-
Name
Benzoylformic acid
- EINECS 210-278-7
- CAS No. 611-73-4
- Article Data360
- CAS DataBase
- Density 1.298 g/cm3
- Solubility methanol: 0.1 g/mL, clear
- Melting Point 60-69 °C
- Formula C8H6O3
- Boiling Point 258.6 °C at 760 mmHg
- Molecular Weight 150.134
- Flash Point 124.4 °C
- Transport Information
- Appearance white to light yellow adhering crystals
- Safety 26-28-36-28A-24-22
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Glyoxylicacid, phenyl- (6CI,7CI,8CI);2-Oxo-2-phenylacetic acid;2-Oxo-2-phenylethanoicacid;Benzoylformic acid;CAA 0869;Formic acid, benzoyl-;LFC 1849;NSC 28293;Oxophenylacetic acid;Phenylgloxylic acid;Phenylglyoxylic acid;Phenyloxoacetic acid;a-Ketophenylacetic acid;a-Oxobenzeneacetic acid;
- PSA 54.37000
- LogP 0.95390
Synthetic route
-
-
15206-55-0
methyl 2-oxo-2-phenylacetate

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide | 100% |
| With water; sodium hydroxide In dichloromethane | 100% |
| With water; sodium hydroxide at 20℃; for 2h; | 99.2% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; | 100% |
| With trifluoroacetic acid at 0℃; for 1h; | 57% |
| With trifluoroacetic acid at 0℃; for 5h; |

| Conditions | Yield |
|---|---|
| With methyl 3,5-bis((1H-1,2,4-triazol-1-yl)methyl)benzoate; oxygen; sodium acetate; nickel dibromide at 120℃; under 760.051 - 912.061 Torr; for 48h; chemoselective reaction; | A 91% B 99% |
| Multi-step reaction with 2 steps 1: sodium hydrogencarbonate; ruthenium trichloride; potassium peroxymonosulfate / acetonitrile; water; ethyl acetate / 0.3 h 2: iron(II) perchlorate hexahydrate; tetramethylammonium hydroxide pentahydrate / acetonitrile; water / 12 h / Inert atmosphere View Scheme |
-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With sodium bromate In acetonitrile for 52h; Heating; | 98% |
| With oxalyl dichloride; methyl dodecyl sulfoxide; triethylamine In dichloromethane at -60 - 20℃; Swern oxidation; | 96% |
| With oxalyl dichloride; methyl dodecyl sulfoxide; triethylamine In dichloromethane at -60 - 20℃; Swern oxidation; | 96% |
-
-
1352131-39-5
5-hydroxy-N,N',5-triphenylimidazolidine-2,4-dione

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In methanol at 20 - 50℃; for 1h; | 98% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 100 - 120℃; for 5h; | 96.6% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iron(III) chloride; thymidyl acetic acid In decane; acetonitrile at 80℃; for 6h; | 96% |
| With Oxone; 2-Iodobenzoic acid In water; acetonitrile at 70℃; for 6h; | 94% |
| With sodium tetrahydroborate; 1% Pd/C; water; potassium hydroxide In methanol at 20℃; for 15h; In air; | 91% |
| With potassium permanganate; acetic acid; Tris(3,6-dioxaheptyl)amine In dichloromethane for 4h; Ambient temperature; | 71% |
| aqua(2,2'-bipyridine)(2,2':6',2''-terpyridine)ruthenium(II)(2+) In phosphate buffer for 6h; pH=7.2; Electrolysis; | 47% |

| Conditions | Yield |
|---|---|
| With tetramethoxytitanium; β-Propiolactone at 60℃; for 2h; Temperature; | 96% |
| Multi-step reaction with 5 steps 1.1: hydrogen bromide; dihydrogen peroxide / water / 4.5 h / 25 - 90 °C 2.1: toluene-4-sulfonic acid; benzene / water / 36 h / 85 °C 3.1: tetrabutylammomium bromide; sodium ethanolate / water / 23 h / 105 °C 4.1: oxalic acid / dichloromethane; water / 5 h / 25 °C 5.1: tetrabutylammomium bromide; hydrogen bromide / water / 0.02 h 5.2: 5 h / 25 - 90 °C View Scheme |

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 2h; Heating; | 94% |

| Conditions | Yield |
|---|---|
| With phosphotungstic acid; ferric nitrate at 55 - 60℃; for 0.166667h; | 93% |
| With D-lactate oxidase from Gluconobacter oxydans 621H; oxygen; catalase In aq. buffer at 30℃; pH=7.4; Reagent/catalyst; Enzymatic reaction; | |
| With NAD at 30℃; for 48h; pH=9.5; Kinetics; Reagent/catalyst; Temperature; pH-value; Enzymatic reaction; |

-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hypobromide; sodium thiosulfate In dichloromethane; water | 91% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; dihydrogen peroxide In water at 25 - 65℃; for 5h; Reagent/catalyst; Time; | 90.1% |
| With hydrogen bromide; dihydrogen peroxide In water Temperature; Reflux; | 79.3% |
| Stage #1: 1-phenyl-2-hydroxyethanone With tetrabutylammomium bromide; hydrogen bromide In water for 0.0166667h; Stage #2: With dihydrogen peroxide In water at 25 - 90℃; for 5h; | 83 %Chromat. |
| With hydrogen bromide; dihydrogen peroxide In water at 90℃; for 7h; |
-
-
80490-41-1
3,4-diphenyl-4-hydroxy-2-isoxazolin-5-one

-
A
-
100-47-0
benzonitrile

-
B
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 3h; Heating; | A n/a B 89.3% |
| With hydrogenchloride; sodium hydroxide; water Mechanism; |
-
-
134918-42-6
epoxide of α,β-difluoro-β-chlorostyrene

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate for 0.5h; | 89% |

| Conditions | Yield |
|---|---|
| With pyridine; selenium(IV) oxide at 120℃; for 18h; Inert atmosphere; | 87% |
| With pyridine; selenium(IV) oxide for 3h; Reflux; | 87% |
| With pyridine; selenium(IV) oxide at 90℃; for 5h; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With potassiuim nitrosodisulfonate In water for 60h; pH 10.0; | 82% |
| With ω-transaminase |
-
-
130749-19-8
2,2,2-Tris-benzotriazol-1-yl-1-phenyl-ethanone

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid In tetrahydrofuran at 50℃; for 48h; | 81% |
-
-
79344-05-1
3,5-diphenyl-4,4-dichloro-5-hydroxy-Δ2-isoxazoline

-
A
-
100-47-0
benzonitrile

-
B
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water for 2.5h; Heating; | A 75.4% B 80% |

| Conditions | Yield |
|---|---|
| Stage #1: phenylglyoxal hydrate With silver perchlorate; sodium hydroxide In water at 20℃; Inert atmosphere; Stage #2: With hydrogenchloride In water Inert atmosphere; | 78% |
| bei der Einw. von gaerender Hefe; | |
| Multi-step reaction with 2 steps 1: 26.5 percent Spectr. / Et3N, pyridine, O2, 3-Angstroem sieves / Cu(NO3)2py2 / methanol / 12 h / 25 °C / 1748 Torr 2: 68.6 percent Spectr. / Et3N, pyridine, O2, H2O / Cu(NO3)2py2 / methanol / 12 h / 25 °C / 1748 Torr View Scheme |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; magnesium In N,N-dimethyl-formamide at 20℃; Temperature; Solvent; | 75% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water; sodium nitrite at 80℃; for 4h; | 73.5% |
| With acetic anhydride bei der Destillation u. Verseifen mit Salzsaeure; |

| Conditions | Yield |
|---|---|
| Stage #1: carbon dioxide; benzaldehyde With potassium cyanide In N,N-dimethyl-formamide at 12℃; for 1h; Schlenk technique; Stage #2: With titanium(IV)isopropoxide; 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N-dimethyl-formamide at 40℃; for 18h; Schlenk technique; | 72% |
-
-
2461-80-5
diphenacyl sulfide

-
A
-
1522397-30-3
C16H10O4S

-
B
-
595565-17-6
(3-phenylthiophene-2,5-diyl)bis(phenylmethanone)

-
C
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide In 1,4-dioxane for 2h; Reflux; | A n/a B 10% C 71% |

-
-
80490-51-3
4-hydroxy-3-methyl-4-phenylisoxazoline-5-one

-
A
-
611-73-4
Benzoylformic acid

-
B
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| With 2,6-dichloro-benzonitrile In benzene for 0.5h; Heating; | A 70% B n/a |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; magnesium In N,N-dimethyl-formamide at 20℃; | 69% |
-
-
614-21-1
2-nitroacetophenone

-
A
-
611-73-4
Benzoylformic acid

-
B
-
65-85-0
benzoic acid

-
C
-
6635-54-7
3,4-dibenzoyl-1,2,5-oxadiazole-N-oxide

| Conditions | Yield |
|---|---|
| With nitric acid; sodium nitrite In nitromethane; water at 60℃; for 0.5h; | A 9.7% B 4.4% C 68.4% |
| With nitric acid; sodium nitrite In nitromethane; water at 60℃; for 0.5h; Product distribution; Mechanism; variation of solvents, reactants; | A 9.7% B 4.4% C 68.4% |

-
-
591-50-4
iodobenzene

-
-
201230-82-2
carbon monoxide

-
A
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
B
-
611-73-4
Benzoylformic acid

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With methyl iodide; sodium tetrahydroborate In 1,4-dioxane; water at 20℃; for 20h; | A 3% B 68% C 5% |
| With methyl iodide; sodium tetrahydroborate In 1,4-dioxane; water at 20℃; for 20h; | A 3% B 64% C 13% |
| With calcium hydroxide; water; dichlorobis(dimethylphenylphosphine)palladium(II) In isopropyl alcohol at 100℃; for 12h; P(CO) = 150 atm at room temperature; further catalysts, solvents and hydroxide; | A 69.0 % Chromat. B 3.8 % Chromat. C 9.3 % Chromat. |
| With calcium hydroxide; water; bis(benzonitrile)palladium(II) dichloride In isopropyl alcohol at 100℃; for 12h; P(CO) = 150 atm at room temperature; further catalysts, solvents and hydroxide; | A 46.0 % Chromat. B 8.7 % Chromat. C 25.5 % Chromat. |
| With sodium sulfide; CoCl2 In 1,4-dioxane; water at 20℃; for 20h; Product distribution; other reag.; other solvent; | A 3 % Chromat. B 62 % Chromat. C 5 % Chromat. |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; methyltrioxorhenium(VII) In dichloromethane for 48h; | A 21% B 68% |
| With dihydrogen peroxide; methyltrioxorhenium(VII) In acetone for 48h; | A 65% B 11% |

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate; oxygen In acetonitrile at 60℃; | A 26% B 65% |
-
-
611-71-2, 611-72-3, 17199-29-0, 90-64-2
MANDELIC ACID

-
A
-
611-73-4
Benzoylformic acid

-
B
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With oxone; 2-iodo-3,4,5,6-tetramethylbenzoic acid In water; acetonitrile at 20℃; for 3h; | A 63% B 30% |
-
-
611-73-4
Benzoylformic acid

-
-
25726-04-9
phenylglyoxylyl chloride

| Conditions | Yield |
|---|---|
| With Dichloromethyl methyl ether at 50℃; for 0.5h; | 100% |
| With Dichloromethyl methyl ether In dichloromethane at 15 - 20℃; for 1h; Inert atmosphere; | 100% |
| With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane | 95% |

| Conditions | Yield |
|---|---|
| With sulfur In benzene at 80℃; for 0.166667h; | 100% |
| With sulfur In benzene for 0.166667h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With sulfur In benzene at 80℃; for 0.75h; | 100% |
| With sulfur In benzene at 80℃; for 1h; |

| Conditions | Yield |
|---|---|
| With sulfur In benzene at 80℃; for 0.75h; | 100% |

| Conditions | Yield |
|---|---|
| With NH4S2O8; sulfuric acid; silver nitrate In dichloromethane; water at 40℃; for 2.5h; | 100% |
| Stage #1: 1,4-pyrazine; Benzoylformic acid With ammonium peroxydisulfate; sulfuric acid; silver nitrate In dichloromethane; water at 50℃; for 2h; Stage #2: With water; sodium hydrogencarbonate In dichloromethane | 91% |
| With ferrous(II) sulfate heptahydrate; ammonium peroxydisulfate; formic acid; dimethyl sulfoxide In dichloromethane; water at 40℃; Minisci Aromatic Substitution; | 74% |
| With ammonium peroxydisulfate; silver nitrate; trifluoroacetic acid In dichloromethane; water at 60℃; for 3h; | 68% |
| With dipotassium peroxodisulfate; silver orthophosphate In dichloromethane; water at 40℃; for 24h; | 26% |
-
-
1122-54-9
methyl (4-pyridyl) ketone

-
-
611-73-4
Benzoylformic acid

-
-
132883-96-6
1-(2-benzoylpyridin-4-yl)-ethanone

| Conditions | Yield |
|---|---|
| With NH4S2O8; sulfuric acid; silver nitrate In dichloromethane; water at 40℃; for 2.5h; | 100% |
| 33% |

| Conditions | Yield |
|---|---|
| With NH4S2O8; sulfuric acid; silver nitrate In dichloromethane; water at 40℃; for 2.5h; | 100% |
-
-
91-19-0
2,3-diethynylquinoxaline

-
-
611-73-4
Benzoylformic acid

-
-
13481-33-9
phenyl(quinoxalin-2-yl)methanone

| Conditions | Yield |
|---|---|
| With NH4S2O8; sulfuric acid; silver nitrate In dichloromethane; water at 40℃; for 2.5h; | 100% |
| With dipotassium peroxodisulfate In water; acetonitrile at 20 - 37℃; for 15h; Irradiation; Green chemistry; regioselective reaction; | 80% |
| With dipotassium peroxodisulfate; silver orthophosphate In dichloromethane; water at 40℃; for 24h; | 58% |

| Conditions | Yield |
|---|---|
| 100% |
-
-
91-21-4
1,2,3,4-tetrahydroisoquinoline

-
-
611-73-4
Benzoylformic acid

-
-
122709-96-0
(3,4-dihydroisoquinoline-2(1H)-yl)(phenyl)methanethione

| Conditions | Yield |
|---|---|
| With sulfur In benzene at 80℃; for 0.25h; | 100% |
| With sulfur In benzene at 80℃; for 0.25h; |

| Conditions | Yield |
|---|---|
| In dichloromethane at 40℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: oxalyl dichloride; Benzoylformic acid With copper diacetate; N,N-dimethyl-formamide In dichloromethane at 20℃; for 2h; Stage #2: diallylamine With triethylamine In dichloromethane | 100% |

-
-
64868-20-8
(1S,2R)-(1-methyl-2-phenyl-2-hydroxy)ethyltrimethylammonium iodide

-
-
611-73-4
Benzoylformic acid

-
-
1639366-24-7
[(1R,2S)-N,N-dimethylephedrinium][phenylglyoxylate]

| Conditions | Yield |
|---|---|
| With Dowex Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In chloroform for 0.5h; Inert atmosphere; Reflux; | 100% |
| In chloroform for 0.5h; Inert atmosphere; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| In water; acetone | 100% |

-
-
611-73-4
Benzoylformic acid

| Conditions | Yield |
|---|---|
| In isopropyl alcohol for 1h; Heating; | 99.2% |

| Conditions | Yield |
|---|---|
| With chlorobenzene In methanol at 40℃; for 5h; Sealed tube; Green chemistry; chemoselective reaction; | 99% |
| With phosphonic Acid; methanesulfonic acid; sodium iodide In water at 95℃; for 48h; Inert atmosphere; | 94.3% |
| With hydrazine hydrate anschliessend Erhitzen mit KOH; | |
| With phosphorus; hydrogen iodide at 160℃; |
-
-
65253-04-5
(-)-8-phenylmenthol

-
-
611-73-4
Benzoylformic acid

-
-
88002-15-7
Phenylglyoxylsaeure-<(1R,2S,5R)-5-methyl-2-(1-methyl-1-phenylethyl)-1-cyclohexyl>ester

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene Heating; | 99% |
| With toluene-4-sulfonic acid In toluene at 130℃; for 2h; Dean-Stark; | 25% |
| With hydrogen cation |
-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
611-73-4
Benzoylformic acid

-
-
141694-25-9
N-methoxy-N-methyl-α-oxo-benzeneacetamide

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride In acetonitrile at 20℃; for 1.5h; | 99% |
| Stage #1: Benzoylformic acid With N-Bromosuccinimide; triphenylphosphine In dichloromethane at 0℃; for 0.25h; Stage #2: N,O-dimethylhydroxylamine*hydrochloride With triethylamine In dichloromethane at 20℃; for 1h; chemoselective reaction; | 75% |
| With 2-chloro-1-methyl-pyridinium iodide; triethylamine In dichloromethane for 0.75h; Heating; | 53% |
-
-
611-73-4
Benzoylformic acid

-
-
415919-99-2
(1R,2R)-1,2-diphenyl-2-(4-trifluoromethylbenzyloxy)ethanol

-
-
415920-05-7
(1R,2R)-1,2-diphenyl-2-(4-trifluoromethylbenzyloxy)ethyl phenylglyoxylate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 25℃; | 99% |
-
-
866457-05-8
(1R,2S)-2-(1,1-dimethylethoxy)-1,2-diphenylethanol

-
-
611-73-4
Benzoylformic acid

-
-
866457-10-5
oxophenylacetic acid (1R,2S)-2-(1,1-dimethylethoxy)-1,2-diphenylethyl ester

| Conditions | Yield |
|---|---|
| With dmap; diisopropyl-carbodiimide In dichloromethane at 20℃; | 99% |
| With dmap; diisopropyl-carbodiimide In dichloromethane at 20℃; |
-
-
916525-75-2
(1R,2S)-1-(2-methoxyethoxy)-1,2-bis-(2-methoxyphenyl)-ethanol

-
-
611-73-4
Benzoylformic acid

-
-
916525-80-9
oxophenylacetic acid (1R,2S)-2-(2-methoxyethoxy)-1,2-bis(2-methoxyphenyl)ethyl ester

| Conditions | Yield |
|---|---|
| With dmap; diisopropyl-carbodiimide In dichloromethane at 20℃; for 12h; | 99% |
| With dmap; diisopropyl-carbodiimide In dichloromethane at 20℃; for 12h; |
-
-
15022-08-9
diallylcarbonate

-
-
611-73-4
Benzoylformic acid

-
-
35660-91-4, 35845-66-0, 495-41-0
(E)-1-phenyl-2-buten-1-one

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); Tri(p-tolyl)phosphine In 1,4-dioxane at 100℃; for 12h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate In water; acetonitrile at 70℃; for 24h; Inert atmosphere; Schlenk technique; Sealed tube; | 99% |
| With ammonium peroxydisulfate; copper(II) oxide In water; dimethyl sulfoxide at 80℃; for 12h; | 73% |
-
-
106-45-6
para-thiocresol

-
-
611-73-4
Benzoylformic acid

-
A
-
103-19-5
di(p-tolyl) disulfide

-
B
-
10371-42-3
S-(4-methylphenyl) thiobenzoate

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate In water; acetonitrile at 70℃; for 24h; Inert atmosphere; Schlenk technique; Sealed tube; | A 5.5% B 99% |


| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate In water; acetonitrile at 70℃; for 24h; Reagent/catalyst; Temperature; Solvent; Inert atmosphere; Schlenk technique; Sealed tube; | 99% |
-
-
100-63-0
phenylhydrazine

-
-
611-73-4
Benzoylformic acid

-
-
74470-05-6
phenyl-((Z)-phenylhydrazono)-acetic acid

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 3h; Reflux; | 99% |
Benzoylformic acid Chemical Properties
The density of Benzoylformic acid(611-73-4) is 1.38 and it has a melting point of 62-65 °C(lit.). The boiling point is 84 °C0.1 mm Hg. Its flash point is 147-151°C/12mm.
The chemical synonyms of Benzoylformic acid(611-73-4) are PHENYLGLYOXYLIC ACID;OXO(PHENYL)ACETIC ACID;RARECHEM AL BO 0070;2-Oxo-2-phenylacetic acid;alpha-Ketophenylacetic acid;alpha-ketophenylaceticacid;alpha-oxo-benzeneaceticaci;alpha-oxobenzeneaceticacid
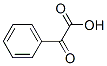
The molecular structure of Benzoylformic acid(611-73-4):
Benzoylformic acid Toxicity Data With Reference
| 1. | ivn-mus LD50:180 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#04110 . |
Benzoylformic acid Safety Profile
Hazard Codes Xi
Risk Statements 36/37/38
Safety Statements 26-28-36-28A-24-22
WGK Germany 3
RTECS DA1480000
HS Code 29183000
Benzoylformic acid Specification
Conditions to Avoid: Incompatible materials, strong oxidants.
Incompatibilities with Other Materials Strong oxidizing agents, strong reducing agents, strong acids, strong bases.
Hazardous Decomposition Products Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization Has not been reported.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View