-
Name
Benzofuran
- EINECS 205-982-6
- CAS No. 271-89-6
- Article Data161
- CAS DataBase
- Density 1.11 g/cm3
- Solubility Insoluble in water
- Melting Point <-18 °C
- Formula C8H6O
- Boiling Point 174 °C at 760 mmHg
- Molecular Weight 118.135
- Flash Point 56.1 °C
- Transport Information UN 1993 3/PG 3
- Appearance Colorless to light yellow liquid
- Safety 36/37-16
- Risk Codes 40-52-10
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi,  Xn
Xn
- Synonyms 1-Oxidene;1-Oxindene;2,3-Benzofuran;Coumarone;
- PSA 13.14000
- LogP 2.43280
Synthetic route

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With Grubbs catalyst first generation In dichloromethane at 20℃; for 8h; | 100% |
-
-
54008-77-4
2-bromo-1-benzofuran

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium tetrahydroborate; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran at 25℃; for 0.7h; Inert atmosphere; | 89% |
-
-
32438-31-6
(2,2-diethoxy-ethoxy)-benzene

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With polyphosphoric acid In 1,2-dichloro-ethane for 4h; Reflux; | 83.9% |
| With tin-exchanged H-b zeolite (Sn-b) for 0.5h; Reagent/catalyst; Time; Reflux; | 78% |
| With oxalic acid | |
| With polyphosphoric acid In toluene for 12h; Reflux; | |
| With polyphosphoric acid In toluene for 12h; Reflux; |

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; copper hydroxide In 1-methyl-pyrrolidin-2-one at 190℃; for 4h; Inert atmosphere; Green chemistry; | 82% |
| at 140℃; for 4h; Ionic liquid; | 71% |
| With [Au(1,3-bis(2,6-diisopropylphenyl)-4,5-dihydroimidazol-2-ylidene)(O2CAd)] In toluene at 120℃; for 16h; | 67% |
-
-
496-16-2
2,3-Dihydrobenzofuran

-
-
558-37-2
tert-butylethylene

-
A
-
271-89-6
1-benzofurane

-
B
-
75-83-2
2,2-Dimethylbutane

| Conditions | Yield |
|---|---|
| With (PSCOP)IrHCl; sodium t-butanolate In para-xylene at 120℃; for 12h; Inert atmosphere; Schlenk technique; Glovebox; Sealed tube; | A 80% B n/a |


-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 0.5h; Heating; | 76% |

| Conditions | Yield |
|---|---|
| at 75 - 80℃; for 4h; Temperature; Reagent/catalyst; | 74.5% |
| With sodium hydrogen sulfite; silica gel at 75 - 80℃; for 4h; Temperature; | 74.5% |
-
-
96853-36-0
β-(2-hydroxyphenyl)nitroethane

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| Stage #1: β-(2-hydroxyphenyl)nitroethane With sodium hydroxide at 20℃; for 0.5h; Nef reaction; Stage #2: With sulfuric acid at 0 - 20℃; Nef Reaction; | 74% |
| Stage #1: β-(2-hydroxyphenyl)nitroethane With sodium hydroxide In water at 20℃; for 0.25h; Stage #2: With sulfuric acid at 0℃; for 0.5h; | 68% |
-
-
152842-85-8
(E)-3-(2-Allyloxy-phenyl)-acrylic acid methyl ester

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| at 650℃; under 0.001 - 0.01 Torr; | 68% |

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| at 650℃; under 0.01 Torr; for 0.25h; | 68% |

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate | 67% |
| With perchloric acid In water at 25℃; Rate constant; Mechanism; |

| Conditions | Yield |
|---|---|
| at 600℃; under 0.1 Torr; flash vacuum pyrolysis; | A 65% B 3% |
-
-
59214-70-9
3-bromobenzofuran

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran at 20℃; for 6h; Reagent/catalyst; Time; Inert atmosphere; | 64% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium tetrahydroborate; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran at 25℃; for 22h; Reagent/catalyst; Inert atmosphere; | 64% |
-
-
121045-14-5
2-<2-hydroxy-1-(isopropylthio)ethyl>phenol

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 2-methoxy-ethanol for 4h; Heating; | 61% |

| Conditions | Yield |
|---|---|
| In benzene at 120℃; for 72h; Diels-Alder reaction; | 58% |

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With 2-(trimethylsilyl)phenyl trifluoromethanesulfonate; cesium fluoride In acetonitrile at 20℃; for 12h; Inert atmosphere; Glovebox; | 58% |

-
-
88284-48-4
2-(trimethylsilyl)phenyl trifluoromethanesulfonate

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile at 20℃; for 12h; Inert atmosphere; | 58% |
-
-
199010-94-1
(2-ethynylphenoxy)triisopropylsilane

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve; tetrabutyl ammonium fluoride In tetrahydrofuran at 50℃; | 56% |

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium tetrahydroborate; N,N,N,N,-tetramethylethylenediamine In tetrahydrofuran at 23℃; for 0.7h; Reagent/catalyst; Inert atmosphere; regioselective reaction; | A 31% B 56% |

| Conditions | Yield |
|---|---|
| at 650℃; under 0.001 Torr; for 1.5h; | A n/a B 55% C n/a |


| Conditions | Yield |
|---|---|
| With sodium anthraquinone-2-sulfonate; oxygen; palladium diacetate; magnesium sulfate; toluene-4-sulfonic acid In chlorobenzene at 110℃; under 760.051 Torr; for 24h; | 53% |
| With copper(I) oxide; dmap; N-hydroxyphthalimide; oxygen In acetonitrile at 120℃; for 12h; Sealed tube; | 45% |
| With [2,2]bipyridinyl; iron(III) chloride; di-tert-butyl peroxide In toluene at 135℃; for 14h; Reagent/catalyst; | 31% |
-
-
6280-80-4
2-Formylphenoxyacetic acid

-
A
-
271-89-6
1-benzofurane

-
B
-
496-41-3
Benzofuran-2-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic anhydride at 110℃; for 3h; | A 22.2% B 53% |
| With sodium acetate; acetic anhydride at 110℃; for 3h; | A 22.2% B 53% |

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With [bis(trifluoromethanesulfonyl)imidate](triphenylphosphine)gold (I) In dichloromethane at 20℃; for 2h; | 53% |

| Conditions | Yield |
|---|---|
| With triethylsilane; palladium diacetate; tricyclohexylphosphine In toluene at 100℃; for 20h; Inert atmosphere; Sealed tube; | A 8 %Chromat. B 50% |

| Conditions | Yield |
|---|---|
| With quinoline; copper; sodium hydroxide at 190℃; | 39% |
| at 860℃; durch ein verzinntes Eisenrohr; | |
| With nickel at 350℃; |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -70℃; for 2h; | A 35% B 38% |
-
-
496-16-2
2,3-Dihydrobenzofuran

-
-
558-37-2
tert-butylethylene

-
A
-
271-89-6
1-benzofurane

-
B
-
75-83-2
2,2-Dimethylbutane

-
C
-
41014-29-3
2,2'-bibenzofuranyl

| Conditions | Yield |
|---|---|
| With (PSCOP)IrHCl; sodium t-butanolate In para-xylene at 120℃; for 6h; Inert atmosphere; Schlenk technique; Glovebox; Sealed tube; | A 11% B n/a C 37% |


-
-
682356-47-4, 202598-29-6, 105445-61-2
tributyl(o-methoxystyryl)stannane

-
A
-
271-89-6
1-benzofurane

-
B
-
174535-20-7, 131428-32-5
(E)-o-methoxystyryl-lead triacetate

-
C
-
174535-20-7, 131428-32-5
(Z)-o-methoxystyryl-lead triacetate

| Conditions | Yield |
|---|---|
| In chloroform-d1 room temp., 2 min; not isolated; (1)H-NMR monitoring; | A 35% B n/a C n/a |

-
-
85267-53-4
(1α,2α,4α,5α,6α,8α)-3-Oxatetracyclo<3.3.1.02,4.06,8>nonan-9-one

-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene at 80℃; for 3h; Mechanism; | 33% |
-
-
271-89-6
1-benzofurane

-
-
69626-75-1
2-iodobenzofuran

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at -40℃; for 1h; Inert atmosphere; Stage #2: With iodine In tetrahydrofuran at 20℃; Inert atmosphere; | 100% |
| Stage #1: 1-benzofurane With 2,2,6,6-tetramethyl-piperidine; n-butyllithium; N,N,N,N,-tetramethylethylenediamine; zinc(II) chloride In tetrahydrofuran at 0 - 20℃; for 2h; Stage #2: With iodine In tetrahydrofuran for 16h; | 99% |
| Stage #1: 1-benzofurane In tetrahydrofuran; diethyl ether; hexane at -78℃; for 3h; Stage #2: With iodine In tetrahydrofuran; diethyl ether; hexane at 20℃; for 16h; Further stages.; | 95% |

| Conditions | Yield |
|---|---|
| indium(III) triflate In acetonitrile at 50℃; for 0.191667h; Product distribution / selectivity; Microwave irradiation; | 100% |
| montmorillonit KSF In acetonitrile at 50℃; for 0.191667h; Product distribution / selectivity; Microwave irradiation; | 79% |
| ytterbium(III) triflate In acetonitrile at 50℃; for 0.191667h; Product distribution / selectivity; Microwave irradiation; | 77% |


| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; Stage #2: 1-bromo dodecane In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 100% |
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran at -78℃; for 2h; Schlenk technique; Inert atmosphere; Stage #2: 1-bromo dodecane In tetrahydrofuran at 20℃; for 24h; Schlenk technique; Inert atmosphere; | 30% |

| Conditions | Yield |
|---|---|
| With methanol; palladium diacetate; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In dichloromethane at 25℃; for 12h; Sealed tube; Inert atmosphere; chemoselective reaction; | 99% |
| With hydrogen In methanol at 80 - 90℃; under 750.075 - 22502.3 Torr; Reagent/catalyst; Autoclave; | 97.8% |
| With ethanol; (BQ‑NCOP)IrHCl; sodium t-butanolate at 60℃; for 5h; Inert atmosphere; Schlenk technique; Sealed tube; chemoselective reaction; | 96% |
-
-
271-89-6
1-benzofurane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
402503-13-3
2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzo[b]furan

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butyl-2,2'-bipyridine; (1,5-cyclooctadiene)(methoxy)iridium(I) dimer In hexane at 25℃; for 1h; | 99% |
| With (iPrPNP)CoCH2SiMe3 In neat (no solvent) at 23℃; for 0.25h; Reagent/catalyst; Temperature; Inert atmosphere; Glovebox; | 99% |
| With (iPrPNP)CoCH2SiMe3 In neat (no solvent) at 23℃; for 0.25h; Reagent/catalyst; | 99% |
-
-
271-89-6
1-benzofurane

-
-
67-56-1
methanol

-
-
56-23-5
tetrachloromethane

-
-
1646-27-1
methyl benzofuran-2-carboxylate

| Conditions | Yield |
|---|---|
| With iron(II) bromide at 160℃; for 4h; Inert atmosphere; sealed ampule; regioselective reaction; | 99% |

-
-
271-89-6
1-benzofurane

-
-
592-82-5
butyl isothiocyanate

-
-
1258217-03-6
N-(n-butyl)benzofuran-2-carbothioamide

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: butyl isothiocyanate In tetrahydrofuran; hexane at -78 - 20℃; for 16h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 2h; Stage #2: formic acid ethyl ester In tetrahydrofuran; hexane at -78℃; for 3h; Stage #3: With ethanol; iodine; potassium carbonate In tetrahydrofuran; hexane at -78 - 20℃; for 13h; | 99% |

| Conditions | Yield |
|---|---|
| With impregnated palladium on magnetite catalyst PdO-Fe3O4 (10 mol percent Pd) In ethanol at 60℃; for 24h; regioselective reaction; | 99% |
| With 5%-palladium/activated carbon In ethanol at 60℃; for 22h; regioselective reaction; | 77% |
| With palladium In tetrahydrofuran at 60℃; for 24h; | 77% |
| With palladium diacetate In ethanol at 60℃; for 24h; Kinetics; Reagent/catalyst; | 31% |
-
-
271-89-6
1-benzofurane

-
-
27124-07-8
2-deuteriobenzofuran

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In diethyl ether; hexane at -20℃; for 1h; Stage #2: With water-d2 In diethyl ether; hexane at -20 - 23℃; | 99% |
| Stage #1: 1-benzofurane With n-butyllithium In diethyl ether at -20℃; for 1h; Stage #2: With water-d2 In diethyl ether at -20 - 23℃; |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; C24H36N2O2Pd*2C2H4O2; cesium trifluoroacetate; trifluoroacetic acid; p-benzoquinone; silver(l) oxide In water at 30℃; for 20h; regioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With C13H18Cl2N4OPd; lithium tert-butoxide In N,N-dimethyl-formamide at 130℃; for 24h; Catalytic behavior; Inert atmosphere; Schlenk technique; | 99% |
-
-
271-89-6
1-benzofurane

-
-
1619992-33-4
6-phenylspiro[2.5]oct-1-ene

| Conditions | Yield |
|---|---|
| With dirhodium tetraacetate In dichloromethane at 20℃; for 21h; Inert atmosphere; Sealed tube; diastereoselective reaction; | 99% |
-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With dirhodium tetraacetate In dichloromethane at 20℃; for 21h; Inert atmosphere; Sealed tube; diastereoselective reaction; | 99% |
-
-
271-89-6
1-benzofurane

-
-
93623-88-2
3-bromo-1-[(diphenylmethylene)amino]-2-piperidinone

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: 3-bromo-1-[(diphenylmethylene)amino]-2-piperidinone In tetrahydrofuran at -78℃; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With tert-butylethylene; C25H40O2P2Ru In neat (no solvent) at 120℃; for 24h; Reagent/catalyst; Solvent; Inert atmosphere; Schlenk technique; Glovebox; regioselective reaction; | 98% |
| With norborn-2-ene; (1,5-cyclooctadiene)(methoxy)iridium(I) dimer; 4,4'-di-tert-butyl-2,2'-bipyridine In tetrahydrofuran at 80℃; for 24h; Inert atmosphere; | 83% |
| With bis(norbornadiene)rhodium(l)tetrafluoroborate; 1,2-bis((2R,5R)-2,5-diisopropylphospholano)ethane; cyclohexene In neat (no solvent) at 80℃; for 20h; Solvent; Inert atmosphere; regioselective reaction; | 83% |
| With di-tert-butyl peroxide; copper diacetate In tert-butyl alcohol at 110℃; for 24h; Inert atmosphere; | 55% |
| With potassium tert-butylate In tetrahydrofuran at 60℃; for 45h; Inert atmosphere; Glovebox; regioselective reaction; | 38% |
-
-
271-89-6
1-benzofurane

-
-
67-56-1
methanol

-
-
558-13-4
carbon tetrabromide

-
-
1646-27-1
methyl benzofuran-2-carboxylate

| Conditions | Yield |
|---|---|
| With ferrocene at 140℃; for 4h; Mechanism; Temperature; Reagent/catalyst; Inert atmosphere; sealed ampule; regioselective reaction; | 98% |
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate; potassium carbonate at 20℃; for 24h; Inert atmosphere; Irradiation; regioselective reaction; | 70% |
| With Ru(2,2'-bipyridine)3(PF6)2*H2O; diisopropylamine at 20℃; for 36h; Schlenk technique; Inert atmosphere; Irradiation; regioselective reaction; | 40% |


| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With tert.-butyl lithium In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 98% |
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; | 88% |
| Stage #1: 1-benzofurane With tert.-butyl lithium In tetrahydrofuran; hexane at -78℃; for 2h; Inert atmosphere; Schlenk technique; Stage #2: methyl iodide In tetrahydrofuran; hexane at 20℃; Inert atmosphere; Schlenk technique; Cooling; | 87% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; C24H36N2O2Pd*2C2H4O2; cesium trifluoroacetate; trifluoroacetic acid; p-benzoquinone; silver(l) oxide In water at 30℃; for 20h; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel(0); C29H41N2; sodium t-butanolate In neat (no solvent) at 50℃; for 16h; Reagent/catalyst; Temperature; Glovebox; Sealed tube; | 98% |

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; 2,3-dicyano-5,6-dichloro-p-benzoquinone In 1,2-dichloro-ethane at 100℃; for 6h; Inert atmosphere; Schlenk technique; Sealed tube; | 98% |
-
-
271-89-6
1-benzofurane

-
-
94348-81-9
2-(acetoxyphenylmethyl)-cyclohex-2-enone

| Conditions | Yield |
|---|---|
| With bismuth(lll) trifluoromethanesulfonate In 1,2-dichloro-ethane at 50℃; for 0.166667h; Friedel-Crafts Alkylation; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With 2,2,6,6-tetramethyl-piperidine; n-butyllithium In tetrahydrofuran; hexane at -78 - -10℃; for 1h; Inert atmosphere; Stage #2: 2,3-diethynylquinoxaline In tetrahydrofuran; hexane at -78 - 20℃; for 11.5h; Inert atmosphere; Stage #3: With iodine In tetrahydrofuran; hexane at 20℃; for 5h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With sec.-butyllithium In tetrahydrofuran; cyclohexane at -78 - 20℃; | 97% |
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at -78℃; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -78 - 20℃; for 17h; Inert atmosphere; | 84% |
| Stage #1: 1-benzofurane With sodium 2,2,6,6-tetramethylpiperidide In hexane at 20℃; for 0.5h; Inert atmosphere; Sealed tube; Stage #2: chloro-trimethyl-silane In hexane at 0 - 20℃; for 1h; Inert atmosphere; Sealed tube; | 82% |
-
-
271-89-6
1-benzofurane

-
-
556-96-7
5-bromo-1,3-xylene

-
-
154715-81-8
2-(3,5-dimethylphenyl)-1,3-benzoxazole

| Conditions | Yield |
|---|---|
| With C13H18Cl2N4OPd; lithium tert-butoxide In N,N-dimethyl-formamide at 130℃; for 24h; Catalytic behavior; Inert atmosphere; Schlenk technique; | 97% |
-
-
271-89-6
1-benzofurane

| Conditions | Yield |
|---|---|
| With bismuth(lll) trifluoromethanesulfonate In dichloromethane at 20℃; for 0.166667h; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran at 0℃; for 1h; Stage #2: p-benzoquinone In tetrahydrofuran | 96% |
-
-
271-89-6
1-benzofurane

-
-
126799-87-9
o-bromobenzyl azide

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In dichloromethane at 20℃; for 0.25h; Aube-Schmidt rearrangement; | 96% |
-
-
271-89-6
1-benzofurane

-
-
941294-58-2
tert-butyl 6-bromopyridin-3-carboxylate

-
-
1393880-07-3
tert-butyl 6-(benzofuran-2-yl)nicotinate

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzofurane With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: With zinc(II) chloride In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #3: tert-butyl 6-bromopyridin-3-carboxylate With methanesulfonic acid(2-dicyclohexylphosphino-2′,4′,6′-triisopropyl-1,1′-biphenyl)[2-(2′-amino-1,1′-biphenyl)]palladium(II); XPhos In tetrahydrofuran; hexane at 20℃; for 12h; Negishi Coupling; Inert atmosphere; | 96% |
| Stage #1: 1-benzofurane With n-butyllithium; Triisopropyl borate In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: tert-butyl 6-bromopyridin-3-carboxylate With potassium phosphate; chloro-(2-dicyclohexylphosphino-2,4,6-triisopropyl-1,1-biphenyl)[2-(2-amino-1,1-biphenyl)]palladium(II) In tetrahydrofuran; hexane; water at 40℃; for 2h; Suzuki-Miyaura Coupling; Inert atmosphere; | 95% |
Benzofuran Specification
The 2,3-Benzofuran is an organic compound with the formula C8H6O. The IUPAC name of this chemical is 1-benzofuran. With the CAS registry number 271-89-6, it is also named as Coumarone. The product's category is Furan & Benzofuran. Besides, it is colorless to light yellow liquid, which is used in the manufacture of synthetic resin. It is the fragrance intermediates, which is used for flavoring soap or washing powder.
Physical properties about 2,3-Benzofuran are: (1)ACD/LogP: 2.67; (2)ACD/LogD (pH 5.5): 2.67; (3)ACD/LogD (pH 7.4): 2.67; (4)ACD/BCF (pH 5.5): 62.98; (5)ACD/BCF (pH 7.4): 62.98; (6)ACD/KOC (pH 5.5): 675.27; (7)ACD/KOC (pH 7.4): 675.27; (8)#H bond acceptors: 1; (9)Polar Surface Area: 13.14 Å2; (10)Index of Refraction: 1.6; (11)Molar Refractivity: 36.39 cm3; (12)Molar Volume: 106.3 cm3; (13)Polarizability: 14.42×10-24cm3; (14)Surface Tension: 38.6 dyne/cm; (15)Density: 1.11 g/cm3; (16)Flash Point: 56.1 °C; (17)Enthalpy of Vaporization: 39.35 kJ/mol; (18)Boiling Point: 174 °C at 760 mmHg; (19)Vapour Pressure: 1.65 mmHg at 25°C.
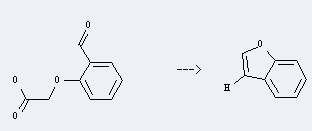
Preparation: this chemical can be prepared by (2-formyl-phenoxy)-acetic acid. This reaction will need reagent sodium acηte, acetic acid anhydride and acetic acid.
Uses of 2,3-Benzofuran: it can be used to produce 2,3-dihydro-benzofuran. It will need reagent alcoholic potash.

When you are using this chemical, please be cautious about it as the following:
It is flammable and harmful to aquatic organisms. Please keep away from sources of ignition - No smoking. Besides, this chemical has limited evidence of a carcinogenic effect. When you are using it, wear suitable protective clothing and gloves. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: o2c1ccccc1cc2
(2)InChI: InChI=1/C8H6O/c1-2-4-8-7(3-1)5-6-9-8/h1-6H
(3)InChIKey: IANQTJSKSUMEQM-UHFFFAOYAU
(4)Std. InChI: InChI=1S/C8H6O/c1-2-4-8-7(3-1)5-6-9-8/h1-6H
(5)Std. InChIKey: IANQTJSKSUMEQM-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 500mg/kg (500mg/kg) | European Journal of Medicinal Chemistry--Chimie Therapeutique. Vol. 12, Pg. 383, 1977. |
Related Products
- Benzofuran
- Benzofuran, 2,3-dihydro-5,7-dinitro-
- Benzofuran, 2,3-dimethyl-
- Benzofuran, 3-bromo-
- Benzofuran, 3-nitroso-
- Benzofuran, 4-chloro-
- Benzofuran, 5-chloro-
- Benzofuran, 5-nitro-
- Benzofuran, polymer with 1H-indene
- Benzofuran,2,3-dihydro-2,2-dimethyl-
- 2719-08-6
- 2719-15-5
- 2719-21-3
- 2719-23-5
- 2719-27-9
- 2719-30-4
- 2719-32-6
- 27193-28-8
- 27193-86-8
- 2719-40-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View