-
Name
4-ethylbenzaldehyde
- EINECS 225-268-8
- CAS No. 4748-78-1
- Article Data74
- CAS DataBase
- Density 1.001 g/cm3
- Solubility Not miscible or difficult to mix in water. Soluble in alcohol.
- Melting Point
- Formula C9H10O
- Boiling Point 221.7 °C at 760 mmHg
- Molecular Weight 134.178
- Flash Point 92.2 °C
- Transport Information
- Appearance clear colourless to slightly yellow liquid
- Safety 26-27-28-36/37/39-37/39
- Risk Codes 36/37/38-22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Benzaldehyde,p-ethyl- (6CI,7CI,8CI);p-Ethylbenzaldehyde;
- PSA 17.07000
- LogP 2.06150
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen In tetrahydrofuran at 50℃; under 760.051 Torr; for 10h; | A 97% B 3% |
| With hydrogen In dichloromethane at 40℃; under 1500.15 Torr; for 24h; Reagent/catalyst; Solvent; Temperature; Pressure; Schlenk technique; | A 67 %Chromat. B 33 %Chromat. |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; nitric acid In acetone at 20℃; for 0.233333h; Sonication; | 95% |
| With 3,3-dichloro-1,2-diphenylcyclopropene; dimethyl sulfoxide; triethylamine In dichloromethane at -78 - 20℃; Inert atmosphere; | 95% |
| With laccase; 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt In tetrahydrofuran; water at 40℃; for 24h; | 92% |

| Conditions | Yield |
|---|---|
| With immobilised amine transaminase from the moderate halophilic bacterium Halomonas elongata; sodium pyruvate In aq. phosphate buffer; dimethyl sulfoxide at 37℃; under 760.051 Torr; for 2h; pH=8; | 94% |
| With steam; nickel at 210℃; |
-
-
1585-07-5
1-bromo-4-ethylbenzene

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With samarium; copper(l) iodide at 20℃; for 4h; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In acetonitrile at 65℃; for 6h; | 92% |

| Conditions | Yield |
|---|---|
| With methanol; formic acid; water In ethyl acetate at 25℃; for 3h; Irradiation; Inert atmosphere; | 90% |
| With aluminum (III) chloride In ethanol; water; acetonitrile at 20℃; for 6h; Irradiation; Inert atmosphere; | 95 %Chromat. |

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; nickel(II) chloride hexahydrate In methanol at 20℃; for 2.5h; chemoselective reaction; | 86% |

| Conditions | Yield |
|---|---|
| With succinic acid anhydride; methylphenylsilane; C58H82O6P2; copper(II) nitrate In tetrahydrofuran at 75℃; for 18h; Schlenk technique; Inert atmosphere; | 85% |
| With potassium phosphate; sodium hypophosphite; 2,2-dimethylpropanoic anhydride; palladium diacetate; tricyclohexylphosphine In tetrahydrofuran at 60℃; for 16h; | 74% |
| With 2,6-dimethylpyridine; nickel(II) bromide trihydrate; phenylsilane; 4,4'-di-tert-butyl-2,2'-bipyridine; zinc; dimethyl dicarbonate In ethyl acetate at 60℃; for 24h; Schlenk technique; Inert atmosphere; | 76 %Chromat. |

| Conditions | Yield |
|---|---|
| Stage #1: formic acid; 4-ethyl-1-iodobenzene With palladium diacetate; acetic anhydride; tricyclohexylphosphine In N,N-dimethyl-formamide at 30℃; for 1h; Inert atmosphere; Green chemistry; Stage #2: With triethylamine In N,N-dimethyl-formamide at 80℃; for 6h; Inert atmosphere; Green chemistry; | 85% |
| With palladium diacetate; triethylamine; dicyclohexyl-carbodiimide; tricyclohexylphosphine In N,N-dimethyl-formamide at 80℃; for 10h; Inert atmosphere; Sealed tube; | 71% |

| Conditions | Yield |
|---|---|
| at 100℃; for 5h; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: carbon dioxide With zinc(II) cyanide; ruthenium trichloride; rhodium (III) acetate; cobalt(II) acetate; zirconium(IV) acetate for 1h; Autoclave; Stage #2: ethylbenzene at 50℃; under 15001.5 Torr; for 5h; | 80% |
-
-
97-94-9
triethyl borane

-
-
17763-69-8
4-(trifluormethanesulfonyloxy)benzaldehyde

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; cesium acetate In tetrahydrofuran Suzuki-Miyaura cross-coupling; Reflux; Inert atmosphere; | 78% |

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With 4-ethylbenzyl alcohol; 1,2-dimethylindazolium chloride for 3h; Heating; | 71% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide at 120℃; for 5h; Sealed tube; Green chemistry; | 70% |
-
-
10217-72-8
p,p'-diethyldiphenylmercury

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With trichlorophosphate In water at 70℃; for 5h; | 65% |
-
-
811-49-4
ethyllithium

-
-
192436-83-2
4-bromo-N-methoxy-N-methylbenzamide

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-N-methoxy-N-methylbenzamide With diisobutylaluminium hydride In hexane; toluene at 0 - 20℃; Inert atmosphere; Schlenk technique; Stage #2: ethyllithium In hexane; toluene at 20℃; Inert atmosphere; Schlenk technique; | 61% |

| Conditions | Yield |
|---|---|
| With (1,2-dimethoxyethane)dichloronickel(II); N,N,N,N,-tetramethylethylenediamine; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; water; N-ethyl-N,N-diisopropylamine In dimethyl sulfoxide at 80℃; under 760.051 Torr; for 24h; Schlenk technique; Sealed tube; Irradiation; | 54% |

| Conditions | Yield |
|---|---|
| With (1,2-dimethoxyethane)dichloronickel(II); N,N,N,N,-tetramethylethylenediamine; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; water; N-ethyl-N,N-diisopropylamine In dimethyl sulfoxide at 80℃; under 760.051 Torr; for 24h; Schlenk technique; Sealed tube; Irradiation; | 50% |
-
-
35076-76-7
4-ethyl-1-cyclopropylbenzene

-
A
-
127506-37-0
(E)-3-(4-ethylphenyl)acrylaldehyde

-
B
-
119200-33-8
5-(4-ethylphenyl)isoxazoline

-
C
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With dinitrogen tetraoxide; triethylamine In dichloromethane at -30℃; for 1h; | A 32% B 48% C 8% |
| With dinitrogen tetraoxide In dichloromethane at -30℃; for 1h; | A 35% B 40% C 8% |

-
-
6921-43-3
1-cyclopropyl-4-methyl-benzene

-
A
-
127506-37-0
(E)-3-(4-ethylphenyl)acrylaldehyde

-
B
-
119200-33-8
5-(4-ethylphenyl)isoxazoline

-
C
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With dinitrogen tetraoxide In dichloromethane at -30℃; for 1h; | A 35% B 40% C 8% |

-
-
67302-72-1
(4-ethyl-benzyliden)-aniline

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With sulfuric acid |
-
-
246161-07-9
N-(4-ethyl-benzylidene)-p-toluidine

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With hexamethylenetetramine |

| Conditions | Yield |
|---|---|
| With oxygen; iron(II) chloride; zinc; iron(III) In pyridine; acetic acid for 16h; | A 10.3 % Chromat. B 3.1 % Chromat. |
| With oxygen; acetic acid; iron(II) chloride; zinc; iron(III) In pyridine for 16h; Product distribution; other alkylbenzenes; without Fe(III)-catalyst; | |
| With oxygen In acetonitrile at 24.84℃; under 760.051 Torr; Reagent/catalyst; Irradiation; Green chemistry; |
-
-
622-96-8
4-methylethylbenzene

-
A
-
622-97-9
1-ethenyl-4-methylbenzene

-
B
-
536-50-5
CH3C6H4CH(CH3)OH

-
C
-
122-00-9
para-methylacetophenone

-
D
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; copper diacetate In water; acetonitrile at 100℃; Yield given. Further byproducts given. Yields of byproduct given; | |
| With ammonium peroxydisulfate; copper diacetate; sodium acetate In water; acetonitrile at 100℃; Yield given. Further byproducts given. Yields of byproduct given; |

-
-
622-96-8
4-methylethylbenzene

-
A
-
622-97-9
1-ethenyl-4-methylbenzene

-
B
-
67035-84-1
4-ethylbenzyl acetate

-
C
-
122-00-9
para-methylacetophenone

-
D
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; copper diacetate In water; acetic acid at 100℃; Yield given. Further byproducts given. Yields of byproduct given; |

-
-
85623-71-8, 137957-42-7
N-tert-butyl α-(4-ethylphenyl) nitrone

-
A
-
16649-50-6
N-tert-Butylhydroxylamine

-
B
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With water at 25℃; Rate constant; hydrolysis; |

| Conditions | Yield |
|---|---|
| With water; hydrogen cation In various solvent(s) at 25℃; Rate constant; Mechanism; pH=1.00; rate constant - octanol/water partition coefficient and relative rate - substituent constants correlations in lamellar solvent; |
-
-
622-96-8
4-methylethylbenzene

-
A
-
622-97-9
1-ethenyl-4-methylbenzene

-
B
-
768-59-2
4-ethylbenzyl alcohol

-
C
-
122-00-9
para-methylacetophenone

-
D
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With nitrate radical under 760 Torr; Kinetics; Oxidation; |

-
-
104-13-2
4-Butylaniline

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1046769-82-7
4-butyl-N-[(1E)-(4-ethylphenyl)methylene]aniline

| Conditions | Yield |
|---|---|
| In ethanol Reflux; Inert atmosphere; | 100% |
-
-
107-10-8
propylamine

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1246401-39-7
N-(4-ethylbenzylidene)propan-1-amine

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| at 20℃; for 1h; |

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
| at 20℃; for 1h; | |
| In dichloromethane at 20℃; for 4h; |
-
-
77086-38-5
ketene t-butyldimethylsilyl methyl acetal

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1231743-50-2
C18H30O3Si

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; 2,6-bis-[1-(2,6-diethylphenylimino)ethyl]pyridine iron(II) chloride In dichloromethane at 20℃; Mukaiyama Aldol Addition; Inert atmosphere; | 100% |
| With C33H29FeNO2P(1+)*I(1-) In acetonitrile at 20℃; for 0.25h; Mukaiyama aldol reaction; | 48% |
-
-
2579-20-6
cis, trans-1,3-dimethylaminocyclohexane

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1217526-83-4
C26H34N2

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Molecular sieve; | 100% |
| In methanol at 20℃; Molecular sieve; |

| Conditions | Yield |
|---|---|
| at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 20℃; for 15h; Claisen-Schmidt Condensation; | 100% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0℃; for 0.25h; | 99% |
| With formaldehyd; C42H36ClIrO2P2; triethylamine In 1,2-dimethoxyethane at 80℃; for 20h; Reagent/catalyst; Schlenk technique; Inert atmosphere; chemoselective reaction; | 99% |
| With sodium tetrahydroborate In methanol for 14h; | 98% |

| Conditions | Yield |
|---|---|
| With ammonium acetate; acetic acid for 1h; Reflux; stereoselective reaction; | 99% |
| With ammonium acetate; acetic acid for 1 - 12h; Heating / reflux; |
-
-
617-86-7
triethylsilane

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1163144-08-8
triethyl((4-ethylbenzyl)oxy)silane

| Conditions | Yield |
|---|---|
| With C42H43BN3P2(1+)*C18HBF15(1-) In chloroform at 40℃; for 1h; Reagent/catalyst; Temperature; Schlenk technique; Glovebox; | 99% |
-
-
99-91-2
para-chloroacetophenone

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
343610-66-2
(2E)-1-(4-chlorophenyl)-3-(4-ethylphenyl)prop-2-en-1-one

| Conditions | Yield |
|---|---|
| Stage #1: para-chloroacetophenone; 4-ethylbenzylaldehyde With sodium methylate In methanol at 20℃; for 16h; Stage #2: With hydrogenchloride In methanol; water | 98% |
| With potassium hydroxide In ethanol at 20℃; Claisen-Schmidt Condensation; |
-
-
99-91-2
para-chloroacetophenone

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
782483-60-7
1-(4-chloro-phenyl)-3-(4-ethyl-phenyl)-propenone

| Conditions | Yield |
|---|---|
| Stage #1: para-chloroacetophenone; 4-ethylbenzylaldehyde With sodium methylate In methanol at 20℃; for 16h; Stage #2: With hydrogenchloride In methanol; water | 98% |
-
-
6284-40-8
1-deoxy-1-(methylamino)-D-glucitol

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1042065-80-4
(2R,5S)-2-(4-ethylphenyl)-3-methyl-5-(D-arabino-1,2,3,4-tetrahydroxybutyl)oxazolidine

| Conditions | Yield |
|---|---|
| In benzene Heating; | 98% |
-
-
100-63-0
phenylhydrazine

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
324067-43-8
1-(4-ethylbenzylidene)-2-phenylhydrazine

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane | 98% |
| In dichloromethane at 20℃; for 0.5h; | |
| In ethanol; water at 80℃; |

| Conditions | Yield |
|---|---|
| With cyclohexane-1,2-dione hydrolase In aq. buffer at 30℃; for 48h; pH=6.5; Enzymatic reaction; | 98% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 20℃; for 4h; | 98% |

| Conditions | Yield |
|---|---|
| With sodium acetate In ethanol for 1h; Reflux; | 98% |


| Conditions | Yield |
|---|---|
| With (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate In water at 20℃; for 3h; Schlenk technique; Inert atmosphere; Irradiation; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With yttria doped hydroxyapatites In ethanol at 20℃; Reagent/catalyst; Temperature; Green chemistry; | 98% |


| Conditions | Yield |
|---|---|
| With hydrazine hydrate In water at 60℃; for 0.75h; Green chemistry; | 98% |


| Conditions | Yield |
|---|---|
| Stage #1: p-toluene sulfinic acid; 4-ethylbenzylaldehyde In water at 20℃; for 0.416667h; Stage #2: urea In water at 20℃; for 20h; | 98% |

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
106-50-3
1,4-phenylenediamine

-
-
28784-88-5
bis(4-ethylbenzylidene)-p-phenylenediamine

| Conditions | Yield |
|---|---|
| In ethanol for 3h; | 97% |
| In methanol |
-
-
104-13-2
4-Butylaniline

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
54379-33-8
N-(4-n-ethylbenzylidene)-4'-n-butylaniline

| Conditions | Yield |
|---|---|
| 97% |
-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1174516-81-4
sodium (4-ethylphenyl)(hydroxy)methanesulfonate

| Conditions | Yield |
|---|---|
| With sodium hydrogensulfite In ethanol; water for 20.5h; Inert atmosphere; | 97% |
| With sodium metabisulfite; water In ethanol Cooling; |
-
-
613-94-5
benzoic acid hydrazide

-
-
106-95-6
allyl bromide

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
1449037-68-6
N'-[1-(4-ethylphenyl)but-3-en-1-yl]benzohydrazide

| Conditions | Yield |
|---|---|
| With zinc perchlorate; indium In methanol at 20℃; for 4h; chemoselective reaction; | 97% |

-
-
38622-91-2, 36635-61-7
[(p-methylphenyl)sulfonylmethyl]isonitrile

-
-
4748-78-1
4-ethylbenzylaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate at 80℃; for 1.5h; | 97% |

| Conditions | Yield |
|---|---|
| With biguanide-functionalized hierarchical porous covalent organic framework In water at 20℃; for 0.166667h; Knoevenagel Condensation; | 97% |
| With dimethyl 3-methyl-9-oxo-7-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecyl)-2,4-di(pyridin-2-yl)-3,7-diazabicyclo-[3.3.1]nonane-1,5-dicarboxylate In water at 40℃; for 2h; Knoevenagel Condensation; | 95% |
| Stage #1: 4-ethylbenzylaldehyde With C9H11ClN2O*ClHO4 In neat (no solvent) for 0.166667h; Knoevenagel Condensation; Stage #2: malononitrile In neat (no solvent) at 25 - 30℃; for 0.5h; Knoevenagel Condensation; | 94% |
-
-
1067-74-9
Methyl diethylphosphonoacetate

-
-
4748-78-1
4-ethylbenzylaldehyde

-
-
87087-37-4
3-(4-ethylphenyl)acrylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: Methyl diethylphosphonoacetate With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 0.5h; Inert atmosphere; Stage #2: 4-ethylbenzylaldehyde In tetrahydrofuran; hexane at 20℃; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With 2.5 wt% vanadium oxide loaded on fluorapatite In ethanol at 20℃; for 0.5h; Green chemistry; | 97% |

4-Ethylbenzaldehyde Specification
The Benzaldehyde, 4-ethyl- is a derivative of benzaldehyde with the formula C9H10O. The IUPAC name of this chemical is 4-ethylbenzaldehyde. With the CAS registry number 4748-78-1 and EINECS 225-268-8, it is also named as Ethylbenzaldehyde, p-. The product's categories are Aromatic Aldehydes & Derivatives (substituted); Benzaldehyde; Benzaldehyde (Building Blocks for Liquid Crystals); Building Blocks for Liquid Crystals; Functional Materials; Alphabetical Listings; Flavors and Fragrances; Aldehydes; Carbonyl Compounds. It is clear colourless to slightly yellow liquid which is stable under normal temperature and pressure. In addition, this chemical should be sealed in the container, stored in the well-ventilated and dry place and avoided direct sunshine.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.63; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.63; (4)ACD/LogD (pH 7.4): 2.63; (5)#H bond acceptors: 1; (6)#H bond donors: 0; (7)#Freely Rotating Bonds: 2; (8)Polar Surface Area: 17.07 Å2; (9)Index of Refraction: 1.548; (10)Molar Refractivity: 42.55 cm3; (11)Molar Volume: 133.9 cm3; (12)Polarizability: 16.87×10-24 cm3; (13)Surface Tension: 36.3 dyne/cm; (14)Density: 1.001 g/cm3; (15)Flash Point: 92.2 °C; (16)Enthalpy of Vaporization: 45.81 kJ/mol; (17)Boiling Point: 221.7 °C at 760 mmHg; (18)Vapour Pressure: 0.106 mmHg at 25°C.
Preparation of Benzaldehyde, 4-ethyl-: It can be obtained by N,N-dimethyl-formamide and bis-(4-ethyl-phenyl)-mercury. This reaction needs reagent POCl3 and solvent water at temperature of 70 °C. The reaction time is 5 hours. The yield is 65%.
.gif)
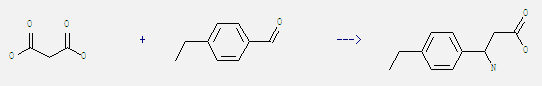
Uses of Benzaldehyde, 4-ethyl-: It can react with malonic acid to get 3-amino-3-(4-ethyl-phenyl)-propionic acid. This reaction needs reagent AcONH4 and solvent ethanol by heating. The reaction time is 6 hours. The yield is 65%.
When you are using this chemical, please be cautious about it as the following:
It is not only harmful if swallowed, but also irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. After contact with skin, wash immediately with plenty of soap-suds. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection.
People can use the following data to convert to the molecule structure.
1. SMILES:CCc1ccc(C=O)cc1
2. InChI:InChI=1/C9H10O/c1-2-8-3-5-9(7-10)6-4-8/h3-7H,2H2,1H3
3. InChIKey:QNGNSVIICDLXHT-UHFFFAOYAN
Related Products
- 4-Ethylbenzaldehyde
- 4748-80-5
- 474893-07-7
- 474918-32-6
- 474918-42-8
- 474922-77-5
- 474922-82-2
- 474922-84-4
- 474922-90-2
- 474960-44-6
- 475-03-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View