-
Name
3-Cyanomethylthiophene
- EINECS 237-433-1
- CAS No. 13781-53-8
- Article Data12
- CAS DataBase
- Density 1.171 g/cm3
- Solubility
- Melting Point
- Formula C6H5NS
- Boiling Point 243.7 °C at 760 mmHg
- Molecular Weight 123.178
- Flash Point 92.4 °C
- Transport Information UN 3276 6.1/PG 2
- Appearance clear yellow to brown liquid
- Safety 26-36/37-45-36/37/39
- Risk Codes 20/21/22-36/37/38-23/24/25
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  T
T
- Synonyms 3-Cyanomethylthiophene;3-Thienylacetonitrile;
- PSA 52.03000
- LogP 1.81418
Synthetic route

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; bis(η3-allyl-μ-chloropalladium(II)) In 1,3,5-trimethyl-benzene at 20 - 140℃; for 5.16667h; Inert atmosphere; Sealed tube; chemoselective reaction; | 78% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; palladium diacetate; sodium carbonate In 1,4-dioxane; water at 100℃; for 12h; Inert atmosphere; Sealed tube; | 53% |
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; palladium diacetate; sodium carbonate In 1,4-dioxane; water at 60℃; for 12h; Inert atmosphere; Sealed tube; |

| Conditions | Yield |
|---|---|
| tetrabutylammomium bromide In dichloromethane; water Heating / reflux; | 44% |
| With ethanol |

| Conditions | Yield |
|---|---|
| With N-benzyl-N,N,N-triethylammonium chloride In water | 28% |

| Conditions | Yield |
|---|---|
| With ethanol |
-
-
498-62-4
3-thiophene carboxaldehyde

-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With potassium tert-butylate 1.) THF, -20 deg C, 30 min, 2.) THF, -20 deg C, 30 min; Yield given. Multistep reaction; |
-
-
81344-59-4
Methyl 2-cyano-2-(3-thienyl)acetate

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With water; triethylamine Product distribution; |
-
-
616-44-4
3-Methylthiophene

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) / tetrachloromethane / 1 h / Reflux 2: tetrabutyl ammonium fluoride / acetonitrile; tetrahydrofuran / 0 - 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: N-Bromosuccinimide; dibenzoyl peroxide 2: N-benzyl-N,N,N-triethylammonium chloride / water View Scheme |
-
-
34846-44-1
3-Bromomethylthiophene

-
-
7677-24-9
trimethylsilyl cyanide

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran; acetonitrile at 0 - 20℃; Inert atmosphere; | 4.06 g |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| With 2,4-dichlorophenoxyacetic acid dimethylamine In toluene Heating; | 100% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
67-56-1
methanol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In diethyl ether at 0 - 5℃; | 99% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
529-20-4
2-methylphenyl aldehyde

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol | 98% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
59311-67-0
2-(3-thienyl)ethylamine

| Conditions | Yield |
|---|---|
| With indium(III) chloride; sodium tetrahydroborate In tetrahydrofuran at 25℃; for 4h; Inert atmosphere; | 97% |
| With diisobutylaluminum borohydride In tetrahydrofuran at 25℃; for 1h; Inert atmosphere; | 71% |
| With indium(III) chloride; sodium tetrahydroborate In tetrahydrofuran at 25℃; for 16h; Inert atmosphere; | 67% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
137-07-5
2-amino-benzenethiol

-
-
1427180-57-1
2-((thiophen-3-yl)methyl)benzo[d]thiazole

| Conditions | Yield |
|---|---|
| With copper diacetate; triethylamine In ethanol at 70℃; for 6h; | 97% |
-
-
98-03-3
thiophene-2-carbaldehyde

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
13781-61-8
(Z)-3-Thiophen-2-yl-2-thiophen-3-yl-acrylonitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 30℃; Kinetics; Thermodynamic data; ΔE*, Σ*, A; | 95% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
100-52-7
benzaldehyde

-
-
13781-54-9
(E)-3-phenyl-2-(3-thienyl)acrylonitrile

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 30℃; Kinetics; Thermodynamic data; ΔE*, Σ*, A; | 95% |
| With sodium ethanolate In ethanol | 94% |
| With potassium tert-butylate In ethanol Heating; | 60% |
| With potassium tert-butylate In ethanol at 20℃; Knoevenagel condensation; | 60% |
| With sodium ethanolate In ethanol at 4℃; for 8h; | 51% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
1942-45-6
4-Octyne

-
-
1180490-30-5
(Z)-2-propyl-3-(3-thienylmethyl)-2-hexenenitrile

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel(0); dimethylaluminum chloride; 2-dicyclohexylphosphino-2',4',6'-trimethylbiphenyl In hexane; toluene at 80℃; for 2h; Inert atmosphere; stereoselective reaction; | 95% |
| With bis(1,5-cyclooctadiene)nickel (0); dimethylaluminum chloride; 2-dicyclohexylphosphino-2',4',6'-trimethylbiphenyl In hexane; tetradecane; toluene at 80℃; for 2h; Inert atmosphere; regioselective reaction; | 95% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
34843-84-0
2-(thiophen-3-yl) ethanamine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With borane-dimethyl sulfide complex In tetrahydrofuran for 16h; Reflux; Stage #2: With hydrogenchloride In tetrahydrofuran; water at 20℃; for 0.333333h; | 94% |
| Stage #1: (thiophen-3-yl)acetonitrile With dimethylsulfide borane complex In tetrahydrofuran for 16h; Heating / reflux; Stage #2: With methanol In tetrahydrofuran | 94% |
| Stage #1: (thiophen-3-yl)acetonitrile With dimethylsulfide borane complex In tetrahydrofuran for 16h; Heating / reflux; Stage #2: With hydrogenchloride; methanol at 20℃; for 0.333333h; | 94% |
| Stage #1: (thiophen-3-yl)acetonitrile With dimethylsulfide borane complex In tetrahydrofuran for 12h; Reflux; Stage #2: With hydrogenchloride In tetrahydrofuran for 0.5h; | 78.5% |
| Stage #1: (thiophen-3-yl)acetonitrile With borane-THF In tetrahydrofuran at 20℃; Inert atmosphere; Reflux; Stage #2: With methanol In tetrahydrofuran Stage #3: With hydrogenchloride In 1,4-dioxane; methanol |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With sodium azide; triethylamine hydrochloride In toluene at 110℃; for 16h; Inert atmosphere; | 93% |
| With aluminium trichloride; sodium azide In tetrahydrofuran for 48h; Heating; | 61% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
17649-86-4
4,4-bis(methylthio)but-3-en-2-one

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With sodium hydride In N,N-dimethyl-formamide; benzene for 0.25h; Metallation; Stage #2: 4,4-bis(methylthio)but-3-en-2-one In N,N-dimethyl-formamide at 20℃; Substitution; | 93% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
64-17-5
ethanol

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 0℃; Pinner reaction; | 93% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
113350-36-0, 102382-37-6
bis(acetonitrile)diiodotricarbonyltungsten(II)

-
-
603-35-0
triphenylphosphine

-
-
211744-19-3
WI2(CO)3(thiophene-3-acetonitrile)(PPh3)

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; stirring W-complex with equimolar amt. of PPh3 for 3 min,filtration into equimolar amt. of ligand soln., stirring for 30 min; solvent removal (vac.); elem. anal.; | 93% |

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; toluene-4-sulfonic acid In ethanol at 50℃; for 0.166667h; Green chemistry; regioselective reaction; | 93% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
298-12-4
Glyoxilic acid

-
-
1076169-87-3
(2Z)-3-cyano-3-(3-thienyl)acrylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol for 3h; Knoevenagel Condensation; Reflux; | 90% |
| With potassium carbonate In methanol for 3h; Heating / reflux; | 90% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
74-88-4
methyl iodide

-
-
88701-59-1
rac-2-(thiophen-2-yl)propanenitrile

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; | 90% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
772-31-6
cyclopropyl(4-fluorophenyl)methanone

-
-
1429308-76-8
9a-(4-fluorophenyl)-7,8,9,9a-tetrahydrothieno[3,2-g]indolizin-5(4H)-one

| Conditions | Yield |
|---|---|
| With tributylphosphine; boron trifluoride diethyl etherate; copper(I) bromide In nitromethane at 90℃; for 18h; Ritter Amidation; | 90% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
113350-36-0, 102382-37-6
bis(acetonitrile)diiodotricarbonyltungsten(II)

-
-
211744-17-1
WI2(CO)3(thiophene-3-acetonitrile)2

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; stirring W-complex with 2 equiv. of ligand (warm water bath, 90 min, then room temp., 1 h); filtration, solvent removal (vac.); elem. anal.; | 89% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
13781-66-3
2-(thiophene-3-yl) acetamide

| Conditions | Yield |
|---|---|
| With water at 130℃; for 1h; Microwave irradiation; | 88% |
| With copper(l) iodide; caesium carbonate; 1,8-diazabicyclo[5.4.0]undec-7-ene In nitromethane; water at 20 - 100℃; for 3h; | 75% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
5874-09-9
methyl (4-methoxyphenyl)carbodithioate

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With potassium tert-butylate In 1,4-dioxane at 0℃; for 0.166667h; Stage #2: methyl (4-methoxyphenyl)carbodithioate In 1,4-dioxane at 0 - 20℃; for 1h; Stage #3: With iodine In 1,4-dioxane at 90℃; | 87% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
57908-42-6
α,α'-dichloroazo compound

| Conditions | Yield |
|---|---|
| With antimonypentachloride In dichloromethane at -60 - 23℃; for 2.16667h; | 86% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
109-94-4
formic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol for 2h; Heating; | 85% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
106-95-6
allyl bromide

-
-
62716-51-2
2-allyl-2-(thiophen-3-yl)pent-4-enenitrile

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With sodium hydride In dimethyl sulfoxide at 20℃; for 0.5h; Inert atmosphere; Stage #2: allyl bromide In dimethyl sulfoxide at 20℃; for 2h; Inert atmosphere; | 85% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
6640-25-1
4-chlorophenyl cyclopropyl ketone

-
-
1429308-77-9
9a-(4-chlorophenyl)-7,8,9,9a-tetrahydrothieno[3,2-g]indolizin-5(4H)-one

| Conditions | Yield |
|---|---|
| With tributylphosphine; boron trifluoride diethyl etherate; copper(I) bromide In nitromethane at 90℃; for 18h; Ritter Amidation; | 84% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
3481-02-5
cyclopropyl phenyl ketone

-
-
1429308-75-7
9a-phenyl-7,8,9,9a-tetrahydrothieno[3,2-g]indolizin-5(4H)-one

| Conditions | Yield |
|---|---|
| With tributylphosphine; boron trifluoride diethyl etherate; copper(I) bromide In nitromethane at 90℃; for 18h; Ritter Amidation; | 84% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
925252-85-3, 155601-61-9
N-Hydroxy-2-thiophen-3-yl-acetamidine

| Conditions | Yield |
|---|---|
| With potassium carbonate; ammonium chloride In methanol at 60℃; for 12h; | 83% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
29540-62-3
bis-(1-chloro-1-methyl-ethyl)-diazene

| Conditions | Yield |
|---|---|
| With antimonypentachloride In dichloromethane at -60 - 23℃; for 2.16667h; | 83% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
1117-71-1
methyl 4-bromocrotonate

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With lithium diisopropyl amide In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: methyl 5-(dimethylamino)thiophene-2-carbodithioate In tetrahydrofuran at 0℃; for 0.5h; Stage #3: methyl 4-bromocrotonate In tetrahydrofuran at 0 - 20℃; for 8.5h; | 82% |

-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
118-74-1
hexachlorobenzene

| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether 1.) -70 deg C, 2 h, 2.) -70 deg C to r.t.; r.t., overnight; | 80% |
-
-
13781-53-8
(thiophen-3-yl)acetonitrile

-
-
5969-47-1
methyl fluorobenzenedithiocarboxylate

| Conditions | Yield |
|---|---|
| Stage #1: (thiophen-3-yl)acetonitrile With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0℃; for 0.166667h; Stage #2: methyl fluorobenzenedithiocarboxylate In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 1h; Stage #3: With tetrabutylammomium bromide; copper diacetate; palladium diacetate In N,N-dimethyl-formamide; mineral oil at 90℃; | 80% |
| Stage #1: (thiophen-3-yl)acetonitrile With potassium tert-butylate In 1,4-dioxane at 0℃; for 0.166667h; Stage #2: methyl fluorobenzenedithiocarboxylate In 1,4-dioxane at 0 - 20℃; for 1h; Stage #3: With iodine In 1,4-dioxane at 90℃; | 77% |
3-Thiopheneacetonitrile Chemical Properties
The MF of 3-Thiopheneacetonitrile(13781-53-8): C6H5NS
The MW of 3-Thiopheneacetonitrile(13781-53-8): 123.18
EINECS: 237-433-1
bp: 124-125 °C16 mm Hg(lit.)
density: 1.08 g/mL at 25 °C(lit.)
refractive index: n20/D 1.545(lit.)
Fp: >230 °F
Appearance: Clear brown liquid.
Categories: Aromatic Nitriles;Thiophene&Benzothiophene;Nitriles;Thiophens
Synonyms: THIOPHENE-3-ACETONITRILE;3-Thienylacetonitrile;LABOTEST-BB LT00848266;3-THIOPHENEACETONITRILE;3-Cyanomethylthiophene;thiophen-3-acetonitrile;2,3-thiopheneacetonitrile;THIEN-3-YLACETONITRILE
The Structure of 3-Thiopheneacetonitrile(13781-53-8):
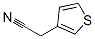
3-Thiopheneacetonitrile Safety Profile
 Xn,
Xn, T
T Risk Statements: 20/21/22-36/37/38-23/24/25
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
23/24/25: Toxic by inhalation, in contact with skin and if swallowed
Safety Statements: 26-36/37-45-36/37/39
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37: Wear suitable protective clothing and gloves
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
RIDADR: UN 3276 6.1/PG 2
WGK Germany: 3
PackingGroup: III
3-Thiopheneacetonitrile Specification
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of soap and water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Eyes:
Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
2. Handling and Storage
Storage:
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Related Products
- 3-Thiopheneacetonitrile
- 13781-67-4
- 13782-33-7
- 137823-39-3
- 137823-42-8
- 137832-55-4
- 13784-61-7
- 137848-28-3
- 137848-29-4
- 13785-65-4
- 13785-66-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View