-
Name
4-Dodecylaniline
- EINECS 203-201-3
- CAS No. 104-42-7
- Article Data19
- CAS DataBase
- Density 0.902 g/cm3
- Solubility
- Melting Point 35-39 °C(lit.)
- Formula C18H31N
- Boiling Point 364 °C at 760 mmHg
- Molecular Weight 261.451
- Flash Point 162.7 °C
- Transport Information
- Appearance WHITE TO TAN POWDER, CRYSTALS AND/OR CHUNKS
- Safety 22-24/25
- Risk Codes 22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Aniline,p-dodecyl- (6CI,7CI,8CI);(p-Aminododecyl)benzene;1-(p-Aminophenyl)dodecane;4-Dodecylbenzenamine;4-Dodecylphenylamine;4-n-Dodecylaniline;p-Dodecylaniline;
- PSA 26.02000
- LogP 6.31340
Synthetic route

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 255 - 258℃; for 21h; | 45% |
-
-
1022975-57-0
1-(dodec-1-yn-1-yl)-4-nitrobenzene

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With 5%-palladium/activated carbon; hydrogen In ethanol; ethyl acetate at 20℃; under 15001.5 Torr; for 24h; Autoclave; | 41% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 240℃; |

| Conditions | Yield |
|---|---|
| With cobalt(II) chloride at 247℃; |
-
-
101874-34-4
1-chloro-4-dodecylbenzene

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With ammonia; water; copper(l) chloride |
-
-
101778-21-6
4'-aminododecanoylphenone

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; amalgamated zinc | |
| With potassium hydroxide; hydrazine hydrate; diethylene glycol |
-
-
72617-54-0
p'-(ω-(trimethylammonio)butyloxy)-p-(dodecylbenzylidene)aniline

-
A
-
104-42-7
4-dodecylaniline

-
B
-
72617-53-9
4-(p-formylphenyloxy)butyltrimethylammonium bromide

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 30℃; Rate constant; Kinetics; ΔE(excit.); influence of pH and concentration of azometine on velocity constant; |
-
-
72621-04-6
[10-(4-{[(E)-4-Dodecyl-phenylimino]-methyl}-phenoxy)-decyl]-trimethyl-ammonium; bromide

-
A
-
104-42-7
4-dodecylaniline

-
B
-
73000-51-8
10-(p-formylphenyloxy)decyltrimethylammonium bromide

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 30℃; Rate constant; Kinetics; ΔE(excit.); influence of pH and concentration of azometine on velocity constant, var. solvents; |
-
-
70987-23-4
(4-{[(E)-4-Dodecyl-phenylimino]-methyl}-phenyl)-trimethyl-ammonium; bromide

-
A
-
104-42-7
4-dodecylaniline

-
B
-
72621-22-8
4-formyl-N,N,N-trimethyl-benzenaminium bromide

| Conditions | Yield |
|---|---|
| With phosphate buffer In water at 30℃; Rate constant; Kinetics; ΔE(excit.); influence of pH and concetration of azometine on velocity constant; |
-
-
3007-74-7
N-dodecylaniline

-
A
-
104-42-7
4-dodecylaniline

-
B
-
854306-07-3
4,N-didodecyl-aniline

-
C
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| at 250℃; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: AlCl3 2: CuCl; aqueous NH3 / 250 - 270 °C 3: amalgamated zinc; aqueous HCl View Scheme | |
| Multi-step reaction with 3 steps 1: AlCl3; CS2 2: ethanolic NaOH 3: N2H4+H2O; KOH; diethylene glycol View Scheme | |
| Multi-step reaction with 3 steps 1: aluminum (III) chloride 2: hydrazine 3: aluminum (III) chloride; acetyl chloride View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: cobalt (II)-chloride / 247 °C View Scheme |
-
-
408333-43-7
1-(4-chloro-phenyl)-dodecan-1-one

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: CuCl; aqueous NH3 / 250 - 270 °C 2: amalgamated zinc; aqueous HCl View Scheme | |
| Multi-step reaction with 4 steps 1: aluminium isopropylate; isopropyl alcohol 2: KHSO4 / 200 °C / 10 Torr 3: Raney nickel; ethanol / Hydrogenation 4: water; CuCl; NH3 View Scheme |
-
-
112508-20-0
acetic acid-(4-lauroyl-anilide)

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ethanolic NaOH 2: N2H4+H2O; KOH; diethylene glycol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: cobalt (II)-chloride / 247 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: AlCl3 2: CuCl; aqueous NH3 / 250 - 270 °C 3: amalgamated zinc; aqueous HCl View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: AlCl3; CS2 2: ethanolic NaOH 3: N2H4+H2O; KOH; diethylene glycol View Scheme |
-
-
101887-54-1
1ξ-(4-chloro-phenyl)-dodec-1-ene

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Raney nickel; ethanol / Hydrogenation 2: water; CuCl; NH3 View Scheme |
-
-
101874-35-5
1-(4-chloro-phenyl)-dodecan-1-ol

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: KHSO4 / 200 °C / 10 Torr 2: Raney nickel; ethanol / Hydrogenation 3: water; CuCl; NH3 View Scheme |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride; methyl tributylammonium chloride at 46 - 160℃; for 27.8h; |

| Conditions | Yield |
|---|---|
| Stage #1: 1-dodecylbenzene With aluminum (III) chloride; acetyl chloride Stage #2: Schmidt reaction; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrazine 2: aluminum (III) chloride; acetyl chloride View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aluminum (III) chloride 2: hydrazine 3: aluminum (III) chloride; acetyl chloride View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine / 2 h / 80 °C / Inert atmosphere 2: 5%-palladium/activated carbon; hydrogen / ethanol; ethyl acetate / 24 h / 20 °C / 15001.5 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine / 2 h / 80 °C / Inert atmosphere 2: 5%-palladium/activated carbon; hydrogen / ethanol; ethyl acetate / 24 h / 20 °C / 15001.5 Torr / Autoclave View Scheme |
-
-
40938-30-5
2-dodecylaniline

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With hydrogen sulfide |
-
-
104-42-7
4-dodecylaniline

-
-
148402-69-1
5,11,17,23-tetraformyl-25,26,27,28-tetrakis(methoxy)calix[4]arene

-
-
143519-67-9
C108H148N4O4

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In chloroform for 29h; Heating; | 100% |

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| In not given ligand reacted with Co salt instaneously; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: cycl-isopropylidene malonate; trimethyl orthoformate for 1h; Reflux; Stage #2: 4-dodecylaniline for 2h; Reflux; | 98% |


| Conditions | Yield |
|---|---|
| Stage #1: 4-dodecylaniline With 1H-imidazole; triethylamine In tetrahydrofuran for 0.166667h; Stage #2: acryloyl chloride In tetrahydrofuran at 0 - 25℃; for 15.33h; Inert atmosphere; | 95% |
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 15.16h; Inert atmosphere; | 95% |
-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With iodine; sodium hydrogencarbonate In water; toluene at 20℃; Inert atmosphere; Schlenk technique; | 94% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 18h; | 91.5% |

| Conditions | Yield |
|---|---|
| With acid | 90% |
-
-
104-42-7
4-dodecylaniline

-
-
118-91-2
ortho-chlorobenzoic acid

-
-
1373551-67-7
2-((4-dodecylphenyl)amino)benzoic acid

| Conditions | Yield |
|---|---|
| With copper diacetate; potassium carbonate In N,N-dimethyl-formamide for 3h; Inert atmosphere; Reflux; | 90% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; under 760.051 Torr; for 0.25h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-dodecylaniline With potassium carbonate In dichloromethane at 20℃; for 0.5h; Stage #2: chloroacetyl chloride In dichloromethane for 4h; Reflux; | 89% |
| Stage #1: chloroacetyl chloride With potassium carbonate In dichloromethane at 20℃; for 0.5h; Stage #2: 4-dodecylaniline In dichloromethane for 4h; Reflux; | 89% |
-
-
108-73-6
3,5-dihydroxyphenol

-
-
104-42-7
4-dodecylaniline

-
-
1198578-78-7
1,3,5-tris(4-n-dodecylanilino)benzene

| Conditions | Yield |
|---|---|
| iodine In toluene at 130℃; for 2h; | 87.8% |
-
-
104-42-7
4-dodecylaniline

-
-
111772-62-4
2-hydroxy-4-n-dodecyloxybenzaldehyde

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 18h; Reflux; | 86% |
-
-
1333471-38-7
trans-(2,6-diformyl-3,5-bis(2,5,8,11-tetraoxadecyl)phenyl)chlorobis(triphenylphosphine)palladium

-
-
104-42-7
4-dodecylaniline

-
-
1333471-36-5
(3,5-bis(2,5,8,11-tetraoxadecyl)-2,6-bis(N-4-dodecylphenylimino)phenyl)chloropalladium

| Conditions | Yield |
|---|---|
| With (H2N)2CO*H2O2 In acetonitrile (N2); refluxing soln. of palladium compd. and aniline deriv. in anhyd. CH3CN for 72 h, cooling to 25°C, addn. of urea hydrogen peroxide, stirring at 50°C for 11 h; evapn., chromy. (silica gel, 0-5% methanol/ethyl acetate), gel permeating chromy. (CHCl3), elem. anal.; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-dodecylaniline; phenylpropyolic acid With dmap In tetrahydrofuran at 0℃; for 0.166667h; Inert atmosphere; Stage #2: With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 80% |
-
-
104-42-7
4-dodecylaniline

-
-
147622-34-2
4'-formyl-2-diphenylphosphino-acetophenone

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 2h; Heating; | 79% |
-
-
104-42-7
4-dodecylaniline

-
-
148347-18-6, 139141-81-4
1,2-epox-[60]fullerene

-
A
-
1092060-75-7
1-(4-dodecylanilino)-2-hydroxy-1,2-dihydro[60]fullerene

-
B
-
1092060-70-2
5'-dodecylindolino[2',3':1,2][60]fullerene

| Conditions | Yield |
|---|---|
| With sepiolite In chlorobenzene at 100℃; | A n/a B 78% |

-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
104-42-7
4-dodecylaniline

-
-
90850-59-2
(4,6-Dichloro-[1,3,5]triazin-2-yl)-(4-dodecyl-phenyl)-amine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; acetone 1. 0 deg C, 1 h; 2. 20 deg C, 1 h; | 76% |

| Conditions | Yield |
|---|---|
| With acetic acid | 76% |

| Conditions | Yield |
|---|---|
| at 20℃; | 74% |
| for 6h; Ambient temperature; |
-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With benzyltriethylammonium tribromide; calcium carbonate In methanol; dichloromethane at 20℃; for 1h; | 74% |
| With hydrogenchloride; bromine In water for 3h; |

| Conditions | Yield |
|---|---|
| Stage #1: 4-dodecylaniline; Propiolic acid With dmap In tetrahydrofuran at 0℃; for 0.166667h; Inert atmosphere; Stage #2: With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 74% |
-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 100℃; Inert atmosphere; | 73% |
-
-
104-42-7
4-dodecylaniline

-
-
104010-92-6
2,3-bis(tert-butoxycarbonyl amino)propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-bis(tert-butoxycarbonyl amino)propanoic acid With 1-hydroxy-pyrrolidine-2,5-dione; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In chloroform at 20℃; for 2h; Inert atmosphere; Stage #2: 4-dodecylaniline With dmap In chloroform at 50℃; Inert atmosphere; | 72% |
-
-
104-42-7
4-dodecylaniline

-
-
47173-80-8
N-Boc-D-serine(Bzl)-OH

-
-
448959-69-1
[2-benzyloxy-1-(4-decyl-phenylcarbamoyl)-ethyl]-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate; N-ethyl-N,N-diisopropylamine | 71% |

-
-
104-42-7
4-dodecylaniline

| Conditions | Yield |
|---|---|
| In neat (no solvent) Co complex mixed with ligand; heated on steam bath for 1 h; purified by preparative TLC (alumina, CHCl3); elem. anal.; | 70% |
-
-
13669-05-1
1,4-bis(thiophen-2-yl)butane-1,4-dione

-
-
104-42-7
4-dodecylaniline

-
-
499793-93-0
1-(p-dodecylphenyl)-2,5-di(2-thienyl)pyrrole

| Conditions | Yield |
|---|---|
| In acetic acid; toluene at 140 - 150℃; | 69% |
-
-
104-42-7
4-dodecylaniline

-
-
891780-19-1
2-bromo-4-dodecylaniline

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane at 20℃; for 20h; | 66% |
| With sodium perborate; acetic acid; potassium bromide; sodium vanadate at 20℃; for 10h; | 59% |
| With N-Bromosuccinimide; ammonium acetate In acetonitrile at 20℃; for 3h; |
-
-
104-42-7
4-dodecylaniline

-
-
170698-90-5
1-dodecyl-4-iodobenzene

| Conditions | Yield |
|---|---|
| With C5H11NO2; potassium iodide | 65% |
| Stage #1: 4-dodecylaniline With hydrogenchloride; sodium nitrite at 0℃; Stage #2: With potassium iodide at 40℃; for 0.333333h; | 49% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-dodecylaniline With hydrogenchloride; sodium nitrite In tetrahydrofuran; water at 0℃; for 2h; Inert atmosphere; Stage #2: phenol With sodium carbonate; potassium carbonate In tetrahydrofuran; water at 20℃; for 12h; | 65% |
| With hydrogenchloride; sodium carbonate; sodium nitrite In water | |
| Stage #1: 4-dodecylaniline With hydrogenchloride; sodium nitrite In water at 0℃; for 0.5h; Stage #2: phenol With sodium carbonate In water at 0℃; for 1h; |
4-Dodecylaniline Chemical Properties
Product Name: 4-Dodecylaniline
The MF of 4-Dodecylaniline (104-42-7) is C18H31N.
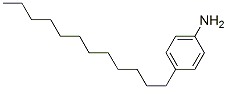
The MW of 4-Dodecylaniline (104-42-7) is 261.45.
Synonyms of 4-Dodecylaniline (104-42-7): 4-Dodecylaniline ; p-Dodecylaniline ; 4-Dodecyl-benzenamin ; 4-N-Dodecylamine ; Benzenamine, 4-dodecyl-
Product Categories: Intermediates of Dyes and Pigments;Alkyl;Alkylanilines;Nitrogen Compounds;Organic Building Blocks;Polyamines
EINECS: 203-201-3
Index of Refraction: 1.508
Density: 0.902 g/ml
Flash Point: 162.7 °C
Boiling Point: 364 °C
Melting Point: 35-39 °C(lit.)
4-Dodecylaniline Uses
4-Dodecylaniline (104-42-7) is used for the manufacture of Kapu Lun Huang 3GS, Kapu Lun pink BS, Kapu Lun blue dye BS, etc.
4-Dodecylaniline Safety Profile
Safety information of 4-Dodecylaniline (104-42-7):
Hazard Codes ![]() Xn
Xn
Risk Statements
22 Harmful if swallowed
Safety Statements
22 Do not breathe dust
24/25 Avoid contact with skin and eyes
WGK Germany 3
Related Products
- 4-Dodecylaniline
- 1044281-58-4
- 10442-83-8
- 10443-65-9
- 104436-60-4
- 104-43-8
- 104439-77-2
- 10444-89-0
- 1044-49-1
- 104449-98-1
- 104-45-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View