 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
-
Name
4-Nitroaniline
- EINECS 202-810-1
- CAS No. 100-01-6
- Article Data828
- CAS DataBase
- Density 1.333 g/cm3
- Solubility 0.8 g/L (20 °C) in water
- Melting Point 147 °C
- Formula C6H6N2O2
- Boiling Point 333.1 °C at 760 mmHg
- Molecular Weight 138.126
- Flash Point 165 °C
- Transport Information UN 1661
- Appearance Yellow solid with a mild odor
- Safety 28-36/37-45-61-28A-16-1/2-7
- Risk Codes 23/24/25-33-52/53-39/23/24/25-11
-
Molecular Structure
-
Hazard Symbols
 T,
T, F
F
- Synonyms Aniline,p-nitro- (8CI);1-Amino-4-nitrobenzene;4-Amino-1-nitrobenzene;4-Aminonitrobenzene;4-Nitro-1-aminobenzene;
- PSA 71.84000
- LogP 2.28140
Synthetic route

| Conditions | Yield |
|---|---|
| With formic acid In acetonitrile at 20℃; for 1h; Irradiation; | 100% |
| In water at 25℃; for 0.333333h; Sonication; | 99% |
| With sodium tetrahydroborate In water at 95℃; for 0.333333h; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With 1,1,1,3',3',3'-hexafluoro-propanol; tetrabutylammonium perchlorate In N,N-dimethyl-formamide electrolytic reduction at a Pt gauze electrode at a potential of -0.30 V; | 100% |
| With Decaborane; palladium on activated charcoal In methanol at 20℃; for 0.25h; | 99% |
| With triethylsilane; indium(III) chloride In acetonitrile at -20℃; for 0.25h; | 99% |
-
-
77853-01-1
(4-nitro-phenyl)-m-tolyl sulfone

-
A
-
100-32-3
di(p-nitrophenyl) disulfide

-
B
-
77853-09-9
Toluene-3-thiosulfonic acid; compound with ammonia

-
C
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With sulfur; ammonia at 100℃; for 2h; titanium autoclave; | A 43% B 100% C 40% |

-
-
22865-57-2
1-methoxy-4-((4-nitrophenyl)sulfonyl)benzene

-
A
-
100-32-3
di(p-nitrophenyl) disulfide

-
B
-
77853-03-3
4-Methoxy-benzenethiosulfonic acid; compound with ammonia

-
C
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With sulfur; ammonia at 100℃; for 2h; titanium autoclave; | A 56% B 100% C 32% |
| With sulfur; ammonia at 100℃; for 2h; Product distribution; titanium autoclave; various reactions temp. and amounts of S8; |


| Conditions | Yield |
|---|---|
| With graphitic carbon nitride; hydrazine hydrate In water at 70℃; for 8h; Time; Darkness; Sealed tube; Green chemistry; chemoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 120 - 190℃; under 45004.5 - 46504.7 Torr; for 4h; Temperature; Pressure; | 99.31% |
| With ammonium hydroxide; copper(l) iodide at 200℃; for 6.5h; Reagent/catalyst; Temperature; Time; Autoclave; Green chemistry; | 97% |
| With copper(ll) sulfate pentahydrate; ammonium hydroxide In PEG1000-DIL; methyl cyclohexane at 60℃; for 4h; | 96% |
-
-
18437-63-3
tert-butyl N-(4-nitrophenyl)carbamate

-
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With EPZG clay In dichloromethane for 1.5h; deacylation; Heating; | 99% |
| With H-β zeolite In dichloromethane for 4h; Heating; | 98% |
| With water at 150℃; for 10h; Subcritical conditions; | 97% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; ascorbic acid In ammonia at 25℃; for 18h; Kinetics; liquid NH3; | 99% |
| With ammonium hydroxide; copper(l) iodide; phosphate potassium salt In N,N-dimethyl-formamide at 20℃; for 36h; Inert atmosphere; | 98% |
| Stage #1: p-nitrobenzene iodide With copper(l) iodide; D-glucosamine hydrochloride; potassium carbonate In water; acetone at 90℃; for 0.166667h; Stage #2: With ammonia In water; acetone at 90℃; for 28h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: para-nitrophenyl bromide With copper(l) iodide; D-glucosamine hydrochloride; potassium carbonate In water; acetone at 90℃; for 0.166667h; Stage #2: With ammonia In water; acetone at 90℃; for 28h; | 98% |
| With ammonium hydroxide at 20℃; for 3h; Catalytic behavior; | 98% |
| With ammonia; triethylamine In water at 20℃; for 2.5h; | 98% |
-
-
63-68-3
L-methionine

-
-
74197-40-3
3,5-dinitro-1-(4-nitrophenyl)-4-pyridone

-
A
-
100-01-6
4-nitro-aniline

-
B
-
92782-41-7
(S)-2-(3,5-Dinitro-4-oxo-4H-pyridin-1-yl)-4-methylsulfanyl-butyric acid

| Conditions | Yield |
|---|---|
| In pyridine; water for 3h; Ambient temperature; | A n/a B 98% |

-
-
90-15-3
α-naphthol

-
-
64168-52-1
4'-nitrobenzenesulfenanilide

-
A
-
68143-73-7
2-phenylthio-1-naphthol

-
B
-
19133-53-0
4-(phenylthio)naphthalen-1-ol

-
C
-
100-01-6
4-nitro-aniline

-
D
-
103929-98-2
2,4-bis(phenylthio)naphthalen-1-ol

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In benzene for 0.166667h; Ambient temperature; Further byproducts given; | A 42% B 33% C 98% D 10% |

-
-
90-15-3
α-naphthol

-
-
64168-52-1
4'-nitrobenzenesulfenanilide

-
A
-
68143-73-7
2-phenylthio-1-naphthol

-
B
-
19133-53-0
4-(phenylthio)naphthalen-1-ol

-
C
-
100-01-6
4-nitro-aniline

-
D
-
882-33-7
diphenyldisulfane

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In benzene for 0.166667h; Ambient temperature; Further byproducts given; | A 42% B 33% C 98% D 14% |

-
-
90-15-3
α-naphthol

-
-
64168-52-1
4'-nitrobenzenesulfenanilide

-
A
-
68143-73-7
2-phenylthio-1-naphthol

-
B
-
19133-53-0
4-(phenylthio)naphthalen-1-ol

-
C
-
100-01-6
4-nitro-aniline

-
D
-
103929-98-2
2,4-bis(phenylthio)naphthalen-1-ol

-
E
-
882-33-7
diphenyldisulfane

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In benzene for 0.166667h; Product distribution; Mechanism; 1.5 eqiv. TFA; | A 42% B 33% C 98% D 10% E 14% |

-
-
64168-52-1
4'-nitrobenzenesulfenanilide

-
A
-
1212-08-4
S-Phenyl benzenethiosulfonate

-
B
-
100-01-6
4-nitro-aniline

-
C
-
882-33-7
diphenyldisulfane

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In benzene at 25℃; Product distribution; Mechanism; further sulfenanilide; | A 32% B 98% C 56% |

-
-
76-05-1
trifluoroacetic acid

-
A
-
2168-93-6
butyl sulfoxide

-
B
-
404-27-3
2,2,2-trifluoro-N-(4-nitrophenyl)acetamide

-
C
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| at 72℃; for 72h; Elimination; acylation; retro-Pummerer reaction; | A 79% B 98% C 2% |


| Conditions | Yield |
|---|---|
| With ammonium iodide; hydrazine hydrate In ethanol at 60℃; for 42h; Inert atmosphere; Sealed tube; | 97% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol for 1h; Product distribution; Further Variations:; Solvents; reaction time; Heating; | 96% |
| With 40% potassium fluoride/alumina at 85℃; for 0.0666667h; Microwave irradiation; Neat (no solvent); | 94% |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol for 1.5h; Product distribution; Further Variations:; Solvents; reaction time; Heating; | 97% |
| In methanol |
-
-
16169-16-7
4-Nitro-N-phenylhydroxylamine

-
-
88867-70-3
C11H14N2O4

-
A
-
4485-08-9
4-nitrosonitrobenzene

-
B
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With iron(II) chloride In water; acetonitrile at 40℃; pH=4.7, ionic strength=0.5 M; | A 39% B 95% |
| With iron(II) chloride In water; acetonitrile at 40℃; Mechanism; pH=4.7, ionic strength=0.5 M; |

-
-
88867-70-3
C11H14N2O4

-
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With iron(II) chloride In water; acetonitrile at 40℃; pH=4.7, ionic strength=0.5 M; | 95% |
| With FeCl2 or CuCl In water; acetonitrile at 40℃; Product distribution; Mechanism; pH=4.7, ionic strength=0.5 M; |
-
-
108047-30-9
3-methyl-1-(4-nitrophenyl)-5-nitrouracil

-
-
74-89-5
methylamine

-
A
-
41613-26-7
1,3-dimethyl-5-nitrouracil

-
B
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| In methanol for 0.5h; Ambient temperature; | A 95% B n/a |

-
-
1516-60-5
4-nitrophenyl azide

-
-
105-53-3
diethyl malonate

-
A
-
5256-74-6
1,3-diethyl 2-diazopropanedioate

-
B
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With tetrabutylammonium perchlorate In N,N-dimethyl-formamide electrolytic reduction at a Pt gauze electrode at a potential of -0.30 V; | A 95% B 101 % |

-
-
78596-42-6
ethyl α-ethoxycarbonyl-β-(4-nitroanilino)acrylate

-
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With ethylenediamine In ethanol at 20℃; for 2.3h; | 95% |
-
-
110-86-1
pyridine

-
-
74197-40-3
3,5-dinitro-1-(4-nitrophenyl)-4-pyridone

-
A
-
100-01-6
4-nitro-aniline

-
B
-
139055-63-3
1-Hydroxy-3,5-dinitro-1H-pyridin-4-one; compound with pyridine

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride at 20℃; for 2h; | A 94% B 83% |

-
-
77863-74-2
C14H13NO5S

-
A
-
100-32-3
di(p-nitrophenyl) disulfide

-
B
-
77853-06-6
3-Ethoxy-benzenethiosulfonic acid; compound with ammonia

-
C
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With sulfur; ammonia at 100℃; for 2h; titanium autoclave; | A 47% B 94% C 40% |

-
-
88867-70-3
C11H14N2O4

-
-
823-86-9
N-(4-chlorophenyl)hydroxylamine

-
A
-
932-98-9
1-chloro-4-nitroso-benzene

-
B
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With iron(II) chloride In water; acetonitrile at 40℃; pH=4.7, ionic strength=0.5 M; | A 53% B 94% |
| With iron(II) chloride In water; acetonitrile at 40℃; Mechanism; pH=4.7, ionic strength=0.5 M; |


| Conditions | Yield |
|---|---|
| With pyrene; tetramethyl ammoniumhydroxide; benzamide; potassium carbonate In dimethyl sulfoxide at 90℃; for 2.08333 - 2.16667h; Conversion of starting material; | A 94% B 6% |
| With potassium hydroxide; pyrene; potassium carbonate; urea In dimethyl sulfoxide at 90℃; for 2.08333 - 2.16667h; Conversion of starting material; | A 87% B 12% |
| With urea, monopotassium salt; urea In dimethyl sulfoxide at 90℃; for 2h; | A 56% B 5.5% |

| Conditions | Yield |
|---|---|
| With Vilsmeier reagent; potassium nitrate In neat (no solvent) at 100℃; under 1500.15 Torr; Reagent/catalyst; Temperature; Microwave irradiation; | 93% |
| With nitric acid; sodium dodecyl-sulfate In water at 25℃; for 0.333333h; Micellar solution; Green chemistry; regioselective reaction; | 86% |
| With nitric acid; sodium dodecyl-sulfate In acetonitrile at 24.84℃; for 3h; Micellar solution; regioselective reaction; | 85% |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride at 20℃; for 2h; | A 93% B 88% |


| Conditions | Yield |
|---|---|
| With 1,2-bis(mercaptomethyl)benzene; (Et4N) In acetonitrile at 26℃; for 1.5h; | A 93% B 2% |
| With D-glucose; bakers' yeast In phosphate buffer; ethanol at 30℃; for 0.5h; pH=6.5; Product distribution; Further Variations:; time; | A 83% B n/a |
| With sodium tetrahydroborate; selenium In ethanol at 23 - 26℃; for 0.25h; | A 81% B 16% |
| With 1,2-bis(mercaptomethyl)benzene; (Et4N) In acetonitrile at 26℃; for 1.5h; Product distribution; |
-
-
77853-02-2
1,3-Dichloro-5-(4-nitro-benzenesulfonyl)-benzene

-
A
-
100-32-3
di(p-nitrophenyl) disulfide

-
B
-
77853-10-2
3,5-Dichloro-benzenethiosulfonic acid; compound with ammonia

-
C
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With sulfur; ammonia at 100℃; for 2h; titanium autoclave; | A 42% B 93% C 40% |

-
-
555-16-8
4-nitrobenzaldehdye

-
-
100-01-6
4-nitro-aniline

-
-
10480-05-4
N-(4-nitrobenzylidene)-4-nitroaniline

| Conditions | Yield |
|---|---|
| at 60℃; for 2h; | 100% |
| at 83 - 85℃; | 100% |
| With sulfuric acid In neat (no solvent) Microwave irradiation; Sealed tube; Green chemistry; | 72% |
-
-
100-52-7
benzaldehyde

-
-
100-01-6
4-nitro-aniline

-
-
785-81-9, 1613-94-1, 40339-44-4
4-nitro-N-(phenylmethylene)benzenamine

| Conditions | Yield |
|---|---|
| In toluene at 120℃; for 24h; | 100% |
| With aqueous extract of pericarp of Sapindus trifoliatus fruits at 20℃; for 0.0333333h; | 98% |
| sodium hydrogen sulfate; silica gel at 52 - 54℃; for 0.0222222h; microwave irradiation; | 95% |

| Conditions | Yield |
|---|---|
| With NMM In dichloromethane | 100% |
| With triethylamine In dichloromethane at 20℃; | 71% |
| With sodium hydroxide; acetone |
-
-
100-01-6
4-nitro-aniline

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
734-25-8
N-(4-nitrophenyl)-4-methylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 4℃; | 100% |
| With (Na1752K0.144Ca0365Mg0.065)(Al2044Si2774O96)*19.16H2O In ethanol at 25 - 30℃; for 2h; Sonication; Green chemistry; | 96% |
| With pyridine In dichloromethane at 20℃; for 12h; | 94% |
-
-
100-01-6
4-nitro-aniline

-
-
625-36-5
2-chloropropionyl chloride

-
-
19313-88-3
3-chloro-N-(4-nitrophenyl)propanamide

| Conditions | Yield |
|---|---|
| at 0 - 50℃; Neat (no solvent); | 100% |
| at 20 - 50℃; | 100% |
| at 50℃; for 16h; | 97% |
-
-
100-01-6
4-nitro-aniline

-
-
87-13-8
diethyl 2-ethoxymethylenemalonate

-
-
78596-42-6
ethyl α-ethoxycarbonyl-β-(4-nitroanilino)acrylate

| Conditions | Yield |
|---|---|
| at 120℃; for 3h; Heating / reflux; | 100% |
| for 0.025h; Gould-Jacob reaction; Irradiation; | 98% |
| In ethanol at 28℃; for 6h; | 95% |

| Conditions | Yield |
|---|---|
| With potassium iodide; 10-camphorsulfonic acid; sodium nitrite In acetic acid at 20℃; for 24h; Time; | 100% |
| With Nitrogen dioxide; sodium iodide In acetonitrile at -20℃; for 10h; | 98% |
| Stage #1: 4-nitro-aniline With toluene-4-sulfonic acid In water at 20℃; Stage #2: With potassium iodide In water at 20℃; for 1.5h; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-aniline With toluene-4-sulfonic acid; sodium nitrite In water at 20℃; for 0.333333h; Stage #2: With sodium azide In water at 20℃; Reagent/catalyst; | 100% |
| Stage #1: 4-nitro-aniline With trifluoroacetic acid; sodium nitrite In water at 0℃; for 0.166667h; Stage #2: With sodium azide In water at 20℃; for 5h; | 99% |
| Stage #1: 4-nitro-aniline With hydrogenchloride In water at 0℃; Stage #2: With sodium nitrite In water for 0.166667h; Further stages; | 99% |

| Conditions | Yield |
|---|---|
| With nitrosylchloride under 750.075 Torr; for 24h; | 100% |
| With sodium nitrite In hydrogenchloride; water at 0.2℃; Rate constant; | |
| With hydrogenchloride; sodium nitrite In hydrogenchloride; water at 25℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With t-butyl thionitrite; copper dichloride In acetonitrile at 25℃; for 2h; | 100% |
| With t-butyl thionitrite; copper dichloride In acetonitrile at 25℃; for 0.0333333h; Product distribution; Mechanism; var. temperature, var. time, other copper(II) halides, other reagents, other substituted aniline derivatives; | 98% |
| Stage #1: 4-nitro-aniline With tert.-butylnitrite In acetonitrile at 20℃; Sandmeyer Reaction; Flow reactor; Stage #2: With copper dichloride In ethylene glycol; acetonitrile at 82℃; Sandmeyer Reaction; Flow reactor; | 91% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; carbon monoxide; triphenylphosphine In water; acetic acid at 56℃; under 532 Torr; for 11h; | 100% |
| With triethylsilane; palladium dichloride In ethanol at 20℃; for 1h; Inert atmosphere; | 100% |
| With 10% palladium on activated charcoal; hydrogen for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate; water; sodium nitrite for 0.0333333h; Diazotization; coupling; microwave irradiation; | 100% |
| With hydrogenchloride; sodium nitrite In water at 20℃; for 1h; Time; | 99% |
| With hydrogenchloride; sodium nitrite In water at 20℃; for 0.0833333h; Reagent/catalyst; Temperature; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-aniline With hydrogenchloride; sodium nitrite In water at 2℃; for 1.5h; Stage #2: phenol With sodium hydroxide In water at 2 - 20℃; for 4h; | 100% |
| Stage #1: 4-nitro-aniline With carboxyl- and nitrite- functionalized graphene quantum dots at 20℃; for 0.0833333h; Stage #2: phenol In water at 20℃; for 0.166667h; | 96% |
| Stage #1: 4-nitro-aniline With hydrogenchloride In water; acetone at 0℃; Inert atmosphere; Stage #2: With sodium nitrite In water; acetone at 0℃; for 1h; Inert atmosphere; Stage #3: phenol With sodium carbonate; sodium hydroxide In water; acetone at 0 - 20℃; for 3h; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium acetate; sodium nitrite In acetic acid at 0℃; for 1h; | 100% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
100-01-6
4-nitro-aniline

-
-
15450-66-5
N-(4-methoxybenzylidene)-4-nitroaniline

| Conditions | Yield |
|---|---|
| With pyrrolidine In dichloromethane at 20℃; for 16h; Molecular sieve; | 100% |
| With TiO2 nanotubes at 20℃; for 8h; Neat (no solvent); Sunlight; | 98% |
| sodium hydrogen sulfate; silica gel at 64 - 66℃; for 0.025h; microwave irradiation; | 96% |
-
-
117452-48-9, 117475-50-0
6,7-dideoxy-α,β-D-galacto-hept-6-enopyranose

-
-
100-01-6
4-nitro-aniline

-
-
128357-86-8
N-p-nitrophenyl-6,7-dideoxy-L-galacto-hept-6-enosylamine

| Conditions | Yield |
|---|---|
| In methanol; acetic acid at 110℃; for 0.166667h; | 100% |
-
-
100-01-6
4-nitro-aniline

-
-
103-80-0
phenylacetyl chloride

-
-
13140-77-7
N-(4-nitrophenyl)-2-phenylacetamide

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran at 0 - 20℃; for 2h; | 100% |
| With potassium phosphate In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; | 95% |
| In dichloromethane at 20℃; for 1h; | 70% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 0.25h; | 100% |
| In tetrahydrofuran at 20℃; for 3h; Schlenk technique; Inert atmosphere; | 77% |
| In tetrahydrofuran |
-
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| With Nitrogen dioxide In 1,4-dioxane at 10 - 40℃; | 100% |
| With Nitrogen dioxide at 0℃; for 6h; | 92% |
| With Nitrogen dioxide; N,N-dimethyl-formamide In N,N-dimethyl-formamide for 0.0833333h; | 91% |
| With tributyl phosphate-NO2 In various solvent(s) at 10 - 15℃; | 90% |
| With polyethylene glycol - NO2 In various solvent(s) | 84% |
-
-
33842-02-3, 529510-96-1
dichloromethylenedimethyliminium chloride

-
-
100-01-6
4-nitro-aniline

| Conditions | Yield |
|---|---|
| at -20℃; for 1h; ball mill; | 100% |
-
-
104-88-1
4-chlorobenzaldehyde

-
-
100-01-6
4-nitro-aniline

-
-
5340-14-7
(4-chlorobenzylidene)(4-nitrophenyl)amine

| Conditions | Yield |
|---|---|
| for 36h; Ambient temperature; | 100% |
| In neat (no solvent) at 20℃; for 72h; | 80% |
| In ethanol for 2h; Reflux; | 76% |
-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
104-94-9
4-methoxy-aniline

-
-
100-01-6
4-nitro-aniline

-
-
97221-15-3
(E)-N-(4-methoxybenzylidene)-4-nitroaniline

| Conditions | Yield |
|---|---|
| In dichloromethane Condensation; Heating; | 100% |


| Conditions | Yield |
|---|---|
| In dichloromethane Condensation; Heating; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane Condensation; Heating; | 100% |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
18144-47-3
tert-butyl 4-aminobenzoate

-
-
100-01-6
4-nitro-aniline

-
-
301317-92-0
1-(4'-nitrophenyl)-3-(4''-tert-butylcarboxyphenyl)urea

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; 4-nitro-aniline With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 0 - 20℃; for 0.833333h; Substitution; Stage #2: tert-butyl 4-aminobenzoate In tetrahydrofuran at 20℃; for 42h; Substitution; | 100% |

-
-
100-01-6
4-nitro-aniline

-
-
14628-57-0
4-chloro-5-methoxy-2-methyl-2H-pyridazin-3-one

| Conditions | Yield |
|---|---|
| With potassium carbonate; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; palladium diacetate In toluene at 120℃; for 7h; Substitution; Amination; | 100% |
4-Nitroaniline Specification
The 4-Nitroaniline with CAS registry number of 100-01-6 is also known as 1-Amino-4-nitrobenzene. The IUPAC name and product name are the same. It belongs to product categories of Anilines, Aromatic Amines and Nitro Compounds; Organics; Aniline; Functional Materials; Organic Nonlinear Optical Materials; NA - NI; Alpha Sort; Amines; AromaticsVolatiles/ Semivolatiles; Chemical Class; N; N-OAlphabetic; Analytical Standards; AromaticsChemical Class; NA - NIAnalytical Standards; Nitro Compounds; Indicators for non-aqueous titrationsTitration;Metal Titration Indicators; Indicators; Titration; I-N, Puriss p.a.Spectroscopy; Analytical Reagents for General Use; Mass Spectrometry (MS)&LC-MS; Puriss p.a.; Reagents for Mass Spectrometry (MS); C2 to C6Photonic and Optical Materials; NLO Chromophores and Intermediates; Non-Linear Optical (NLO) Materials; C2 to C6Stains and Dyes; Nitrogen Compounds; Stains&Dyes, A to. Its EINECS registry number is 202-810-1. In addition, the formula is C6H6N2O2 and the molecular weight is 138.12. This chemical is a yellow solid with a mild odor and should be sealed in ventilated, cool room away from fire and heat. Besides, it is slightly soluble in cold water, soluble in boiling water, ethanol, ether, benzene and acid.
Physical properties about 4-Nitroaniline are: (1)ACD/LogP: 1.20; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.195; (4)ACD/LogD (pH 7.4): 1.195; (5)ACD/BCF (pH 5.5): 4.769; (6)ACD/BCF (pH 7.4): 4.769; (7)ACD/KOC (pH 5.5): 106.469; (8)ACD/KOC (pH 7.4): 106.472; (9)#H bond acceptors: 4; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 2; (12)Index of Refraction: 1.634; (13)Molar Refractivity: 37.034 cm3; (14)Molar Volume: 103.555 cm3; (15)Surface Tension: 60.318 dyne/cm; (16)Density: 1.334 g/cm3; (17)Flash Point: 165 °C; (18)Enthalpy of Vaporization: 57.588 kJ/mol; (19)Boiling Point: 333.051 °C at 760 mmHg; (20)Vapour Pressure: 0 mmHg at 25 °C.
Preparation of 4-Nitroaniline: it is prepared by nitration, hydrolysis reaction of acetanilide.
C6H4ClNO2 + 2 NH3 → NH4Cl + C6H6N2O2
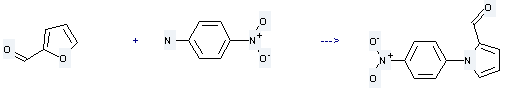
Uses of 4-Nitroaniline. This chemical is commonly used as an intermediate in the synthesis of dyes, antioxidants, pharmaceuticals and gasoline, in gum inhibitors, poultry medicines, and as a corrosion inhibitor. What's more, it can be used to produce 1-(4-nitro-phenyl)-pyrrole-2-carbaldehyde by reaction with furfural. The reaction occurs with reagent 2n-HCl and solution ethanol with other condition of heating for 2 hours. The yield is about 90%.
When you are using this chemical, please be cautious about it. As a chemical, it is harmful to aquatic organisms that may cause long-term adverse effects in the aquatic environment. Beside, it has danger of very serious irreversible effects through inhalation, in contact with skin and if swallowed. Furthermore, it has danger of cumulative effects and it is highly flammable. During using it, wear suitable protective clothing, gloves and keep away from sources of ignition. In case of accident or if you feel unwell seek medical advice immediately. After contact with skin, wash immediately. Afer using it, keep container tightly closed, locked up and out of the reach of children. Avoid release to the environment.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C1=CC(=CC=C1N)[N+](=O)[O-]
2. InChI: InChI=1S/C6H6N2O2/c7-5-1-3-6(4-2-5)8(9)10/h1-4H,7H2
3. InChIKey: TYMLOMAKGOJONV-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| bird - wild | LD50 | oral | 75mg/kg (75mg/kg) | Toxicology and Applied Pharmacology. Vol. 21, Pg. 315, 1972. | |
| guinea pig | LD50 | oral | 450mg/kg (450mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Voprosy Kommunal'noi Gigieny. Problems of Communal Hygiene. Vol. 6, Pg. 89, 1966. |
| guinea pig | LD50 | skin | > 500mg/kg (500mg/kg) | National Technical Information Service. Vol. OTS0572389, | |
| mammal (species unspecified) | LDLo | intravenous | 40mg/kg (40mg/kg) | BLOOD: METHEMOGLOBINEMIA-CARBOXYHEMOGLOBIN | U.S. Public Health Service, Public Health Bulletin. Vol. 271, Pg. 34, 1941. |
| mouse | LD50 | intramuscular | 800mg/kg (800mg/kg) | Igiena. Vol. 15, Pg. 151, 1966. | |
| mouse | LD50 | intraperitoneal | 250mg/kg (250mg/kg) | National Technical Information Service. Vol. AD691-490, | |
| mouse | LD50 | oral | 810mg/kg (810mg/kg) | Toxicology and Applied Pharmacology. Vol. 42, Pg. 417, 1977. | |
| quail | LD50 | oral | 1gm/kg (1000mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 12, Pg. 355, 1983. | |
| rat | LD50 | oral | 750mg/kg (750mg/kg) | Ceskoslovenska Hygiena. Czechoslovak Hygiene. Vol. 23, Pg. 168, 1978. | |
| rat | LDLo | intraperitoneal | 600mg/kg (600mg/kg) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 92, 1982. |
Related Products
- 4-Nitroaniline
- 100016-62-4
- 100021-45-2
- 100021-46-3
- 100021-85-0
- 1000269-51-1
- 100-02-7
- 1000279-69-5
- 1000-30-2
- 1000306-34-2
- 1000335-21-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View