-
Name
1-(5-Fluoro-2-hydroxyphenyl)-1-ethanone
- EINECS 206-893-5
- CAS No. 394-32-1
- Article Data42
- CAS DataBase
- Density 1.247 g/cm3
- Solubility 0.68 g/L in water
- Melting Point 56-58 °C(lit.)
- Formula C8H7FO2
- Boiling Point 182.2 °C at 760 mmHg
- Molecular Weight 154.141
- Flash Point 97.7 °C
- Transport Information
- Appearance light yellow to beige-brown crystals
- Safety 26-37/39-36/37/39
- Risk Codes 36/37/38-22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Acetophenone,5'-fluoro-2'-hydroxy- (6CI,7CI,8CI);1-(5-fluoro-2-hydroxyphenyl)ethanone;2-Acetyl-4-fluorophenol;2'-Hydroxy-5'-fluoroacetophenone;3-Fluoro-6-hydroxyacetophenone;NSC 46624;
- PSA 37.30000
- LogP 1.73390
Synthetic route

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride at 140℃; for 3h; Fries Phenol Ester Rearrangement; | 100% |
| With trifluorormethanesulfonic acid at 0 - 20℃; for 16h; Fries rearrangement; regiospecific reaction; | 92% |
| With hydrogenchloride; aluminum (III) chloride In water at 140 - 145℃; Fries Phenol Ester Rearrangement; | 92% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride at 130℃; for 2h; | 98.4% |
| With aluminium trichloride at 130℃; for 2h; | 89% |
| With aluminium trichloride at 180℃; for 0.5h; | 65% |
| Stage #1: 4-Fluorophenol; acetyl chloride In toluene at 40℃; Stage #2: With aluminium trichloride In toluene at 80℃; Further stages.; | |
| Stage #1: 4-Fluorophenol; acetyl chloride In neat (no solvent) at 0 - 20℃; for 2h; Stage #2: With aluminum (III) chloride In neat (no solvent) at 130℃; for 2h; |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at -5 - 25℃; | 90% |
| With pyridine hydrochloride | |
| With boron tribromide In dichloromethane at -78 - 23℃; for 1h; |

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium hydride In N,N-dimethyl acetamide at 25℃; for 4h; Inert atmosphere; | 82% |

| Conditions | Yield |
|---|---|
| With aluminum oxide; methanesulfonic acid at 120℃; for 2h; | 60% |
| With boron trifluoride at 150℃; unter Druck; |
-
-
459-60-9
1-fluoro-4-methoxybenzene

-
-
75-36-5
acetyl chloride

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| With aluminium trichloride In tetrachloromethane | 44.1% |
-
-
459-60-9
1-fluoro-4-methoxybenzene

-
-
75-36-5
acetyl chloride

-
A
-
445-82-9
1-(5-fluoro-2-methoxyphenyl)ethanone

-
B
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| With carbon disulfide; aluminium trichloride | |
| Stage #1: 1-fluoro-4-methoxybenzene; acetyl chloride With aluminum (III) chloride In dichloromethane Friedel-Crafts Acylation; Inert atmosphere; Stage #2: With water; acetic acid; sodium hydroxide at 0 - 8℃; Cooling with ice; Overall yield = 97%; Overall yield = 13.0g; |

-
-
7446-70-0
aluminium trichloride

-
-
405-51-6
4-fluorophenyl acetate

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| at 150℃; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: AlCl3 / 130 °C View Scheme | |
| Multi-step reaction with 2 steps 1: H2SO4 / 120 °C 2: AlCl3 / 160 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 84.55 percent / pyridine / CH2Cl2 / 5 °C 2: 44 percent / aluminium chloride / 2 h / 140 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 80 percent / conc. H2SO4 2: 85 percent / AlCl3 / 2 h / 160 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: AlCl3; benzene 2: 100 °C 3: AlCl3 / 150 °C View Scheme | |
| Multi-step reaction with 2 steps 1: AlCl3; CS2 2: pyridine hydrochloride View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: AlCl3; benzene 2: 100 °C 3: AlCl3 / 150 °C View Scheme |
-
-
189628-38-4
3-fluoro-6-methoxybenzonitrile

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: tetrahydrofuran / 6 h / Reflux 1.2: 20 h / 75 °C 2.1: boron tribromide / dichloromethane / 1 h / -78 - 23 °C View Scheme |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
874388-48-4
2-acetyl-4-fluorophenyl triflate

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 20℃; | 100% |
| With pyridine at 0 - 20℃; for 12h; | 92% |
| With pyridine at 0 - 20℃; | 68% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
105-39-5
chloroacetic acid ethyl ester

-
-
34849-57-5
(2-acetyl-4-fluorophenoxy)acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 16h; Reflux; Inert atmosphere; | 100% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
623-48-3
ethyl iodoacetae

-
-
34849-57-5
(2-acetyl-4-fluorophenoxy)acetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; acetone | 99% |
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 20℃; for 1h; Cooling with ice; | 99% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
70978-39-1
5'-fluoro-2'-hydroxy-3'-nitroacetophenone

| Conditions | Yield |
|---|---|
| With nitric acid; acetic acid In acetic acid at 0 - 20℃; for 3h; | 98.48% |
| With nitric acid In tetrachloromethane at 20℃; Reflux; | 97% |
| With nitric acid; acetic acid at 0 - 20℃; for 16h; | 89% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
77-78-1
dimethyl sulfate

-
-
445-82-9
1-(5-fluoro-2-methoxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 18h; Reflux; | 98% |
-
-
6165-76-0
propargyl p-toluenesulfonate

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
879216-16-7
1-(5-fluoro-2-(prop-2-yn-1-yloxy)phenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| With 2,3,5,6,8,9,11,12,14,15-decahydro-1,4,7,10,13,16-benzohexaoxacyclooctadecin; potassium carbonate In acetonitrile Heating; | 98% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride In pyridine; methanol at 20℃; for 14h; | 98% |
| With hydroxylamine hydrochloride; sodium acetate In methanol at 78℃; for 4h; | 88% |
| With hydroxylamine hydrochloride; sodium acetate In methanol at 70℃; for 4h; |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 65 - 70℃; Claisen Condensation; | 97% |
| With sodium ethanolate In ethanol at 80℃; for 1.33333h; Cooling with ice; |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
100-07-2
4-methoxy-benzoyl chloride

-
-
850799-88-1
1-(5-fluoro-2-hydroxyphenyl)-3-(4-methoxyphenyl)propane-1,3-dione

| Conditions | Yield |
|---|---|
| Stage #1: 1-(5-fluoro-2-hydroxyphenyl)ethan-1-one With lithium hexamethyldisilazane In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #2: 4-methoxy-benzoyl chloride In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 96.31% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
106-95-6
allyl bromide

-
-
1092306-36-9
1-[2-(allyloxy)-5-fluorophenyl]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In 4-methyl-2-pentanone for 8h; Reflux; | 96% |
| With potassium carbonate In acetone Reflux; |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 1-(5-fluoro-2-hydroxyphenyl)ethan-1-one With (3aR)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole In tetrahydrofuran at 0℃; for 0.333333h; Inert atmosphere; Stage #2: With borane-THF In tetrahydrofuran for 0.5h; Inert atmosphere; Heating; | 95% |
| With (-)-diisopinocamphenylborane chloride In tetrahydrofuran for 12h; Ambient temperature; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 16h; | 95% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
403-43-0
4-fluorobenzoyl chloride

-
-
1225785-23-8
1-(5-fluoro-2-hydroxyphenyl)-3-(4-fluorophenyl)propane-1,3-dione

| Conditions | Yield |
|---|---|
| Stage #1: 1-(5-fluoro-2-hydroxyphenyl)ethan-1-one With lithium hexamethyldisilazane In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #2: 4-fluorobenzoyl chloride In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 94.5% |
-
-
74-96-4
ethyl bromide

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
1466-79-1
2'-ethoxy-5'-fluoroacetophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 94% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
37050-18-3
5-chloro-1-methyl-1H-imidazole-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol Ambient temperature; | 93% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
446-52-6
2-Fluorobenzaldehyde

-
-
224294-29-5
3-(2-fluorophenyl)-1-(5-fluoro-2-hydroxyphenyl)-2-propen-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol | 93% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
74-88-4
methyl iodide

-
-
445-82-9
1-(5-fluoro-2-methoxyphenyl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 93% |
| With potassium carbonate In acetone at 20℃; |
-
-
437-81-0
2,6-difluorobenzaldehyde

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
224294-27-3
3-(2,6-difluorophenyl)-1-(5-fluoro-2-hydroxyphenyl)-2-propen-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol | 92% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
120-92-3
cyclopentanone

-
-
111478-02-5
6-fluorospiro[chroman-2,1'-cyclopentan]-4-one

| Conditions | Yield |
|---|---|
| With pyrrole In toluene for 20h; Reflux; | 92% |
| pyrrolidine In acetonitrile |

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
209810-53-7, 209743-84-0
2-acetyl-4-fluorophenolato-[o-(di-o-tolylphosphino)benzyl]palladium(II)

| Conditions | Yield |
|---|---|
| With potassium tert-butoxide In toluene Ar-atmosphere; stirring (12 h); filtering, evapn. (vac.), crystn. (toluene / pentane); elem. anal.; | 92% |

| Conditions | Yield |
|---|---|
| With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; triethylamine In acetic acid butyl ester; ethyl acetate at 120℃; Perkin condensation; | 92% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
105-58-8
Diethyl carbonate

-
-
1994-13-4
6-fluoro-4-hydroxycoumarin

| Conditions | Yield |
|---|---|
| With sodium hydride In mineral oil at 0 - 100℃; | 91% |
| With potassium tert-butylate In tetrahydrofuran at 20℃; for 10h; Inert atmosphere; | 78% |
| With sodium hydride In dimethyl sulfoxide at 0 - 100℃; for 2h; | 29% |

| Conditions | Yield |
|---|---|
| With C19H25F6N3S In toluene at 0℃; for 96h; Aldol Addition; Inert atmosphere; enantioselective reaction; | 91% |
-
-
63944-31-0
6-chloro-8-formyl-1,3-benzodioxane

-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
147722-92-7
(E)-3-(6-Chloro-4H-benzo[1,3]dioxin-8-yl)-1-(5-fluoro-2-hydroxy-phenyl)-propenone

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol Ambient temperature; | 90% |
-
-
394-32-1
1-(5-fluoro-2-hydroxyphenyl)ethan-1-one

-
-
459-57-4
4-fluorobenzaldehyde

-
-
224294-26-2
(E)-1-(2-hydroxy-5-fluorophenyl)-3-(4-fluorophenyl)prop-2-en-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol | 90% |
| With potassium hydroxide In ethanol; water at 20℃; Claisen-Schmidt Condensation; | 80% |
| With sodium hydroxide In ethanol; water at 30℃; for 24h; | 19% |

| Conditions | Yield |
|---|---|
| In toluene at 80℃; Sealed tube; Inert atmosphere; | 90% |
| at 95℃; for 4h; | 69% |
| at 100℃; for 2h; |
5'-Fluoro-2'-hydroxyacetophenone Specification
The IUPAC name of Ethanone,1-(5-fluoro-2-hydroxyphenyl)- is 1-(5-fluoro-2-hydroxyphenyl)ethanone. With the CAS registry number 394-32-1, it is also named as 5'-Fluoro-2'-hydroxyacetophenone. The product's categories are Alcohols and Derivatives; Carbonyl Compounds; Aromatic Acetophenones & Derivatives (substituted); ketone; (intermediate of nebivolol); Benzenes; Adehydes, Acetals & Ketones; Fluorine Compounds; Phenols; C7 to C8; Carbonyl Compounds; Ketones; Acetophenone Series. Besides, it is light yellow to beige-brown crystals, which is soluble in water. In addition, its molecular formula is C8H7FO2 and molecular weight is 154.14.
The other characteristics of this product can be summarized as: (1)EINECS: 206-893-5; (2)ACD/LogP: 2.30; (3)# of Rule of 5 Violations: 0; (4)ACD/LogD (pH 5.5): 2.3; (5)ACD/LogD (pH 7.4): 2.3; (6)ACD/BCF (pH 5.5): 33.1; (7)ACD/BCF (pH 7.4): 33.04; (8)ACD/KOC (pH 5.5): 426.07; (9)ACD/KOC (pH 7.4): 425.36; (10)H bond acceptors: 2; (11)H bond donors: 1; (12)Freely Rotating Bonds: 2; (13)Index of Refraction: 1.529; (14)Molar Refractivity: 38.15 cm3; (15)Molar Volume: 123.6 cm3; (16)Surface Tension: 41.9 dyne/cm; (17)Density: 1.247 g/cm3; (18)Flash Point: 97.7 °C; (19)Melting point: 54-59 °C; (20)Water solubility: soluble 0.68 g/L; (21)Enthalpy of Vaporization: 43.56 kJ/mol; (22)Boiling Point: 182.2 °C at 760 mmHg; (23)Vapour Pressure: 0.603 mmHg at 25 °C.
Preparation of Ethanone,1-(5-fluoro-2-hydroxyphenyl)-: this chemical can be prepared by Acetic acid-(4-fluoro-phenyl ester).

This reaction needs AlCl3 at temperature of 160 °C. The reaction time is 2 hours. The yield is 85 %.
Uses of Ethanone,1-(5-fluoro-2-hydroxyphenyl)-: it can react with 6-Chloro-4H-benzo[1,3]dioxin-8-carbaldehyde to get 3-(6-Chloro-4H-benzo[1,3]dioxin-8-yl)-1-(5-fluoro-2-hydroxy-phenyl)-propenone.
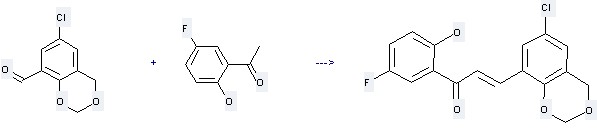
This reaction needs 50percent aq. NaOH and Ethanol at ambient temperature. The yield is 90 %.
When you are using this chemical, please be cautious about it as the following: it is harmful if swallowed. It is also irritating to eyes, respiratory system and skin. You should wear suitable protective clothing, gloves and eye / face protection when use it. Moreover, in case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
People can use the following data to convert to the molecule structure.
(1)SMILES:O=C(c1cc(F)ccc1O)C
(2)InChI:InChI=1/C8H7FO2/c1-5(10)7-4-6(9)2-3-8(7)11/h2-4,11H,1H3
(3)InChIKey:KOFFXZYMDLWRHX-UHFFFAOYAY
(4)Std. InChI:InChI=1S/C8H7FO2/c1-5(10)7-4-6(9)2-3-8(7)11/h2-4,11H,1H3
(5)Std. InChIKey:KOFFXZYMDLWRHX-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | > 1gm/kg (1000mg/kg) | Medicina Experimentalis. Vol. 11, Pg. 137, 1964. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View