|---|
| With di(n-butyl)tin oxide at 100℃; for 1.5h; Sealed tube; Inert atmosphere; | A n/a B n/a C 66% |

-
-
2483-57-0
methyl 2-nitroacetate

-
-
107-18-6
allyl alcohol

-
A
-
75-52-5
nitromethane

-
B
-
124-38-9
carbon dioxide

-
C
-
221116-46-7
allyl 2-nitroacetate

| Conditions | Yield |
|---|---|
| With di(n-butyl)tin oxide at 100℃; for 1.5h; Sealed tube; Inert atmosphere; | A n/a B n/a C 66% |

-
-
64-17-5
ethanol

-
-
2483-57-0
methyl 2-nitroacetate

-
A
-
626-35-7
nitroacetic acid ethyl ester

-
B
-
75-52-5
nitromethane

-
C
-
124-38-9
carbon dioxide

| Conditions | Yield |
|---|---|
| With di(n-butyl)tin oxide at 100℃; for 1.5h; Catalytic behavior; Reagent/catalyst; Temperature; Sealed tube; Inert atmosphere; | A 61% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With tetrabutylammonium nitrite In chloroform-d1 at 20℃; | A 60% B 29 % Spectr. |
-
-
22755-24-4
sodium salt of nitromethane

-
A
-
75-52-5
nitromethane

-
B
-
1493-05-6
difluoro-nitro-methane

-
C
-
21824-10-2
fluorotrinitromethane

| Conditions | Yield |
|---|---|
| With calcium fluoride; fluorine at -70 - -65℃; | A 24% B 20% C 45% |


| Conditions | Yield |
|---|---|
| With tetrabutylammonium nitrite In chloroform-d1 at 20℃; | A 41% B 41 % Spectr. |

| Conditions | Yield |
|---|---|
| With tetrabutylammonium nitrite In chloroform-d1 at 20℃; | A 38% B 56 % Spectr. |

| Conditions | Yield |
|---|---|
| With calcium fluoride; fluorine at -80 - -75℃; | A 24% B 20% C 23% |


| Conditions | Yield |
|---|---|
| With Cu(MeNO2)2+ Thermodynamic data; free energie for exchange of both ligands; | |
| With water; sodium hydrogencarbonate; iron(II) sulfate; sodium nitrite | |
| With sulfuric acid; sodium nitrite |

| Conditions | Yield |
|---|---|
| With sodium nitrite bei pH 6-10; |

| Conditions | Yield |
|---|---|
| With sodium nitrite bei pH 6-10; |

| Conditions | Yield |
|---|---|
| With nitric acid at 400℃; Nebenprod.2:Nitroaethan; |

| Conditions | Yield |
|---|---|
| With magnesium hydrosilicate at 120 - 130℃; |

| Conditions | Yield |
|---|---|
| at 160 - 219℃; Kinetics; unter vermindertem Druck.Pyrolysis; |


| Conditions | Yield |
|---|---|
| at 95℃; unter vermindertem Druck.Photolysis; |

-
-
34557-54-5
methane

-
-
75-52-5
nitromethane

| Conditions | Yield |
|---|---|
| With nitric acid at 475℃; | |
| With nitric acid | |
| With oxygen; nitric acid |

| Conditions | Yield |
|---|---|
| at 420℃; beim Nitrieren in der Dampfphase; |

| Conditions | Yield |
|---|---|
| bei der Nitrierung; |

| Conditions | Yield |
|---|---|
| bei der Nitrierung; | |
| With nitric acid | |
| With oxygen; nitric acid | |
| With nitric acid; acetic acid In water at 235℃; under 72402.6 Torr; for 0.05h; |

| Conditions | Yield |
|---|---|
| bei der Nitrierung; |

| Conditions | Yield |
|---|---|
| bei der Nitrierung; |

| Conditions | Yield |
|---|---|
| bei der Nitrierung; |

| Conditions | Yield |
|---|---|
| With water at 130 - 150℃; | |
| at 245.6℃; under 55 Torr; Kinetics; Further Variations:; Pressures; Temperatures; also in the presence of free radical inhibitors; |
-
-
625-75-2
nitroacetic acid

-
A
-
75-52-5
nitromethane

-
B
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

| Conditions | Yield |
|---|---|
| at 25℃; Geschwindigkeit in saurer Loesung bei verschiedenen pH und Einfluss von verschiedenen anorganischen und organischen Salzen; | |
| at 15℃; Geschwindigkeit in saurer Loesung bei verschiedenen pH und Einfluss von verschiedenen anorganischen und organischen Salzen; | |
| at 15℃; Geschwindigkeit in saurer Loesung bei verschiedenen pH und Einfluss von Glucose und Rohrzucker; |
-
-
4397-53-9
p-benzyloxybenzaldehyde

-
-
75-52-5
nitromethane

-
-
2982-55-0
(E)-1-(benzyloxy)-4-(2-nitrovinyl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: p-benzyloxybenzaldehyde; nitromethane With sodium methylate In methanol at 0 - 20℃; for 0.166667h; Stage #2: With hydrogenchloride; water In methanol at 0 - 20℃; for 0.25h; | 100% |
| Stage #1: p-benzyloxybenzaldehyde; nitromethane With sodium methylate In methanol at 0 - 20℃; for 0.166667h; Stage #2: With hydrogenchloride In methanol; water at 0 - 20℃; for 0.25h; | 100% |
| Stage #1: p-benzyloxybenzaldehyde; nitromethane With sodium methylate In methanol at 0 - 20℃; Stage #2: With hydrogenchloride In methanol; water at 0 - 20℃; | 100% |
-
-
75-52-5
nitromethane

-
-
623-43-8
crotonic acid methyl ester

-
-
16507-06-5
methyl 3-methyl-4-nitro-butyrate

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethylguanidine at 20℃; for 576h; | 100% |
| With DBN In methanol at 60℃; for 6h; Inert atmosphere; | 71.44% |
| With methanol; potassium carbonate | |
| With N-benzyl-trimethylammonium hydroxide In methanol | |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol Michael addition; |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran; butan-1-ol at 23℃; Inert atmosphere; | 100% |
| With silica gel; triethylamine at 20℃; for 3h; Henry Nitro Aldol Condensation; | 95% |
| With potassium tert-butylate In tetrahydrofuran; tert-butyl alcohol at 20℃; for 18.5h; Henry reaction; | 94% |
-
-
75-52-5
nitromethane

-
-
5417-17-4
2-chloro-3,4-dimethoxybenzaldehyde

-
-
41122-35-4
(E)-2-chloro-3,4-dimethoxy-1-(2-nitrovinyl)benzene

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 4h; Inert atmosphere; Reflux; | 100% |
| With ammonium acetate In acetic acid for 3h; Heating; | 95% |
| With ammonium acetate; acetic acid |

| Conditions | Yield |
|---|---|
| With C62H54N2O2; copper diacetate In ethanol at 10℃; for 48h; Henry Nitro Aldol Condensation; enantioselective reaction; | 100% |
| With C62H48CuN2O2; copper diacetate; benzaldehyde In ethanol at 10℃; for 48h; Reagent/catalyst; Henry Nitro Aldol Condensation; | 100% |
| With (2-hydroxyethyl)ammonium formate at 20℃; for 0.8h; Knoevenagel condensation; Ionic liquid; | 95% |

| Conditions | Yield |
|---|---|
| With copper(II) ferrite In dimethyl sulfoxide at 120℃; for 0.0333333h; Microwave irradiation; | 100% |
| With bridged diamino-functionalized periodic mesoporous organosilicas with high nitrogen contents at 90℃; for 5h; Henry Nitro Aldol Condensation; | 99% |
| With acetic acid; benzylamine at 78 - 80℃; for 3.5h; Product distribution / selectivity; | 97.1% |

| Conditions | Yield |
|---|---|
| With polymer supported 4-DMAP at 20℃; for 0.416667h; Reagent/catalyst; Henry Nitro Aldol Condensation; | 100% |
| With triethylamine at 80℃; for 0.0166667h; Henry reaction; microwave irradiation; | 99% |
| With Musca acuminata (banana) peel ash In neat (no solvent) at 20℃; for 0.333333h; Henry Nitro Aldol Condensation; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With C57H78N12; copper(II) acetate monohydrate In methanol; dichloromethane for 72h; Henry reaction; Inert atmosphere; | 100% |
| With polymer supported 4-DMAP at 20℃; for 0.333333h; Reagent/catalyst; Henry Nitro Aldol Condensation; | 100% |
| With P(i-PrNCH2CH2)3N; magnesium sulfate for 0.0833333h; Ambient temperature; | 99% |
-
-
75-52-5
nitromethane

-
-
100-52-7
benzaldehyde

-
-
141377-54-0, 145920-96-3, 149495-00-1, 15990-45-1
2-nitro-1-phenylethan-1-ol

| Conditions | Yield |
|---|---|
| palladium/alumina at 60℃; for 1h; | 100% |
| With 1-butyl-4-aza-1-azoniabicyclo[2.2.2]octane hydroxide In neat (no solvent) at 20℃; for 0.166667h; Reagent/catalyst; Henry Nitro Aldol Condensation; Green chemistry; | 99% |
| With 1-butyl-4-aza-1-azoniabicyclo[2.2.2]octane hydroxide In neat (no solvent) at 20℃; for 0.166667h; Catalytic behavior; Reagent/catalyst; Henry Nitro Aldol Condensation; Ionic liquid; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 0℃; for 0.166667h; Addition; Henry reaction; | 100% |
| With P(i-PrNCH2CH2)3N; magnesium sulfate for 0.666667h; Ambient temperature; | 98% |
| With rac-1',2',3',4'-tetrahydro-1,1'-bisisoquinoline In tetrahydrofuran at 20℃; for 24h; Henry Nitro Aldol Condensation; | 98% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran; tert-butyl alcohol at 0 - 20℃; for 24h; Henry reaction; | 100% |
| With potassium tert-butylate In tetrahydrofuran; tert-butyl alcohol Addition; | 99% |
| With (S,S)-1,2-diaminocyclohexane; copper(II) acetate monohydrate In ethanol at 20℃; for 12h; Henry reaction; Inert atmosphere; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol Michael condensation; Reflux; | 100% |
| With alkaline hydroxide In ethanol at 60℃; for 12h; Michael addition; | 100% |
| With potassium fluoride In ethanol at 20℃; for 2h; Addition; | 98% |
-
-
75-52-5
nitromethane

-
-
119-67-5
o-carboxybenzaldehyde

-
-
3598-68-3
3-(nitromethyl)-isobenzofuran-1-(3H)-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 15 - 30℃; for 1h; | 100% |
| With alkaline solution |
-
-
75-52-5
nitromethane

-
-
2043-61-0
cyclohexanecarbaldehyde

-
-
141377-56-2, 141434-99-3, 149559-83-1, 50598-90-8
1-cyclohexyl-2-nitroethanol

| Conditions | Yield |
|---|---|
| tris(bis(trimethylsilyl)amido)samarium(III) In tetrahydrofuran for 25h; Ambient temperature; | 100% |
| With potassium fluoride In isopropyl alcohol at 0 - 20℃; for 24h; Inert atmosphere; | 100% |
| With calcium oxide at 50℃; for 12h; | 99% |
-
-
75-52-5
nitromethane

-
-
108-94-1
cyclohexanone

-
-
100-53-8
phenylmethanethiol

-
-
335458-24-7
1-benzylthio-1-nitromethylcyclohexane

| Conditions | Yield |
|---|---|
| With piperidine In benzene | 100% |
| With piperidine In acetonitrile for 4h; Heating; | 96% |
| With piperidine In acetonitrile for 4h; Heating / reflux; | 22% |
| With piperidine In benzene Heating; |

-
-
75-52-5
nitromethane

-
-
120-92-3
cyclopentanone

-
-
100-53-8
phenylmethanethiol

-
-
335458-23-6
1-benzylthio-1-nitromethylcyclopentane

| Conditions | Yield |
|---|---|
| With piperidine In benzene | 100% |
| With piperidine In acetonitrile for 4h; Heating; | 94% |
| With piperidine In benzene Heating; |

-
-
104-53-0
3-phenyl-propionaldehyde

-
-
75-52-5
nitromethane

-
-
141377-53-9, 141434-97-1, 149494-99-5, 85290-51-3
1-nitro-4-phenylbutan-2-ol

| Conditions | Yield |
|---|---|
| (anthracenebisresorcinol(4-):2La(3+))polymer In benzene at 20℃; for 0.333333h; Addition; nitroaldol reaction; | 100% |
| With potassium fluoride In isopropyl alcohol at 0 - 20℃; for 24h; Inert atmosphere; | 100% |
| With silica gel; triethylamine at 20℃; for 2h; Catalytic behavior; Reagent/catalyst; Henry Nitro Aldol Condensation; | 99% |
-
-
75-52-5
nitromethane

-
-
133157-50-3
2-Formyl-2,3-dihydronaphtho<1,8-bc>pyran

-
-
133157-51-4
1-(1-Hydroxy-2-nitroethyl)-2,3-dihydronaphtho<1,8-bc>pyran

| Conditions | Yield |
|---|---|
| With triethylamine 1) 15 min, cooling 2) 2 h, r.t.; | 100% |
-
-
75-52-5
nitromethane

-
-
127181-13-9
(+)-(4S,5S,6R)-6-acetamido-N-(benzyloxycarbonyl)-4,5-(isopropylidenedioxy)-3-piperidinone

-
-
141781-97-7
(-)-(2R,3S,4S,5R)-2-acetamido-N-(benzyloxycarbonyl)-3,4-O-isopropylidene-5-(nitromethyl)-3,4,5-piperidinetriol

| Conditions | Yield |
|---|---|
| With sodium hydride In 1,2-dimethoxyethane for 2h; Ambient temperature; | 100% |
-
-
75-52-5
nitromethane

-
-
94-41-7
benzalacetophenone

-
A
-
6277-67-4, 68047-61-0, 122348-60-1, 77670-36-1
(R)-1,3-diphenyl-4-nitrobutane-1-one

-
B
-
6277-67-4, 77670-36-1, 122348-60-1, 68047-61-0
(S)-4-nitro-1,3-diphenylbutan-1-one

| Conditions | Yield |
|---|---|
| With (+)-quinidine-acrylonitrile copolymer In toluene under 661957 Torr; for 5h; | A n/a B 100% |
| With 1-[3,5-bis(trifluoromethyl)phenyl]-3-[(1S,2S)-2-(dimethylamino)cyclohexyl]thiourea In toluene at 20℃; for 192h; Kinetics; Reagent/catalyst; Michael addition; enantioselective reaction; | |
| Stage #1: nitromethane With (1R,1'R,2S,2'S)-1,1'-(propane-1,3-diyl)bis(2-((2,6-diisopropylphenyl)carbamoyl)pyrrolidine-1-oxide); ytterbium(III) triflate In tetrahydrofuran at 30℃; for 0.5h; Asymmetric Michael addition; Inert atmosphere; Stage #2: benzalacetophenone With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 30℃; for 48h; Asymmetric Michael addition; Inert atmosphere; optical yield given as %ee; enantioselective reaction; |

-
-
75-52-5
nitromethane

-
-
529-20-4
2-methylphenyl aldehyde

-
-
28638-59-7, 34222-71-4
methyl-2 β-nitrostyrene

| Conditions | Yield |
|---|---|
| ammonium acetate at 90℃; for 2h; | 100% |
| With sodium hydroxide |
-
-
75-52-5
nitromethane

-
-
100-53-8
phenylmethanethiol

-
-
67-64-1
acetone

-
-
105780-12-9
benzyl(2-methyl-1-nitropropan-2-yl)sulfane

| Conditions | Yield |
|---|---|
| With piperidine In benzene | 100% |
| With piperidine In tetrahydrofuran for 12h; Heating; | 96% |
| With piperidine for 15h; Heating; | 49% |
| In benzene at 100℃; for 22h; Dean-Stark; | 23% |
| With piperidine In benzene for 10h; Reflux; Inert atmosphere; |

-
-
75-52-5
nitromethane

-
-
122-57-6
1-Phenylbut-1-en-3-one

-
-
122348-55-4, 122348-59-8, 142232-08-4, 1574-86-3
5-nitro-4-phenylpentan-2-one

| Conditions | Yield |
|---|---|
| With hydrotalcite for 0.1h; Michael addition; Microwave irradiation; | 100% |
| With potassium fluoride; 18-crown-6 ether In acetonitrile for 168h; Heating; | 93% |
| With C10H20NaO5*HO(1-); water In methanol at 25℃; for 0.833333h; Michael addition; | 90% |
-
-
75-52-5
nitromethane

-
-
33985-40-9, 58056-24-9, 63029-10-7, 105368-48-7, 141610-28-8, 141724-46-1
1-O-methyl-2,3-O-isopropylidene-α-D-mannopentoaldo-1,4-furanose

-
-
85339-08-8, 85339-09-9, 118205-71-3, 118205-72-4, 124648-90-4, 129443-53-4
6-Deoxy-2,3-O-isopropylidene-6-nitro-α-D-lyxo-hexofuranoside

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 0℃; for 2h; | 100% |
| With sodium methylate In methanol at 0℃; for 2h; |
-
-
75-52-5
nitromethane

-
-
60656-87-3
benzyloxyacetoaldehyde

-
-
169139-89-3
(+/-)-3-benzyloxy-1-nitropropan-2-ol

| Conditions | Yield |
|---|---|
| With potassium fluoride In isopropyl alcohol at 20℃; for 15h; Addition; Henry reaction; | 100% |
| With aluminum oxide at 21℃; for 24h; | 98% |
| With potassium hydroxide In methanol at 20℃; for 1.5h; | 88% |
-
-
75-52-5
nitromethane

-
-
201684-34-6
Toluene-4-sulfonic acid 5-bromo-3-formyl-1H-indol-7-yl ester

-
-
126260-59-1
(E)-5-bromo-3-nitrovinyl-7-(p-toluenesulfonyloxy)indole

| Conditions | Yield |
|---|---|
| With ammonium acetate for 3h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| aluminum oxide at 90℃; for 0.15h; | 100% |
| With piperazine-grafted reduced graphene oxide (rGO-NH) In ethanol at 60℃; for 8h; Reagent/catalyst; Michael Addition; | 72% |
| With aluminum oxide at 20 - 90℃; for 0.2h; microwave irradiation; | 63% |
| at 60℃; for 4h; Michael Addition; | |
| With quinidine In toluene at 20℃; |
-
-
75-52-5
nitromethane

| Conditions | Yield |
|---|---|
| aluminum oxide at 90℃; for 0.15h; | 100% |
| With aluminum oxide at 20 - 90℃; for 0.2h; microwave irradiation; | 70% |
-
-
75-52-5
nitromethane

-
-
211935-30-7
2-(2-Benzyloxy-3-methoxyphenyl)acetaldehyde

-
-
211935-31-8
1-(2-Benzyloxy-3-methoxyphenyl)-3-nitropropan-2-ol

| Conditions | Yield |
|---|---|
| With cetyltrimethylammonim bromide In sodium hydroxide | 100% |
| With sodium hydroxide; cetyltrimethylammonim bromide Condensation; Henry condensation; | 100% |
-
-
75-52-5
nitromethane

-
-
209175-47-3
(1-Formyl-7-methoxy-naphthalen-2-yloxy)-acetic acid ethyl ester

-
-
209175-48-4
[7-Methoxy-1-((E)-2-nitro-vinyl)-naphthalen-2-yloxy]-acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With ammonium acetate for 3h; Heating; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

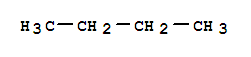














 Xn,
Xn, F,
F, Xi
Xi