-
Name
D-Glucurone
- EINECS 251-053-3
- CAS No. 32449-92-6
- Article Data29
- CAS DataBase
- Density 1.749 g/cm3
- Solubility soluble in water
- Melting Point 172-175 °C(lit.)
- Formula C6H8O6
- Boiling Point 403.5 °C at 760 mmHg
- Molecular Weight 176.126
- Flash Point 174.9 °C
- Transport Information
- Appearance white crystals or crystalline powder
- Safety 24/25-36-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Glucuronicacid, g-lactone, D- (6CI,8CI);D-Glucuronic acid lactone;D-Glucurono-3,6-lactone;D-Glucurono-g-lactone;D-Glucuronolactone;Dicurone;Glucoxy;Glucurone;D-Glucuronic acid, gamma-lactone;Glucuronolactone;Glucuronosan;Glycurone;Guronsan;NSC 656;
- PSA 104.06000
- LogP -2.80660
Synthetic route

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran; N,N-dimethyl-formamide at 0 - 20℃; | A n/a B n/a C 5% |

-
-
6556-12-3
D-Glucuronic acid

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| untersucht wurde das Lactonisierungsgleichgewicht in wss. Loesungen vom pH 7 - 8.5; |
-
-
87-73-0
D-glucaric acid

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With sodium amalgam; sulfuric acid; water |
-
-
87-89-8
D-myo-inositol

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| beim Behandeln mit einem Enzym-Praeparat aus Ratten-Nieren; |
-
-
709-50-2
methyl beta-D-glucopyranoside

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With water; oxygen; sodium hydrogencarbonate in Gegenwart von Platin und Erhitzen des Reaktionsprodukts mit wasserhaltiger Ameisensaeure; |
-
-
97-30-3
methyl-alpha-D-glucopyranoside

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With water; oxygen; sodium hydrogencarbonate in Gegenwart von Platin und Erhitzen des Reaktionsprodukts mit wasserhaltiger Ameisensaeure; | |
| With dinitrogen tetraoxide anschl. mit wasserhaltiger Ameisensaeure; | |
| With tetrachloromethane; dinitrogen tetraoxide Erwaermen des nach Behandeln mit wss. H2SO4 isolierten Reaktionsprodukts mit Butan-1-ol und konz. H2SO4 und anschl. Erwaermen mit wss. H2SO4; |
-
-
2671-19-4
O1,O2-isopropylidene-α-D-glucofuranuronic acid

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water | |
| With water anschl. Einengen der Reaktionsloesung; |
-
-
114127-73-0
(+)-Menthol glucuronide

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With sulfuric acid; water anschl. Einengen der von H2SO4 befreiten Reaktionsloesung; |
-
-
6920-57-6, 7259-41-8, 10345-04-7, 13168-11-1, 46825-92-7, 58865-20-6, 69127-38-4
α-D-glucopyranuronic acid 1-phosphate

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With formic acid; water |
-
-
13100-46-4
1,2,3,4-tetra-O-acetyl-β-D-glucopyranose

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With potassium permanganate; acetic acid; acetone anschl. mit Aceton und Ba(OH)2 in Wasser, Behandeln des danach isolierten Barium-Salzes mit wss. H2SO4 und anschl. Einengen der von H2SO4 befreiten wss. Reaktionsloesung; |

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With sulfuric acid; water anschl. Einengen der von H2SO4 befreiten Reaktionsloesung; |

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ethanol; water anschl. Einengen der Reaktionsloesung; |

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With water; nitric acid anschl. Erhitzen mit wss. HCl auf 150grad; |

| Conditions | Yield |
|---|---|
| Stage #1: D-glucuronic acid With resin-bound fluoride In N,N-dimethyl-formamide for 3h; Stage #2: allyl bromide In N,N-dimethyl-formamide at 40℃; for 20h; Further stages. Title compound not separated from byproducts.; |


| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid for 2h; Reflux; |

-
A
-
3615-41-6
L-Rhamnose

-
B
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid for 2h; Reflux; |

-
A
-
3615-41-6
L-Rhamnose

-
B
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid for 2h; Reflux; |
-
-
123506-40-1
6-hydrazino-1,3-dimethyl-1H-pyrimidine-2,4-dione

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
69471-96-1
6-{N'-[(S)-2-((2R,3R,4S)-3,4-Dihydroxy-5-oxo-tetrahydro-furan-2-yl)-2-hydroxy-eth-(Z)-ylidene]-hydrazino}-1,3-dimethyl-1H-pyrimidine-2,4-dione

| Conditions | Yield |
|---|---|
| In methanol for 5h; Heating; | 94% |
-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
22140-11-0
D-glucuronamide

| Conditions | Yield |
|---|---|
| With ammonia In ethanol at 20℃; for 5h; | 79.6% |
-
-
769-42-6
1,3-dimethylbarbituric acid

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
129878-35-9
disodium 5-(β-D-glucopyranosyluronate)-1,3-dimethylbarbiturate

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 80℃; for 5h; | 77% |
-
-
67-52-7
BARBITURIC ACID

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
129878-34-8
disodium 5-(β-D-glucopyranosysluronate)barbiturate

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 80℃; for 5h; | 73% |
-
-
24016-65-7
D-glucuronic acid sodium salt

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
129878-34-8
disodium 5-(β-D-glucopyranosysluronate)barbiturate

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 80℃; for 5h; | 66% |
-
-
108-24-7
acetic anhydride

-
-
32449-92-6
D-glucurono-6,3-lactone

-
A
-
7355-18-2
(2S,3S,4S,5R,6S)-3,4,5,6-Tetraacetoxy-tetrahydro-pyran-2-carboxylic acid methyl ester

-
B
-
5432-32-6
methyl 1,2,3,4-tetraacetyl-α-D-glucopyranuronate

| Conditions | Yield |
|---|---|
| With pyridine; sodium hydroxide In methanol for 1.5h; | A 39% B 37% |
| Stage #1: D-glucurono-6,3-lactone With sodium In methanol at 0℃; for 5h; Inert atmosphere; Stage #2: acetic anhydride With perchloric acid at 20 - 40℃; Inert atmosphere; Overall yield = 93 %; Overall yield = 96.5 g; |

-
-
67-56-1
methanol

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
17681-01-5, 17681-02-6, 18486-27-6, 18587-77-4, 72521-45-0, 72521-46-1, 73892-53-2, 73892-55-4, 135414-74-3, 135414-75-4
methyl β-D-glucofuranosidurono-6,3-lactone

| Conditions | Yield |
|---|---|
| in Gegenwart eines Kationen-Austauschers; |
-
-
67-56-1
methanol

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
17681-01-5
methyl α-D-glucofuranosidurono-6,3-lactone

| Conditions | Yield |
|---|---|
| in Gegenwart eines Kationen-Austauschers; |
-
-
89897-33-6
2,6-dimethyl-pyrimidine-4-carboxylic acid hydrazide

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
106275-46-1
2,6-dimethyl-pyrimidine-4-carboxylic acid-[(S)-2-((2R)-3c,4c-dihydroxy-5-oxo-tetrahydro-[2r]furyl)-2-hydroxy-ethylLiDenehydrazide]

| Conditions | Yield |
|---|---|
| With ethanol |

| Conditions | Yield |
|---|---|
| With sodium ethanolate | |
| With basic anion-exchanger |
-
-
540-63-6
ethane-1,2-dithiol

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
72251-95-7
Aldehydo-D-glucurono-6,3-lactone ethylene dithioacetal

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
75-87-6
chloral

-
-
32449-92-6
D-glucurono-6,3-lactone

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
100-01-6
4-nitro-aniline

-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
108752-94-9
1-(4-nitro-anilino)-1-deoxy-ξ-D-glucofuranuronic acid-3-lactone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; methanol | |
| With methanol; water; acetone |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
6556-12-3
D-Glucuronic acid

| Conditions | Yield |
|---|---|
| With barium dihydroxide; water | |
| With alkaline solution; water |
-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
3356-20-5
1-amino-1-deoxy-ξ-D-glucopyranuronic acid amide

| Conditions | Yield |
|---|---|
| With ethanol; ammonia |
-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
99094-37-8
O1-methyl-ξ-D-arabinofuranuronic acid methyl ester

| Conditions | Yield |
|---|---|
| With lead(IV) acetate; acetic acid Erwaermen des Reaktionsprodukts mit Chlorwasserstoff enthaltendem Methanol; |
-
-
32449-92-6
D-glucurono-6,3-lactone

-
-
3067-56-9, 20513-98-8, 25159-39-1, 26623-22-3, 29514-28-1, 85080-95-1
D-glucurono-3,6-lactone acetonide

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetone |
D(+)-Glucurono-3,6-lactone Chemical Properties
IUPAC Name: 2-(3,4-dihydroxy-5-oxooxolan-2-yl)-2-hydroxyacetaldehyde
Empirical Formula: C6H8O6
Molecular Weight: 176.1241g/mol
EINECS: 251-053-3
Structure of Glucuronic acid lactone (CAS NO.32449-92-6):
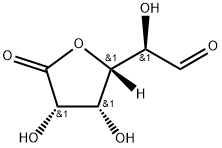
Index of Refraction: 1.597
Molar Refractivity: 34.31 cm3
Molar Volume: 100.6 cm3
Polarizability: 13.6×10-24cm3
Surface Tension: 99.5 dyne/cm
Density: 1.749 g/cm3
Flash Point: 174.9 °C
Enthalpy of Vaporization: 75.68 kJ/mol
Melting Point: 172-175 °C(lit.)
Boiling Point: 403.5 °C at 760 mmHg
Vapour Pressure: 3.42E-08 mmHg at 25°C
Water Solubility: soluble
Product Categories: Miscellaneous Natural Products;Biochemistry;Glucose;Sugar Acids;Sugars;Dextrins、Sugar & Carbohydrates
Canonical SMILES: C(=O)C(C1C(C(C(=O)O1)O)O)O
InChI: InChI=1S/C6H8O6/c7-1-2(8)5-3(9)4(10)6(11)12-5/h1-5,8-10H
InChIKey: UYUXSRADSPPKRZ-UHFFFAOYSA-N
D(+)-Glucurono-3,6-lactone Uses
Glucuronic acid lactone (CAS NO.32449-92-6) is used as a precursor for ascorbic acid synthesis. It is also used as detoxicant.
D(+)-Glucurono-3,6-lactone Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| cat | LD50 | subcutaneous | 4700mg/kg (4700mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| dog | LD50 | intravenous | 940mg/kg (940mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| dog | LD50 | subcutaneous | 4700mg/kg (4700mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| mouse | LD50 | intraperitoneal | 5797mg/kg (5797mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| mouse | LD50 | intravenous | 4586mg/kg (4586mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| mouse | LD50 | oral | > 20gm/kg (20000mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| mouse | LD50 | subcutaneous | 8210mg/kg (8210mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| rabbit | LD50 | intravenous | 940mg/kg (940mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| rabbit | LD50 | subcutaneous | 4700mg/kg (4700mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| rat | LD50 | intravenous | 3200mg/kg (3200mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| rat | LD50 | oral | 10700mg/kg (10700mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. | |
| rat | LD50 | subcutaneous | 4700mg/kg (4700mg/kg) | Drugs in Japan Vol. 6, Pg. 225, 1982. |
D(+)-Glucurono-3,6-lactone Consensus Reports
Reported in EPA TSCA Inventory.
D(+)-Glucurono-3,6-lactone Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 24/25-36-26
S24/25:Avoid contact with skin and eyes.
S36:Wear suitable protective clothing.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
WGK Germany: 2
RTECS: LZ8930000
HS Code: 29322980
D(+)-Glucurono-3,6-lactone Specification
Glucuronic acid lactone , its cas register number is 32449-92-6. It also can be called D-Glucofuranuronic acid, gamma-lactone ; D-Glucuronic acid lactone ; D-Glucurono-3,6-lactone ; D-Glucurono-6,3-lactone ; D-Glucuronolactone ; Dicurone ;
Glucoxy ; Glucurolactona ; Glucurolactone ; Glucurolactonum ; Glucurono-6,3-lactone ; Glucuronolactone ; Glucuronolattone ; Glucuronosan ; Gluronsan ; Guronsan ; Reulatt S.S. ; gamma-Glukurolakton . Glucuronic acid lactone (CAS NO.32449-92-6) is a white crystals or crystalline powder.
Related Products
- D(+)-Glucurono-3,6-lactone
- 32450-25-2
- 32451-85-7
- 32451-88-0
- 324519-54-2
- 324519-65-5
- 324519-66-6
- 324519-67-7
- 324519-68-8
- 324523-20-8
- 32452-45-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View