Synthetic route
-
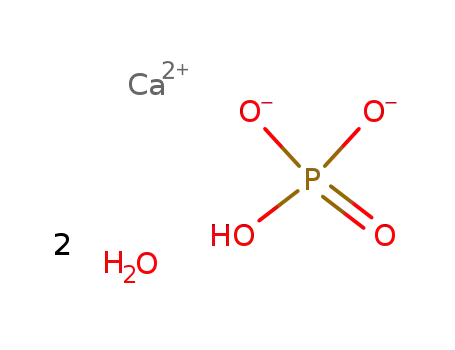
-
calcium hydrogen phosphate dihydrate
Conditions
| Conditions | Yield |
|---|
| In melt byproducts: Ca3(PO4)2*99H2O; melting with decompn. at 36°C;; | 99.85% |
| With (NH4)2CO3 or NH4H2PO4 In water byproducts: CaCO3, hydroxylapatite, P2O5; decompn. with aq. (NH4)2CO3 or aq. NH4H2PO4 soln. at various conditions;; P2O5 in soln.;; | |
| With phosphoric acid In not given byproducts: H2O; dissolving 95 g of dihydrate in 500 g 35 % H3PO4 at 25°C; heating at 100-110°C; filtration of hot soln.;; 6-8 fold washing with abs. alcohol, drying at 100°C; contains crystal water;; | |
-
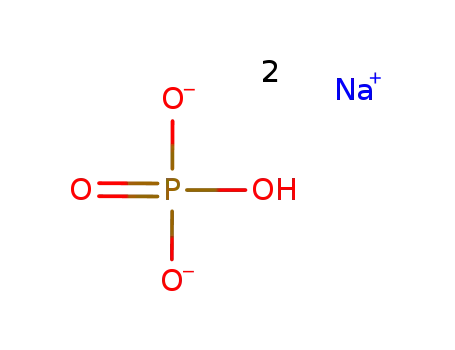
-
disodium hydrogenphosphate
-
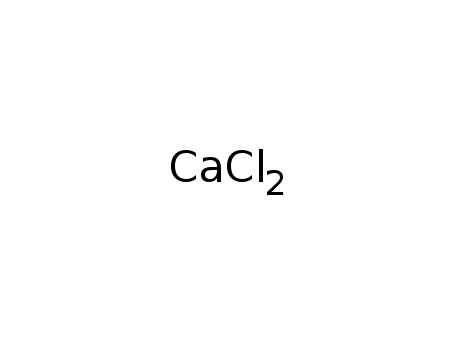
-
calcium chloride
Conditions
| Conditions | Yield |
|---|
| With potassium dihydrogenphosphate In water aq. CaCl2 soln;; 3 times washing with 330 ml aq. H3PO4, washing with abs. ethanol; pure; contains 0.04 mol H2O after calcination at 900.degreee.C;; | 95% |
| In not given addn. of CaCl2 to neutral Na2HPO4 soln.;; | |
| With hydrogenchloride; ammonia In water dissolving ppt. from aq. CaCl2 soln. and Na2HPO4 in HCl, dropwise addn. of 0.6 % aq. NH3 on water bath, replacement of vaporized H2O;; crystn. after 2-3 weeks;; | |
| With HCl; NH3 In water dissolving ppt. from aq. CaCl2 soln. and Na2HPO4 in HCl, dropwise addn. of 0.6 % aq. NH3 on water bath, replacement of vaporized H2O;; crystn. after 2-3 weeks;; | |
| In not given | |
-
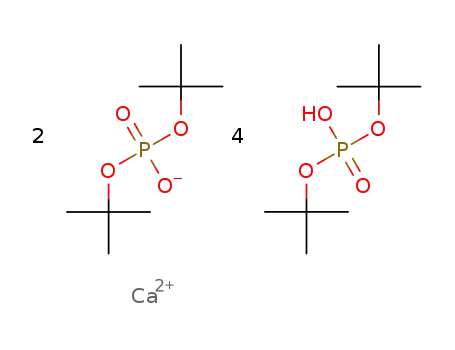
-
2C8H18O4P(1-)*4C8H19O4P*Ca(2+)
Conditions
| Conditions | Yield |
|---|
| With calcium methylate In methanol at 150℃; for 48h; Autoclave; | 45% |
-
-
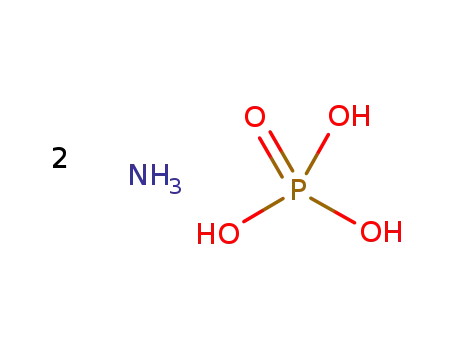
diammonium phosphate
Conditions
| Conditions | Yield |
|---|
| In water for 0.0333333h; pH=5 - 9; | |
-
-
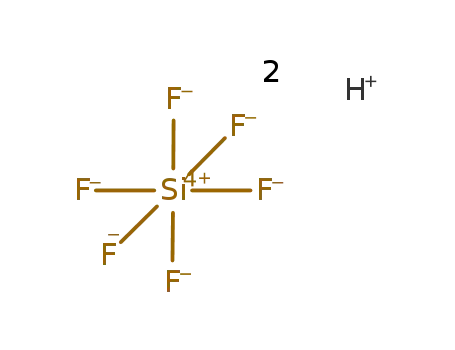
fluorosilicic acid
-

-
calcium oxide
-
B
-
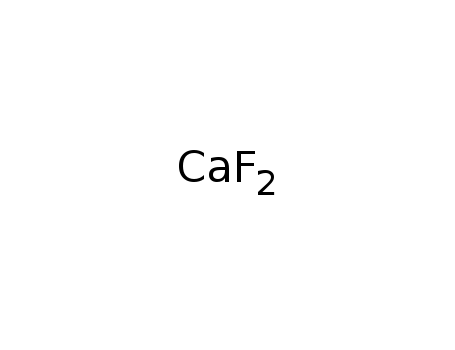
calcium fluoride
-
C
-
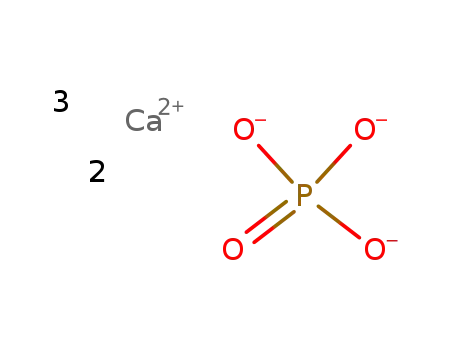
tricalcium diphosphate
-
D
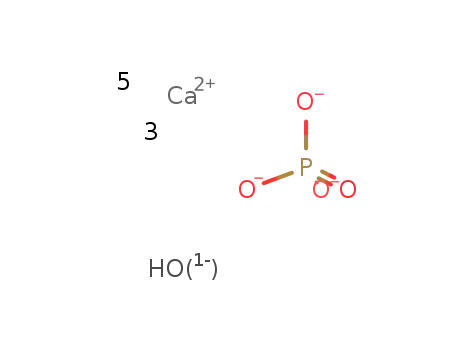
-
hydroxyapatite
Conditions
| Conditions | Yield |
|---|
| In water Kinetics; byproducts: H2O; gastight neutralization reactor, stirring in turbulent regime, 23 °C, addn. of CaO (varying stochiometry) to the acid mixture; withdrawn samples filtered, anal.; | |
-

-
tricalcium diphosphate
Conditions
| Conditions | Yield |
|---|
| reaction of NO2 with moist Ca phosphate;; | |
| reaction of NO2 with moist Ca phosphate;; | |
-
B
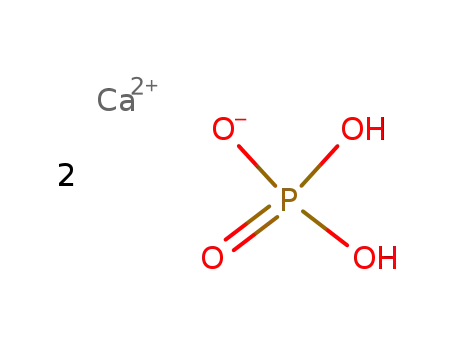
-
calcium dihydrogen phosphate
Conditions
| Conditions | Yield |
|---|
| With phosphate minerals Decompn. of phosphate minerals by HNO3;; | |
-
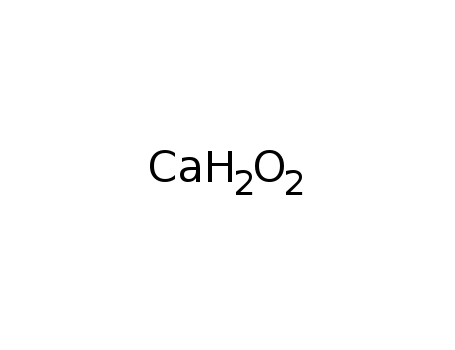
-
calcium hydroxide
-
B
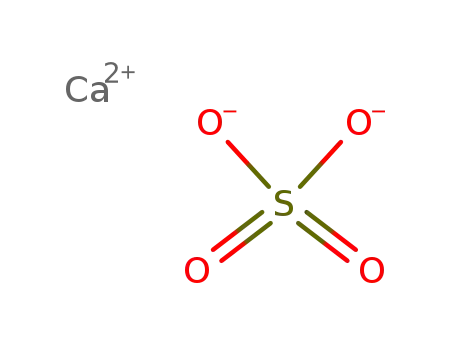
-
calcium sulfate
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) calcination of ppt.;; | |
-
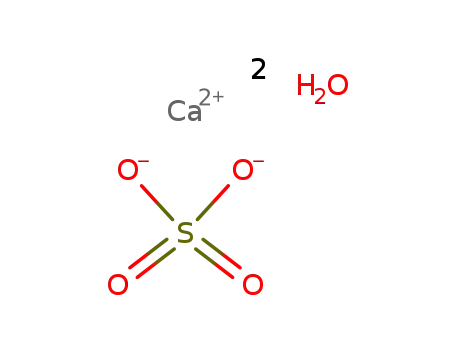
-
calcium sulfate dihydrate
-
B
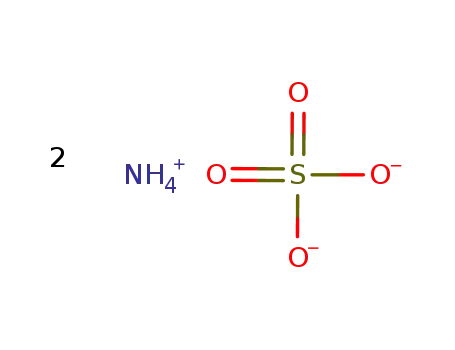
-
ammonium sulfate
Conditions
| Conditions | Yield |
|---|
| With NH3 In neat (no solvent) NH4H2PO4 from reaction of superphosphate with ammonia; addn. of further amounts of ammonia;; | |
| In neat (no solvent) | |
-
B
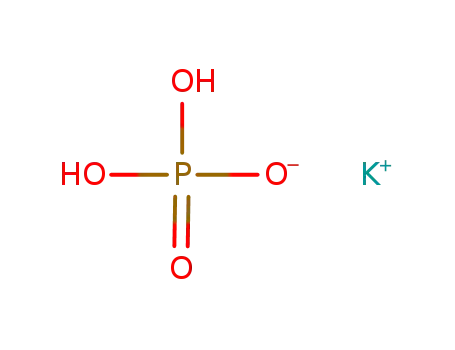
-
potassium dihydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| With CaCO3 In water | |
| With calcium carbonate In water | |
-
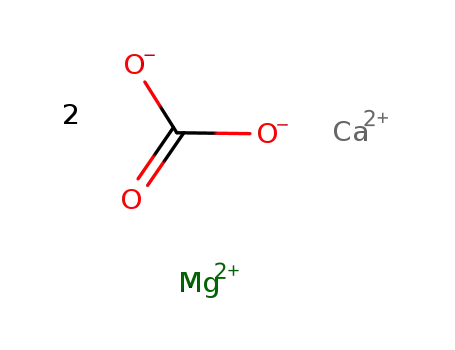
-
dolomite
-
B
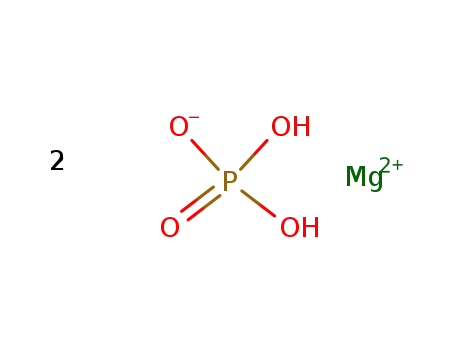
-
magnesium dihydrogen phosphate
-
C

-
magnesium phosphate dibasic trihydrate
Conditions
| Conditions | Yield |
|---|
| Kinetics; byproducts: CO2, H2O; decomposn. by H3PO4 (10, 15, 20, 30% P2O5) at 50 and 80°C; elem. anal., potentiometry, conductometry; | |
-
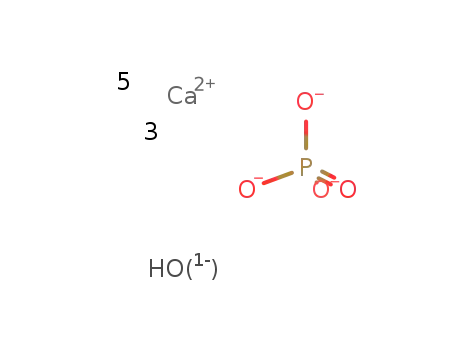
-
hydroxyapatite
Conditions
| Conditions | Yield |
|---|
| With ammonia In hydrogenchloride slow pptn. from a soln. of bone ash in HCl by aq. NH3 at >68°C;; | |
| shaking of bone ash with HCl;; | |
-
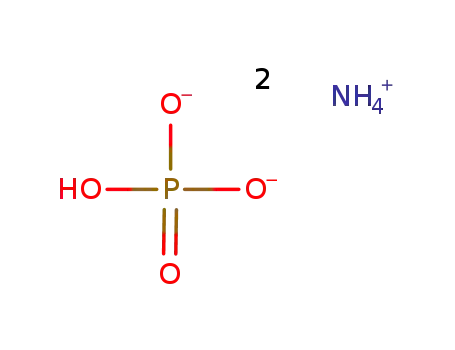
-
diammonium hydrogenphosphate
-
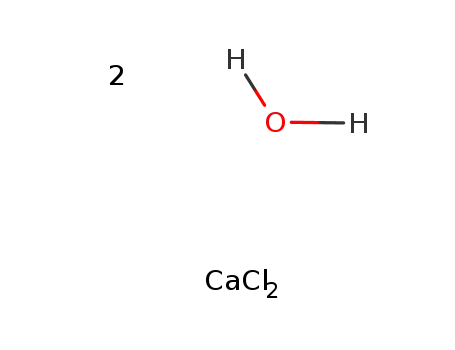
-
calcium(II) chloride dihydrate
Conditions
| Conditions | Yield |
|---|
| In water soln. of (NH4)2HPO4 heated at 373+/-10 K under reflux and stirring; to this hot soln. aq. soln. of CaCl2 added slowly; suspn. aged for 24 h at 373 K; solid filtered; washed (H2O); dried at 573+/-10 K for 24 h; elem. anal.; | |
-
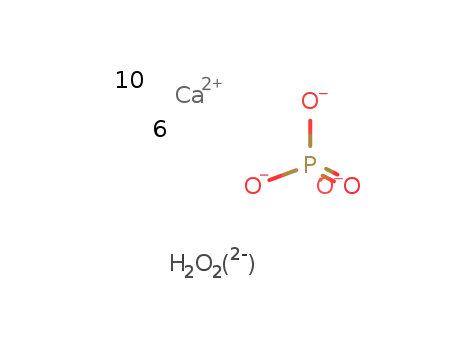
-
hydroxyapatite
Conditions
| Conditions | Yield |
|---|
| In further solvent(s) by treating hydroxyapatite with H3PO4; heat treatment at 25-200°C; XRD- and IR data; | |
-

-
tricalcium diphosphate
-
B

-
calcium dihydrogen phosphate
Conditions
| Conditions | Yield |
|---|
| With carnallite In water | |
| With magnesium chloride In water byproducts: CaCl2; | |
| With potassium salts In water | |
-

-
tricalcium diphosphate
-
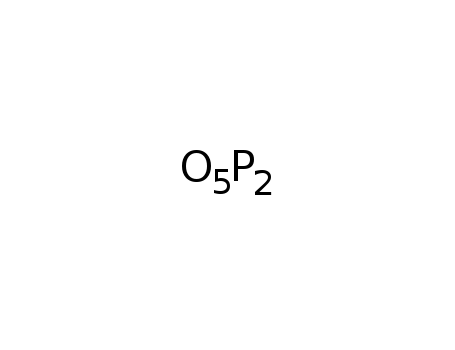
-
phosphorus pentoxide
-

-
calcium hydroxide
-

-
tricalcium diphosphate
-

-
calcium hydroxide
-
B

-
calcium dihydrogen phosphate
Conditions
| Conditions | Yield |
|---|
| In water under pressure;; | |
| In water under pressure;; | |
-
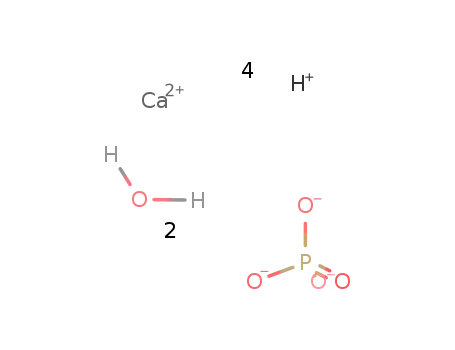
-
calcium tetrahydrogen phosphate
-
B
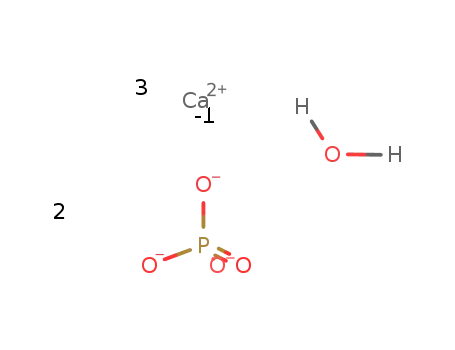
-
tricalcium phosphate hydrate
Conditions
| Conditions | Yield |
|---|
| In water 4 h treatment of 1 g CaHPO4 with 10 g H2O at 280-300°C;; aggregate of fine needles;; | |
-

-
calcium tetrahydrogen phosphate
-

-
calcium hydroxide
Conditions
| Conditions | Yield |
|---|
| In water neutralisation of 60 - 85% H3PO4 with CaCO3;; | |
-

-
calcium hydrogen phosphate dihydrate
-
-
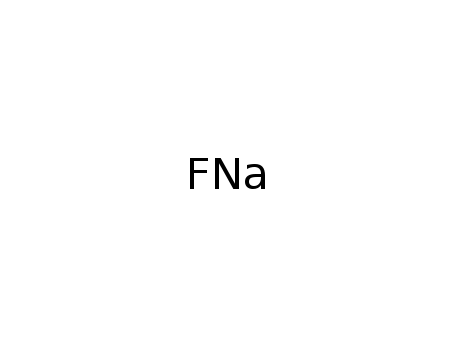
sodium fluoride
-
B

-
calcium fluoride
-
-
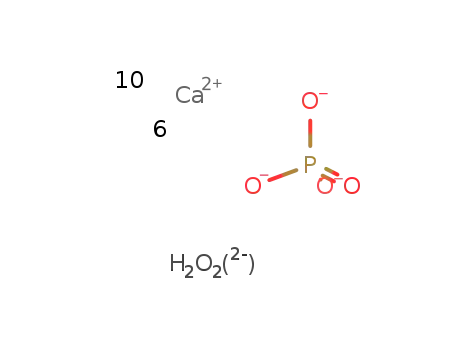
hydroxylapatite
Conditions
| Conditions | Yield |
|---|
| In water byproducts: H2O, PO4(3-); dicalcium phosphate is added to stirred soln. of NaF (pH 7) at 25°C for 1 h; filtn.; XRD; | |
-

-
tricalcium diphosphate
-

-
calcium oxide
Conditions
| Conditions | Yield |
|---|
| With sulfuric acid In water | |
| In water | |
-

-
tricalcium diphosphate
-

-
calcium oxide
-

-
tricalcium diphosphate
-

-
calcium oxide
-
B

-
calcium chloride
Conditions
| Conditions | Yield |
|---|
| With iron(II) chloride In water byproducts: FeCl3; pptn.;; | |
-

-
calcium oxide
Conditions
| Conditions | Yield |
|---|
| In water reaction of alkali phosphates with CaO;; | |
| In water | |
-

-
tricalcium diphosphate
-

-
tricalcium diphosphate
-
B

-
calcium sulfate
Conditions
| Conditions | Yield |
|---|
| With air In water pptn.;; | |
| In water | |
-
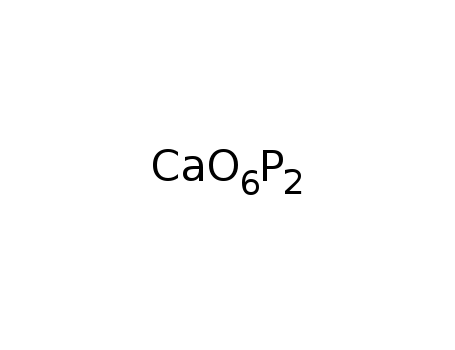
-
calcium metaphosphate
-
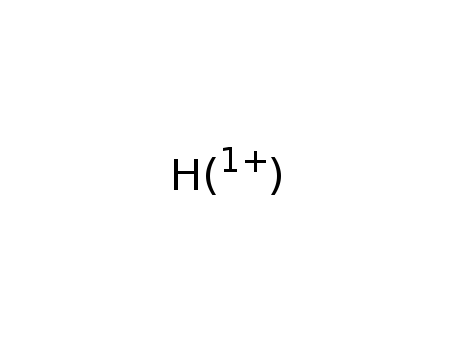
-
hydrogen cation
Conditions
| Conditions | Yield |
|---|
| In not given in acidic soln.;; | |
-

-
tricalcium diphosphate
-
B

-
calcium dihydrogen phosphate
Conditions
| Conditions | Yield |
|---|
| With nitric acid; Nitrate In water use of alkali nitrate;; | |
| With carbon dioxide In water CO2 from industrial process;; | |
| With hydrogenchloride; calcium chloride In water attack by dild. HCl in presence of CaCl2;; | |
-
-
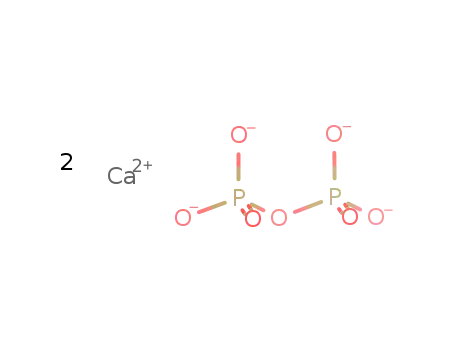
calcium pyrophosphate
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent, solid phase) CaHPO4 (14.5 mmol) was heated at 350°C under vac. (5E-3 Torr) for4 d; XRD studies; | 99.5% |
| In neat (no solvent, solid phase) CaHPO4 was pressed at 8 tons for 60 s and heated under vac. (5E-3 Torr) to 400°C for 6 d; XRD studies; | |
-

-
calcium pyrophosphate
-
-
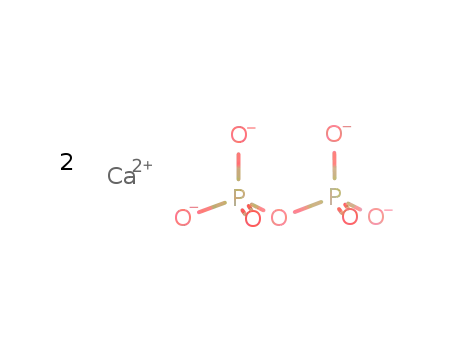
calcium pyrophosphate
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent, solid phase) CaHPO4 (14.7 mmol) was pressed at 8 tons for 60 s and heated at 600°C for 24 h; XRD studies; | A n/a
B 98.5% |
-
-
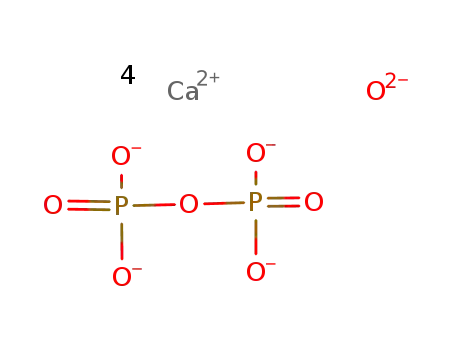
TTCP
-
Reaxys ID: 11388391
-
Reaxys ID: 11388391
-

-
calcium hydroxide
-
-
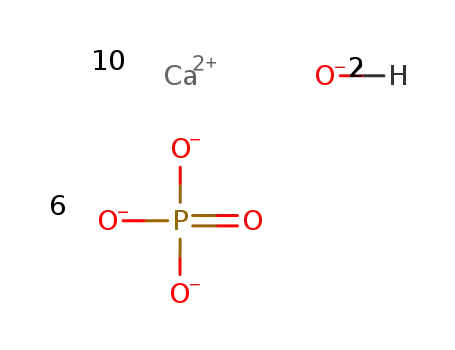
calcium hydroxyapatite
Conditions
| Conditions | Yield |
|---|
| With acetic acid In water at 95℃; for 16 - 24h; pH=7; Product distribution / selectivity; | |
| With propionic acid In water at 95℃; for 24h; Product distribution / selectivity; | |
-

-
sodium fluoride
-

-
disodium hydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| In water byproducts: CaF; pptn. of CaF2 from aq. suspension;; in soln.;; | |
-

-
calcium oxide
Conditions
| Conditions | Yield |
|---|
| With sulfuric acid In water byproducts: CaSO4, Ca3(PO4)2; react. of CaHPO4 with a satd. soln. of NaHSO4 (from technical alkali disulfate) in H2SO4; leaching mass; react. of NaH2PO4 soln. with CaO by pptn. of CaHPO4, reaction of filtrate containing Na2HPO4 with CaO in water;; | |
-

-
disodium hydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| In water heating a CaHPO4/CaSO4-mixture with NaCl and NaHCO3 in aq. suspension, filtration;; soln. contains Na2HPO4, purity 99.6%;; | |
| In water heating a CaHPO4/CaSO4-mixture with NaCl and NaHCO3 in aq. suspension, filtration;; soln. contains Na2HPO4, purity 99.6%;; | |
-

-
disodium hydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| With CO2 or citric acid In water ion-exchange at zeolithe using a soln. of CaHPO4 in CO2-containing H2O or dild. citric acid;; in soln.;; | |
Conditions
| Conditions | Yield |
|---|
| In not given reaction of mono calcium phosphate with >70.8% HNO3-solution;; | |
-
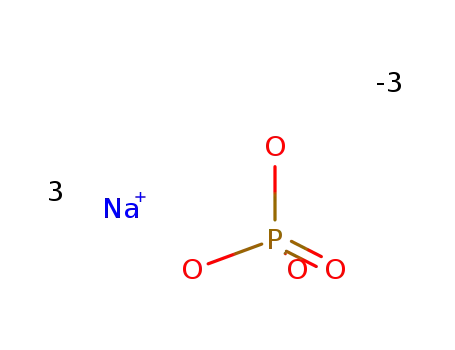
-
sodium phosphate
Conditions
| Conditions | Yield |
|---|
| In water byproducts: Ca3(PO4)2; reaction of CaHPO4 with NaOH, regeneration of CaHPO4 by treatment of Ca3(PO4)2 with Ca(H2PO4)2;; in soln.;; | |
-

-
calcium sulfate
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) | |
| In neat (no solvent) | |
-

-
disodium hydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| In water byproducts: CaCO3; pptn. of CaCO3 with a soln. containing 200g Na2CO3/l;; in soln.;; | |
Conditions
| Conditions | Yield |
|---|
| CaHPO4 was treated with equivalent amount of NH4HSO4;; | |
| CaHPO4 was treated with equivalent amount of NH4HSO4;; | |
-

-
calcium hydroxide
Conditions
| Conditions | Yield |
|---|
| In not given reaction of Na2SO4 soln. with CaHPO4; heating formed Na2HPO4 soln. with Ca(OH)2 under formation of NaOH and CaHPO4;; | |
-

-
disodium hydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| With soda slag In water pptn. of Ca-silicate with soda-slag;; in soln.; since the crude soda slag contains Na2S and phosphate, all heavy metal impurities are coprecipitated and the residual P content of the slag will be regained;; | |
-

-
phosphorus
Conditions
| Conditions | Yield |
|---|
| redn. CaHPO4 prepared from bone ashes; | |
-

-
potassium dihydrogenphosphate
Conditions
| Conditions | Yield |
|---|
| With KHSO4 at 50°C; | |
| With potassium hydrogensulfate at 50°C; | |
-
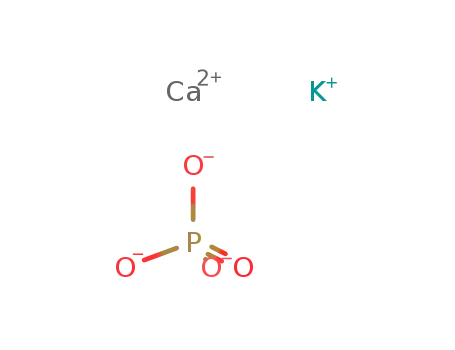
-
potassium calcium phosphate
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent) 3 h calcination at 1200°C;; | |
| In neat (no solvent) 3 h calcination at 1200°C;; | |
-

-
calcium carbonate
-
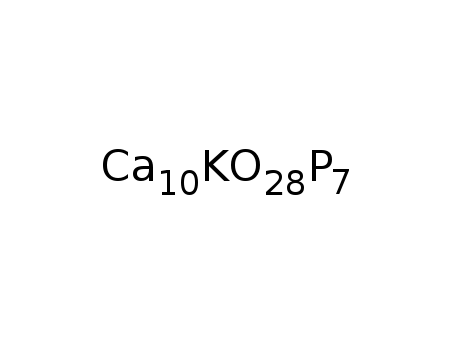
-
Ca10K(PO4)7
Conditions
| Conditions | Yield |
|---|
| In neat (no solvent, solid phase) grounding, annealing at 900 to 950°C for 80 to 100 h; progress of the reaction monitored by X-ray diffraction; | |
Calcium hydrogenphosphate dihydrate Chemical Properties
Product Name:Calcium hydrogenphosphate dihydrate(7789-77-7)
CAS No:7789-77-7
MF: CaH5O6P
MW: 172.09
mp : 109°C
density : 2.44 g.cm3 (20 °C )
Solubility in water:Soluble
Synonyms of Calcium hydrogenphosphate dihydrate(7789-77-7): CALCII HYDROGENOPHOSPHAS DIHYDRICUS;CALCIUM HYDROGEN PHOSPHATE;CALCIUM HYDROGEN PHOSPHATE-2-HYDRATE;CALCIUM HYDROGENPHOSPHATE DIHYDRATE;CALCIUM MONOHYDROGEN PHOSPHATE DIHYDRATE;CALCIUM PHOSPHATE, DIBASIC;CALCIUM PHOSPHATE DIBASIC, DIHYDRATE;CALCIUM PHOSPHATE DIHYDRATE, DIBASIC
Molecular Structure of Calcium hydrogenphosphate dihydrate(7789-77-7):

Calcium hydrogenphosphate dihydrate Uses
Calcium hydrogen phosphate(7789-77-7) is believed to be precursor of apatite and is found in guano-rich caves; formed by the interaction of guano with calcite and clay at a low ph
Calcium hydrogenphosphate dihydrate Toxicity Data With Reference
Inhalation:Material is irritating to mucous membranes and upper respiratory tract. May be harmful if inhaled.
Skin:Calcium hydrogen phosphate may cause skin irritation. May be harmful if absorbed through the skin.
Eyes:Calcium hydrogen phosphate cause eye irritation.
Ingestion:May be harmful if swallowed.
Calcium hydrogenphosphate dihydrate Consensus Reports
First Aid Measures:
Ingestion:If swallowed, wash out mouth with water provided person is conscious. Call a physician.
Inhalation:If inhaled, remove to fresh air. If not breathing give artificial respiration. If breathing is difficult, give oxygen.
Skin:In case of contact, immediately wash skin with soap and copious amounts of water.
Eyes:In case of contact, immediately flush eyes with copious amounts of water for at least 15 minutes.
Storage:Keep tightly closed.
Calcium hydrogenphosphate dihydrate Safety Profile
Hazard Codes : Xi
Risk Statements : 36/37/38
Safety Statements : 26-36
WGK Germany : 1
Calcium hydrogenphosphate dihydrate Specification
Personal Protection:Compatible chemical-resistant gloves. Chemical safety goggles.
Fire Fighting:Extinguish using Water spray. Carbon dioxide, dry chemical powder, or appropriate foam. Wear self-contained breathing apparatus and protective clothing to prevent contact with skin and eyes.
 Xi
Xi

















































































