-
Name
5-Bromo-2-pyridinecarbonitrile
- EINECS -0
- CAS No. 97483-77-7
- Article Data32
- CAS DataBase
- Density 1.72 g/cm3
- Solubility
- Melting Point 128-132 °C(lit.)
- Formula C6H3BrN2
- Boiling Point 260.3 °C at 760 mmHg
- Molecular Weight 183.007
- Flash Point 111.2 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance light yellow cryst
- Safety 26-36/37/39-36/37-9
- Risk Codes 22-37/38-41-36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, T,
T, Xi
Xi
- Synonyms 2-Cyano-5-Bromopyridin;2-Cyano-5-Bromo pyridine;5-Bromo-2-cyanopyridine 97483-77-7;LDP05:5-Bromo-2-Cyanopyridine;5-bromopicolinonitrile;5-bromopyridine-2-carbonitrile;5-Bromo-2-cyanopyridine;
- PSA 36.68000
- LogP 1.71578
Synthetic route
-
-
31181-90-5
5-bromo-pyridine-2-carbaldehyde

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; iodine In tetrahydrofuran at 20℃; for 4h; | 100% |
| With ammonia; iodine In tetrahydrofuran; water at 20℃; for 0.25h; | 85% |
-
-
2402-97-3
3-bromopyridine N-oxide

-
-
7677-24-9
trimethylsilyl cyanide

-
A
-
55758-02-6
3-bromopyridine-2-carbonitrile

-
B
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 4h; Heating; | A 86% B 9% |
| With triethylamine In acetonitrile for 4h; Product distribution / selectivity; Reflux; | |
| With triethylamine In acetonitrile for 4h; Heating / reflux; |


| Conditions | Yield |
|---|---|
| In sulfolane at 160℃; for 6h; Temperature; Solvent; | 83% |
| In N,N-dimethyl-formamide at 150℃; for 7h; | 70% |

-
-
19158-51-1
P-toluenesulfonyl cyanide

-
-
223463-13-6
2-iodo-5-bromopyridine

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2-iodo-5-bromopyridine With isopropylmagnesium chloride In tetrahydrofuran; dichloromethane at 0℃; for 0.75h; Stage #2: P-toluenesulfonyl cyanide In tetrahydrofuran; dichloromethane at 20℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 110 - 120℃; for 12h; | 74% |
| With sodium cyanide In N,N-dimethyl-formamide for 3h; Heating; | 65% |
| In 1-methyl-pyrrolidin-2-one at 110℃; for 20h; | 28% |
| In 1-methyl-pyrrolidin-2-one at 110℃; for 20h; | 28% |
-
-
624-28-2
2,5-dibromopyridine

-
-
143-33-9
sodium cyanide

-
-
544-92-3
copper(I) cyanide

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| In DMF (N,N-dimethyl-formamide) at 20 - 150℃; for 7h; | 70% |


| Conditions | Yield |
|---|---|
| With sodium cyanide In N,N-dimethyl-formamide at 150℃; for 7h; Inert atmosphere; | 70% |
-
-
626-55-1
3-Bromopyridine

-
-
7677-24-9
trimethylsilyl cyanide

-
A
-
55758-02-6
3-bromopyridine-2-carbonitrile

-
B
-
13958-98-0
3-bromo-4-cyanopyridine

-
C
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromopyridine With trifluoromethylsulfonic anhydride In chloroform at 20℃; for 1h; Inert atmosphere; Sealed tube; Stage #2: trimethylsilyl cyanide In chloroform at 60℃; for 3h; Inert atmosphere; Sealed tube; Stage #3: With 4-methyl-morpholine In chloroform at 60℃; for 17h; Inert atmosphere; Sealed tube; Overall yield = 79 %; | A 13% B 55% C 11% |


| Conditions | Yield |
|---|---|
| With copper(l) cyanide In N,N-dimethyl-formamide at 150℃; for 5h; | 53% |
-
-
624-28-2
2,5-dibromopyridine

-
-
773837-37-9
sodium cyanide

-
-
544-92-3
copper(I) cyanide

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 150℃; for 5h; | 53% |


| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 110℃; for 20h; | 28% |
| In 1-methyl-pyrrolidin-2-one at 110℃; for 20h; | 28% |

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With trichlorophosphate In toluene at 110℃; for 5h; |
-
-
624-28-2
2,5-dibromopyridine

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With zinc In ethyl acetate; N,N-dimethyl-formamide |
-
-
624-28-2
2,5-dibromopyridine

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 85 percent / NaI; AcCl / acetonitrile / 3 h / Heating 2.1: iPrMgCl / CH2Cl2; tetrahydrofuran / 0.75 h / 0 °C 2.2: 82 percent / CH2Cl2; tetrahydrofuran / 2 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: n-butyllithium / toluene / 2 h / -78 °C 1.2: toluene / 1 h / -78 °C 2.1: POCl3 / toluene / 5 h / 110 °C View Scheme | |
| In N-methyl-acetamide | |
| With zinc(II) cyanide; zinc; (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In DMF (N,N-dimethyl-formamide) for 5h; Heating / reflux; |

| Conditions | Yield |
|---|---|
| With zinc; (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In dichloromethane; N,N-dimethyl-formamide for 5h; Heating / reflux; | |
| tetrakis(triphenylphosphine) palladium(0) In N,N-dimethyl-formamide at 135℃; for 0.25h; microwave irradiation; |
-
-
30766-11-1
5-bromoisonicotinic acid

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: thionyl chloride / 3 h / Reflux 1.2: 20 °C / Cooling with ice 2.1: trichlorophosphate / toluene / 2 h / Reflux 2.2: pH 12 / Cooling with ice View Scheme | |
| Multi-step reaction with 2 steps 1.1: thionyl chloride / 3 h / Reflux 1.2: Cooling with ice 2.1: trichlorophosphate / toluene / 2 h / Reflux View Scheme |
-
-
90145-48-5
5-bromo-2-pyridinecarboxamide

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| Stage #1: 5-bromo-2-pyridinecarboxamide With trichlorophosphate In toluene for 2h; Reflux; Stage #2: With sodium hydroxide pH=12; Cooling with ice; | |
| With trichlorophosphate In toluene for 2h; Reflux; | 43.6 g |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
41960-47-8
2-cyano-5-iodopyridine

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium iodide; N,N`-dimethylethylenediamine at 110℃; for 24h; Finkelstein reaction; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 20 - 50℃; | 100% |
| With 1-methyl-pyrrolidin-2-one; potassium carbonate at 50℃; for 0.5h; | 100% |
| With potassium carbonate In 1-methyl-pyrrolidin-2-one at 20 - 50℃; | 100% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
5188-07-8
sodium thiomethoxide

-
-
848141-12-8
5-(methylsulfanyl)pyridine-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1-methyl-pyrrolidin-2-one | 99% |
| With potassium carbonate In 1-methyl-pyrrolidin-2-one | 99% |
| With sodium t-butanolate In water; acetonitrile at 0 - 20℃; for 16h; |
-
-
1414782-20-9
tert-butyl 2-[(R)-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-2,3-dihydro-1H-inden-1-yl]-2,7-diazaspiro[3.5]nonane-7-carboxylate

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
1334785-12-4
tert-butyl 2-[(R)-5-(6-cyanopyridin-3-yl)-2,3-dihydro-1H-inden-1-yl]-2,7-diazaspiro[3.5]nonane-7-carboxylate

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In 1,4-dioxane; water Suzuki Coupling; Inert atmosphere; Heating; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 0 - 25℃; for 16h; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at -20 - 0℃; for 16h; | 99% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at -5 - 5℃; for 16h; | 99% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
380380-62-1
5-bromo-N-hydroxypyridine-2-carboxamidine

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; potassium tert-butylate In dimethyl sulfoxide at 20℃; | 98% |
| With sodium hydroxide; hydroxylamine hydrochloride In ethanol; water at 60 - 65℃; for 16h; | 64% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
444120-91-6
6-chloropyridin-3-ylboronic acid

-
-
951658-09-6
5-(2'-chloropyridin-5'-yl)-2-pyridinecarbonitrile

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In methanol; water; toluene at 80℃; Suzuki coupling; Inert atmosphere; | 98% |

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 100℃; | 98% |
| With caesium carbonate In N,N-dimethyl-formamide at 100℃; | 19 g |
-
-
5720-07-0
4-methoxyphenylboronic acid

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
135943-40-7
4-(2-Cyano-5-pyridyl)anisole

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium acetate In tetrahydrofuran; water at 80℃; for 18h; Inert atmosphere; | 98% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
1066-54-2
trimethylsilylacetylene

-
-
1214278-88-2
5-[(trimethylsilyl)ethynyl]pyridine-2-carbonitrile

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine; triphenylphosphine In tetrahydrofuran at 50℃; for 5h; Inert atmosphere; | 96% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 24h; Sonogashira coupling; Inert atmosphere; | 86% |

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); caesium carbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In 1,4-dioxane at 95℃; for 16h; Buchwald-Hartwig Coupling; | 96% |
-
-
1149-23-1
diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
A
-
1149-24-2
2,6-dimethyl-pyridine-3,5-dicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine nickel(II) bromide In N,N-dimethyl acetamide at 20℃; for 24h; Mechanism; Irradiation; Inert atmosphere; | A 96% B 72% |

-
-
6269-89-2
1-(4-Nitrophenyl)piperazine

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; ruphos In 1,4-dioxane at 90℃; for 4h; Sealed tube; | 94% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
380380-62-1
2-(2-amino-1-oxime)-5-bromopyridine

| Conditions | Yield |
|---|---|
| With hydroxylamine; sodium hydrogencarbonate In ethanol for 2h; Heating; | 93% |
-
-
75336-86-6
(2R)-methylpiperazine

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
946399-86-6
1-[(3R)-3-methylpiperazin-1-yl]pyridine-2-carbonitrile

| Conditions | Yield |
|---|---|
| With modified hectorite-loaded ionic liquid In dimethyl sulfoxide at 100 - 110℃; for 8h; Inert atmosphere; | 93% |
| With sodium t-butanolate; tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In toluene at 110℃; for 0.583333h; Microwave irradiation; | 39.1% |
| With sodium t-butanolate; tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In toluene at 110℃; for 0.583333h; Microwave irradiation; | 39.1% |

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); dipotassium hydrogenphosphate; Ir(dF(CF3)ppy)2(bpy)PF6; 4,4'-di-tert-butyl-2,2'-bipyridine In 1,4-dioxane at 24℃; for 24h; Inert atmosphere; Irradiation; Sealed tube; | 93% |

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate; CyJohnPhos In toluene at 100℃; for 24h; Inert atmosphere; | 93% |
-
-
75-89-8
2,2,2-trifluoroethanol

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With C43H59NO2PPd(1+)*CH3O3S(1-); caesium carbonate In toluene at 80℃; Schlenk technique; Inert atmosphere; | 93% |
-
-
4612-26-4
1,4-Phenyldiboronic acid

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
951657-74-2
1,4-bis-(2'-cyanopyridin-5'-yl)phenylene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In methanol; water; toluene at 80℃; Suzuki coupling; Inert atmosphere; | 92% |
-
-
504438-22-6
(R)‐3‐(3‐fluoro‐4‐(4,4,5,5‐tetramethyl‐1,3,2‐dioxaborolan‐2‐yl)phenyl)‐5‐(hydroxymethyl)‐oxazolidin‐2‐one

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In 1,4-dioxane; ethanol; water at 90 - 95℃; for 3h; Inert atmosphere; | 92% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
380380-60-9
5-bromo-2-(2H-tetrazol-5-yl)pyridine

| Conditions | Yield |
|---|---|
| With pyridine; sodium azide; zinc(II) chloride at 40 - 120℃; for 2h; | 91.8% |
| With sodium azide; ammonium chloride In N,N-dimethyl-formamide at 120℃; for 1h; | 84% |
| With sodium azide; ammonium chloride In DMF (N,N-dimethyl-formamide) at 120℃; for 1h; |
-
-
75-16-1
methylmagnesium bromide

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
871723-90-9
1-(5-bromopyridin-2-yl)ethanamine

| Conditions | Yield |
|---|---|
| Stage #1: methylmagnesium bromide; 2-Cyano-5-bromopyridine In tetrahydrofuran at 0 - 20℃; for 0.5h; Stage #2: With sodium tetrahydroborate In tetrahydrofuran; methanol for 10h; | 91% |
| Stage #1: methylmagnesium bromide; 2-Cyano-5-bromopyridine In tetrahydrofuran at -20 - 0℃; Inert atmosphere; Stage #2: With sodium tetrahydroborate In tetrahydrofuran; methanol at 20℃; for 10h; Stage #3: With sodium hydroxide In tetrahydrofuran; methanol; water | 59% |
| Stage #1: methylmagnesium bromide; 2-Cyano-5-bromopyridine In tetrahydrofuran at -20 - 20℃; for 0.5h; Stage #2: With methanol; sodium tetrahydroborate In tetrahydrofuran at 20℃; for 15h; | |
| Stage #1: methylmagnesium bromide; 2-Cyano-5-bromopyridine In tetrahydrofuran at -20 - 20℃; for 1h; Inert atmosphere; Stage #2: With methanol; sodium tetrahydroborate In tetrahydrofuran for 1h; |
-
-
25932-11-0
benzenesulfinic acid sodium dihydrate

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
1424246-19-4
5-(benzenesulfonyl)pyridin-2-carbonitrile

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 120℃; for 4h; | 91% |
-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
101469-92-5
(S)-3-hydroxy-pyrrolidine-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| Stage #1: (S)-3-hydroxy-pyrrolidine-1-carboxylic acid tert-butyl ester With sodium hydride In N,N-dimethyl-formamide at 0 - 25℃; for 0.5h; Inert atmosphere; Stage #2: 2-Cyano-5-bromopyridine In N,N-dimethyl-formamide at 0 - 25℃; for 2h; Inert atmosphere; | 91% |
-
-
110-89-4
piperidine

-
-
97483-77-7
2-Cyano-5-bromopyridine

-
-
1241911-25-0
2-cyano-5-(1-piperidinyl)pyridine

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); XPhos In 1,2-dimethoxyethane at 80℃; for 24h; Inert atmosphere; | 90% |
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); XPhos In 1,2-dimethoxyethane at 80℃; sealed tube; Inert atmosphere; | 63% |
| Stage #1: 2-Cyano-5-bromopyridine With XPhos; tris-(dibenzylideneacetone)dipalladium(0) In 1,2-dimethoxyethane for 0.166667h; Inert atmosphere; Stage #2: piperidine With potassium phosphate In 1,2-dimethoxyethane at 80℃; Inert atmosphere; | 63% |

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine; bis(1,5-cyclooctadiene)nickel (0); Ir(dF(CF3)ppy)2(bpy)PF6; 4,4'-di-tert-butyl-2,2'-bipyridine In methanol; acetone at 20℃; for 24h; Irradiation; | 90% |
-
-
925-90-6
ethylmagnesium bromide

-
-
97483-77-7
2-Cyano-5-bromopyridine

| Conditions | Yield |
|---|---|
| Stage #1: ethylmagnesium bromide; 2-Cyano-5-bromopyridine In tetrahydrofuran; diethyl ether at -20 - 20℃; for 2h; Stage #2: With hydrogenchloride; water In tetrahydrofuran; diethyl ether | 90% |
| In tetrahydrofuran; diethyl ether at -20 - 20℃; for 2h; | 85% |
| Stage #1: ethylmagnesium bromide; 2-Cyano-5-bromopyridine In diethyl ether at -15 - 20℃; for 3h; Inert atmosphere; Stage #2: With hydrogenchloride In diethyl ether; water at 20℃; for 0.25h; Inert atmosphere; | 78% |
5-Bromo-2-pyridinecarbonitrile Chemical Properties
Molecular Formula: C6H3BrN2
Molecular Weight: 183.01 g/mol
Index of Refraction: 1.611
Density: 1.72 g/cm3
Flash Point: 111.2 °C
Enthalpy of Vaporization: 49.79 kJ/mol
Boiling Point: 260.3 °C at 760 mmHg
Vapour Pressure: 0.0124 mmHg at 25 °C
Melting point: 128-132 °C(lit.)
Appearance: Light yellow cryst
Structure of 5-Bromo-2-pyridinecarbonitrile (CAS NO.97483-77-7):
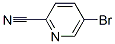
IUPAC Name: 5-Bromopyridine-2-carbonitrile
Product Category of 5-Bromo-2-pyridinecarbonitrile (CAS NO.97483-77-7): Bromides;Carboxes;Pyridine Series;Boron, Nitrile, Thio,& TM-Cpds;Heterocycles;Pyridines, Pyrimidines, Purines and Pteredines;Cyano;Organohalides;Heterocyclic Compounds;Heterocyclic Building Blocks
5-Bromo-2-pyridinecarbonitrile Uses
5-Bromo-2-pyridinecarbonitrile (CAS NO.97483-77-7) has been used for organic synthesis intermediate.
5-Bromo-2-pyridinecarbonitrile Safety Profile
Hazard Symbols:  Xn
Xn
Risk Statements:
22: Harmful if swallowed
41: Risk of serious damage to eyes
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
37/38: Irritating to respiratory system and skin
Safety Statements information:
9: Keep container in a well-ventilated place
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37: Wear suitable protective clothing and gloves
36/37/39: Wear suitable protective clothing, gloves and
5-Bromo-2-pyridinecarbonitrile Specification
5-Bromo-2-pyridinecarbonitrile , its cas register number is 97483-77-7. It also can be called 5-Bromo-2-Cyanopyridine ; 5-bromopyridine-2-carbonitrile ; and 2-Pyridinecarbonitrile, 5-bromo- .
Related Products
- 5-Bromo-2-pyridinecarbonitrile
- 97484-75-8
- 97484-76-9
- 97484-82-7
- 97492-93-8
- 97503-12-3
- 97-50-7
- 97507-71-6
- 97509-75-6
- 97511-05-2
- 97511-06-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View