-
Name
DIBENZO(A,J)ANTHRACENE
- EINECS
- CAS No. 224-41-9
- Article Data26
- CAS DataBase
- Density 1.232g/cm3
- Solubility 8.629ug/L(25 oC)
- Melting Point 197.5°C
- Formula C22H14
- Boiling Point 524.7°Cat760mmHg
- Molecular Weight 278.353
- Flash Point 264.5°C
- Transport Information
- Appearance
- Safety Questionable carcinogen with experimental tumorigenic data. Mutation data reported. When heated to decomposition it emits acrid smoke and irritating fumes. See also ANTHRACENE.
- Risk Codes 25-36/37/38-52/53
-
Molecular Structure
- Hazard Symbols T
- Synonyms 1,2:7,8-Dibenzanthracene;3,4:5,6-Dibenzanthracene; Dibenzo-1,2,7,8-anthracene; NSC 90321
- PSA 0.00000
- LogP 6.29940
Synthetic route
-
-
868847-77-2
2,4',6',2''-tetravinyl-[1,1';3',1'']terphenyl

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With Grubbs catalyst first generation In dichloromethane at 35℃; for 23h; ring-closing olefin metathesis; | 98% |
-
-
130799-92-7
1,2,3,4,10,11,12,13-Octahydrodibenzanthracene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| palladium on activated charcoal In various solvent(s) Heating; | 93% |
-
-
81205-71-2
14-acetoxydibenzanthracene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With potassium hydroxide; zinc In 1,4-dioxane; water Heating; | 90% |
-
-
77321-42-7
2-(Naphthalene-2-carbonyl)-naphthalene-1-carboxylic acid

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With phosphorus; hydrogen iodide In acetic acid for 120h; Heating; | 84% |
-
-
73540-68-8
3-Naphthalen-2-yl-3H-naphtho[1,2-c]furan-1-one

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With hydrogen iodide; hypophosphorous acid In acetic acid for 240h; Heating; | 80% |
-
-
134576-19-5
7-(Methylthio)-dibenzanthracene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With nickel In methanol for 6h; Ambient temperature; | 78% |

| Conditions | Yield |
|---|---|
| With iodine In benzene for 1h; Irradiation; | A 70% B 8% |
| With iodine In benzene Irradiation; | A 70% B 8% |
-
-
103224-51-7
7,14-dihydro-14-ethoxy-7-(trimrthylsilyl)-7,14-epoxydibenzanthracene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With acetic acid; zinc for 14h; Heating; | 55% |

| Conditions | Yield |
|---|---|
| In benzene Irradiation; Yields of byproduct given; | A 39% B n/a C n/a |
| In benzene Irradiation; Yield given. Yields of byproduct given; |

-
-
41774-28-1
(2-methylnaphthalen-1-yl)(naphthalen-1-yl)methanone

-
A
-
53-70-3
dibenzo[a,h]anthracene

-
B
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| at 420℃; for 0.75h; | A 20% B 2% |
-
-
90-12-0
1-Methylnaphthalene

-
A
-
213-46-7
picene

-
B
-
53-70-3
dibenzo[a,h]anthracene

-
C
-
214-17-5
Benzo[b]chrysene

-
D
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| at 750℃; |

-
-
19399-64-5
5,6,8,9-Tetrahydrodibenzoanthracene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With carbon dioxide; palladium on activated charcoal at 300℃; |
-
-
41774-36-1
14H-dibenz[a,j]anthracen-7-one

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; zinc |
-
-
32277-35-3
1,2-dihydrocyclobutene[α]naphthalene

-
A
-
53-70-3
dibenzo[a,h]anthracene

-
B
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| at 890℃; |
-
-
130800-12-3
C22H30O2

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; 2,3-dicyano-5,6-dichloro-p-benzoquinone 1.) CHCl3, r.t., 2.) benzene, reflux; Yield given. Multistep reaction; | |
| Multi-step reaction with 2 steps 1: 1.) methanesulfonic acid, 2.) o-chloranil / 1.) CHCl3, r.t., 2.) benzene, reflux, 48 h 2: 93 percent / 10 percent Pd/C / various solvent(s) / Heating View Scheme |
-
-
81194-74-3
lithium salt of N,N-diethyl-1-naphthtamide

-
-
66-99-9
β-naphthaldehyde

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| 1.) diethyl ether, THF, -60 deg C 2.) Zn, KOH, pyridine, H2O, MeOH, reflux, 2.5 h 3.) ZnCl2, acetic acid, 30 min 4.) HI, hypophosphoric acid, 30 min; Yield given. Multistep reaction; |
-
-
89988-22-7
trans-9,10-dihydro-2-styrylphenanthrene

-
A
-
194-69-4
benzo[c]chrysene

-
B
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With iodine; 2,3-dicyano-5,6-dichloro-p-benzoquinone 1) benzene, irradiation, 7 h, 2) benzene, reflux, 14 h; Yield given. Multistep reaction. Yields of byproduct given; |
-
-
75-15-0
carbon disulfide

-
-
7446-70-0
aluminium trichloride

-
-
86-52-2
1-Chloromethylnaphthalene

-
-
2506-41-4
1-chloromethylnaphthalene

-
A
-
53-70-3
dibenzo[a,h]anthracene

-
B
-
226-88-0
benzo[a]naphthacene

-
C
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| Erhitzen des Reaktionsprodukts mit Kupfer-Pulver, neben groessren Mengen makromolekularer Substanzen; |

-
-
75-15-0
carbon disulfide

-
-
7446-70-0
aluminium trichloride

-
-
86-52-2
1-Chloromethylnaphthalene

-
-
91-57-6
2-Methylnaphthalene

-
A
-
53-70-3
dibenzo[a,h]anthracene

-
B
-
226-88-0
benzo[a]naphthacene

-
C
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| Erhitzen des Reaktionsprodukts mit Kupfer-Pulver; |


-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; zinc |

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| at 340 - 350℃; under 3 - 4 Torr; |
-
-
868847-74-9
1,5-dibromo-2,4-divinylbenzene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 93 percent / tetrakis(triphenylphosphine)palladium(0); sodium carbonate / 1,2-dimethoxy-ethane; ethanol; H2O / 16 h / Heating 2: 98 percent / bis(tricyclohexylphosphine)(benzylidene)ruthenium dichloride / CH2Cl2 / 23 h / 35 °C View Scheme |
-
-
58997-70-9
1,5-dibromo-2,4-bis(dibromomethyl)benzene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: 96 percent / silver nitrate / ethanol; H2O / 0.5 h / Heating 2.1: potassium tert-butoxide / tetrahydrofuran / 0.08 h 2.2: 61 percent / tetrahydrofuran / 25 °C 3.1: 93 percent / tetrakis(triphenylphosphine)palladium(0); sodium carbonate / 1,2-dimethoxy-ethane; ethanol; H2O / 16 h / Heating 4.1: 98 percent / bis(tricyclohexylphosphine)(benzylidene)ruthenium dichloride / CH2Cl2 / 23 h / 35 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 88 percent / N-bromosuccinimide / CCl4 / 9 h / Heating; irradiation 2.1: 96 percent / silver nitrate / ethanol; H2O / 0.5 h / Heating 3.1: potassium tert-butoxide / tetrahydrofuran / 0.08 h 3.2: 61 percent / tetrahydrofuran / 25 °C 4.1: 93 percent / tetrakis(triphenylphosphine)palladium(0); sodium carbonate / 1,2-dimethoxy-ethane; ethanol; H2O / 16 h / Heating 5.1: 98 percent / bis(tricyclohexylphosphine)(benzylidene)ruthenium dichloride / CH2Cl2 / 23 h / 35 °C View Scheme |
-
-
97094-37-6
2,4-dibromobenzene-1,5-dicarboxaldehyde

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: potassium tert-butoxide / tetrahydrofuran / 0.08 h 1.2: 61 percent / tetrahydrofuran / 25 °C 2.1: 93 percent / tetrakis(triphenylphosphine)palladium(0); sodium carbonate / 1,2-dimethoxy-ethane; ethanol; H2O / 16 h / Heating 3.1: 98 percent / bis(tricyclohexylphosphine)(benzylidene)ruthenium dichloride / CH2Cl2 / 23 h / 35 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: 58 percent / Br2; I2 / 16 h / 20 °C 2.1: 88 percent / N-bromosuccinimide / CCl4 / 9 h / Heating; irradiation 3.1: 96 percent / silver nitrate / ethanol; H2O / 0.5 h / Heating 4.1: potassium tert-butoxide / tetrahydrofuran / 0.08 h 4.2: 61 percent / tetrahydrofuran / 25 °C 5.1: 93 percent / tetrakis(triphenylphosphine)palladium(0); sodium carbonate / 1,2-dimethoxy-ethane; ethanol; H2O / 16 h / Heating 6.1: 98 percent / bis(tricyclohexylphosphine)(benzylidene)ruthenium dichloride / CH2Cl2 / 23 h / 35 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: 55 percent / Zn, AcOH / 14 h / Heating View Scheme |
-
-
134576-15-1
7-(Methylthio)-5,6-dihydrodibenzanthracene

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 96 percent / DDQ / dioxane / 24 h / Heating 2: 78 percent / Raney-Nickel / methanol / 6 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: SOCl2, dimethyl formamide / benzene / 2 h 2: 18.5 g / diethyl ether / 1 h / 0 °C 3: 1.) TMEDA, triphenylmethyl chloride, sec-BuLi / 1.) ether, cyclohexane, -78 deg C, 1 h, 2.) a) -78 deg, 1 h, b) room temp., 3 h 4: 80 percent / HI, H3PO2 / acetic acid / 240 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| In benzene Irradiation; | A 91% B 9% |
| In benzene Irradiation; | A 11% B 89% |

-
-
224-41-9
dibenz[a,j]anthracene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; iron(III) chloride In tetrachloromethane Bromination; Heating; | 91% |

| Conditions | Yield |
|---|---|
| In various solvent(s) at 91.5℃; Mechanism; Rate constant; analogous reaction of other polycyclic aromatic hydrocarbons; structure/reactivity correlations, application of theoretical models; |
DIBENZ(a,j)ANTHRACENE Chemical Properties
Product Name: Dibenzo(aj)anthracene (CAS NO.224-41-9)
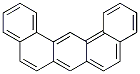
Molecular Formula: C22H14
Molecular Weight: 278.3466 g/mol
Mol File: 224-41-9.mol
Boiling point: 524.7 °C at 760 mmHg
Storage Temperature: 2-8°C
Flash Point: 264.5 °C
Density: 1.232 g/cm3
Surface Tension: 57.7 dyne/cm
Enthalpy of Vaporization: 76.86 kJ/mol
Vapour Pressure: 1.41E-10 mmHg at 25°C
XLogP3: 6.5
H-Bond Donor: 0
H-Bond Acceptor: 0
Heavy Atom Count: 22
Complexity: 363
Covalently-Bonded Unit Count: 1
DIBENZ(a,j)ANTHRACENE Toxicity Data With Reference
| 1. | mma-sat 1 µg/plate | MUREAV Mutation Research. 51 (1978),311. |
DIBENZ(a,j)ANTHRACENE Consensus Reports
IARC Cancer Review: Group 3 IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 7 (1987),p. 56.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) ; Animal Limited Evidence IMEMDT IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Man . 32 (1983),p. 309.(World Health Organization, Internation Agency for Research on Cancer,Lyon, France.: ) (Single copies can be ordered from WHO Publications Centre U.S.A., 49 Sheridan Avenue, Albany, NY 12210) .
DIBENZ(a,j)ANTHRACENE Safety Profile
Questionable carcinogen with experimental tumorigenic data. Mutation data reported. When heated to decomposition it emits acrid smoke and irritating fumes. See also ANTHRACENE.
DIBENZ(a,j)ANTHRACENE Specification
Dibenzo(aj)anthracene ,its CAS NO. is 224-41-9,the synonyms is 3,4,5,6-Dibenzanthracene ; Dibenzo-1,2,7,8-anthracene ; Dibenzo(a,j)anthracene ; Dibenz(a,j)anthracene ; Dibenz[a,j]anthracene (purity) ; 74798, Dibenz[a,j]anthracene (purity) .
Related Products
- Dibenz(a,h)acridine
- Dibenz(a,h)acridine 3,4-diol-1,2-epoxide
- Dibenz(a,h)anthracen-5-ol
- Dibenz(a,h)anthracene
- Dibenz(a,j)aceanthrylene
- Dibenz(a,j)acridine
- Dibenz(a,j)acridine, methosulfate
- DIBENZ(a,j)ANTHRACENE
- 224-42-0
- 2244-21-5
- 22442-48-4
- 224436-97-9
- 224441-73-0
- 224452-66-8
- 22445-41-6
- 22445-42-7
- 22446-12-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View