-
Name
Ethyl acetoacetate
- EINECS 205-516-1
- CAS No. 141-97-9
- Article Data296
- CAS DataBase
- Density 1.021 g/cm3
- Solubility water: 116 g/L (20 °C)
- Melting Point -43 °C
- Formula C6H10O3
- Boiling Point 180.6 °C at 760 mmHg
- Molecular Weight 130.144
- Flash Point 67 °C
- Transport Information UN 1993
- Appearance Colourless liquid
- Safety 26-24/25
- Risk Codes 36
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Ethyl beta-ketobutyrate;FEMA No. 2415;Acetoacetic ester;Ethyl 3-oxobutyrate;Active acetyl acetate;3-Oxobutanoic acid ethyl ester;Ethyl acetylacetate;Ethylester kyseliny acetoctove [Czech];Acetoctan ethylnaty [Czech];1-Ethoxybutane-1,3-dione;Active acetylacetate;Butanoic acid, 3-oxo-, ethyl ester;Ethyl 3-oxobutanoate;
- PSA 43.37000
- LogP 0.52860
Synthetic route
-
-
64-17-5
ethanol

-
-
2911-22-0
6-methyl-4-oxo-2-thioxo-3,4-dihydro-2H-1,3-oxazine

-
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 1h; Ambient temperature; | 100% |

-
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With water at 20℃; under 900.09 Torr; for 0.226667h; Flow reactor; | 98% |

| Conditions | Yield |
|---|---|
| With zinc In tetrahydrofuran 1) 30 deg C, 2 h, 2) reflux, 18 h; | 97% |

| Conditions | Yield |
|---|---|
| With cerium(III) chloride; sodium iodide In acetonitrile for 1.5h; Heating; | 96% |
| With polyaniline-sulfate salt; water for 0.75h; Heating; | 90% |
| With boron trifluoride diethyl etherate; tetraethylammonium iodide In chloroform for 6h; Heating; | 69% |
| palladium (II) ion In acetone for 23h; Ambient temperature; | 97 % Chromat. |
-
-
92208-06-5
3,3-dimethoxybutyric acid ethyl ester

| Conditions | Yield |
|---|---|
| With water at 20℃; under 900.09 Torr; for 0.226667h; Flow reactor; | 96% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 85 - 125℃; for 6h; | 95% |
| With sulfuric acid | |
| With dmap |

| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate at 110℃; for 3h; Neat (no solvent); | 94% |
| In neat (no solvent) at 110℃; for 3h; Green chemistry; | 92% |
| With zinc(II) sulfate In toluene at 80℃; for 6h; | 87% |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 94% B n/a |

-
-
141-78-6
ethyl acetate

-
-
54013-04-6
ethyl 5-chloro-2-pyrazinecarboxylate

-
A
-
1215002-59-7
ethyl 3-(5-chloropyrazin-2-yl)-3-oxopropanoate

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -50 - -30℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 94% B n/a |


| Conditions | Yield |
|---|---|
| Esterification; | 92% |
| In 5,5-dimethyl-1,3-cyclohexadiene Reflux; | 69% |
| In 5,5-dimethyl-1,3-cyclohexadiene at 150℃; |
-
-
141630-41-3
3-(2-Methyl-[1,3]dithiolan-2-yl)-propionic acid ethyl ester

-
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In methanol; water Heating; | 92% |
-
-
614-18-6
3-pyridinecarboxylic acid ethyl ester

-
-
141-78-6
ethyl acetate

-
A
-
6283-81-4
3-oxo-3-pyridin-3-yl-propionic acid ethyl ester

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 92% B n/a |

-
-
26805-39-0
(Z)-ethyl 3-acetoxybut-2-enoate

-
-
65753-93-7
N-methyl-2-dimethylamino-acetohydroxamic acid

-
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Ambient temperature; | 91% |
-
-
7238-62-2
ethyl 2-chloro-4-methylthiazole-5-carboxylate

-
-
141-78-6
ethyl acetate

-
A
-
1215002-61-1
ethyl 3-(2-chloro-4-methylthiazol-5-yl)-3-oxopropanoate

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 91% B n/a |

-
-
66278-17-9
(2-Methyl-1,3-dithiolan-2-yl)essigsaeure-ethylester

-
A
-
141-97-9
ethyl acetoacetate

-
B
-
540-63-6
ethane-1,2-dithiol

| Conditions | Yield |
|---|---|
| With magnesium(II) perchlorate; water; methylene green In acetonitrile Irradiation; | A 90% B n/a |
-
-
141-78-6
ethyl acetate

-
-
6924-68-1
ethyl 2-pyrazinecarboxylate

-
A
-
62124-77-0
3-oxo-3-pyrazin-2-yl-propionic acid ethyl ester

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 88% B n/a |


| Conditions | Yield |
|---|---|
| With palladium diacetate; Dess-Martin periodane In water; acetonitrile at 50℃; Wacker-Tsuji Olefin Oxidation; Inert atmosphere; | 88% |
| With iron(III) sulfate hydrate; palladium dichloride In water; acetonitrile at 45℃; for 2.5h; Wacker Oxidation; Inert atmosphere; | 87% |
| With chromium(VI) oxide; palladium dichloride In water; acetonitrile at 20 - 60℃; for 7h; Wacker-Tsuji Olefin Oxidation; | 85% |
| With manganese(IV) oxide; palladium dichloride In water; acetonitrile at 60℃; for 24h; | 58% |

| Conditions | Yield |
|---|---|
| With potassium fluoride on basic alumina In N,N-dimethyl-formamide at 18 - 25℃; Claisen Condensation; | 87.5% |
| With potassium tert-butylate at 80℃; for 0.333333h; Product distribution; Further Variations:; Reaction partners; Claisen condensation; | 73% |
| With calcium |
-
-
1570-45-2
isonicotinic acid ethylester

-
-
141-78-6
ethyl acetate

-
A
-
26377-17-3
ethyl 3-oxo-3-(4-pyridyl)propanoate

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 87% B n/a |


| Conditions | Yield |
|---|---|
| With zinc In tetrahydrofuran 1) 30 deg C, 2 h, 2) reflux, 18 h; | 86% |
-
-
89415-67-8
ethyl 2,2-dibromoacetoacetate

-
A
-
84911-18-2, 609-13-2
ethyl 2-bromoacetoacetate

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With water; 1-methyl-3-pentyl-1H-imidazolium tetrafluoroborate at 125℃; under 2585.74 Torr; for 0.0333333h; microwave irradiation; | A 85% B n/a |
-
-
50458-78-1
ethyl isoquinoline-1-carboxylate

-
-
141-78-6
ethyl acetate

-
A
-
141-97-9
ethyl acetoacetate

-
B
-
92200-07-2
ethyl 3-(isoquinolin-1-yl)-3-oxopropanoate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A n/a B 85% |

-
-
141-78-6
ethyl acetate

-
-
42839-08-7
pyrimidine-2-carboxylic acid ethyl ester

-
A
-
1093114-80-7
ethyl β-oxo-2-pyrimidininepropanoate

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 85% B n/a |

-
-
109-97-7
pyrrole

-
-
122135-84-6
ethyl (Z)-3-(((trifluoromethyl)sulfonyl)oxy)but-2-enoate

-
B
-
4341-76-8
Ethyl 2-butynoate

-
C
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| Stage #1: pyrrole With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); XPhos In toluene at 60℃; for 0.5h; Stage #2: ethyl (Z)-3-(((trifluoromethyl)sulfonyl)oxy)but-2-enoate In toluene at 60℃; for 3.5h; | A 84% B n/a C n/a |

-
-
92208-06-5
3,3-dimethoxybutyric acid ethyl ester

-
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With water at 20℃; under 900.09 Torr; for 0.226667h; Flow reactor; | 96% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 85 - 125℃; for 6h; | 95% |
| With sulfuric acid | |
| With dmap |

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 84% B n/a |

-
-
115479-07-7
2-Acetyl-4,4,5,5,6,6,7,7,8,8,9,9,9-tridecafluoro-3-oxo-nonanoic acid ethyl ester

-
-
95-54-5
1,2-diamino-benzene

-
A
-
141-97-9
ethyl acetoacetate

-
B
-
119403-54-2
2-(perfluorohexyl)benzimidazole

| Conditions | Yield |
|---|---|
| In ethanol for 4h; Heating; | A n/a B 83% |

-
-
141-78-6
ethyl acetate

-
-
75154-68-6
methyl (2S)-N-tritylaziridine-2-carboxylate

-
A
-
141-97-9
ethyl acetoacetate

-
B
-
236742-97-5
(4S)-3-oxo-4,5-(N-triphenylmethylepimino)pentanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: ethyl acetate With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 1h; Metallation; Stage #2: methyl (2S)-N-tritylaziridine-2-carboxylate In tetrahydrofuran; hexane at -78℃; for 12h; Claisen condensation; | A n/a B 78% |

-
-
2524-52-9
ethyl-2-picolinate

-
-
141-78-6
ethyl acetate

-
A
-
26510-52-1
ethyl 2-pyridylcarbonylacetate

-
B
-
141-97-9
ethyl acetoacetate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -40℃; for 0.333333h; Claisen condensation; Inert atmosphere; | A 78% B n/a |

-
-
30645-78-4
3-(4-chloro-phenyl)-6-methyl-[1,3]oxazine-2,4-dione

-
A
-
101-92-8
4'-Chloroacetoacetanilide

-
B
-
141-97-9
ethyl acetoacetate

-
C
-
2621-80-9
ethyl p-chlorophenylcarbamate

| Conditions | Yield |
|---|---|
| With triethylamine at 95 - 100℃; for 2h; | A 10% B 30% C 77% |

-
-
30645-78-4
3-(4-chloro-phenyl)-6-methyl-[1,3]oxazine-2,4-dione

-
-
121-44-8
triethylamine

-
A
-
101-92-8
4'-Chloroacetoacetanilide

-
B
-
141-97-9
ethyl acetoacetate

-
C
-
2621-80-9
ethyl p-chlorophenylcarbamate

| Conditions | Yield |
|---|---|
| With ethanol at 95 - 100℃; for 2h; | A 10% B 30% C 77% |

-
-
504-29-0
2-aminopyridine

-
-
141-97-9
ethyl acetoacetate

-
-
1693-94-3
2-methyl-4H-pyrido[1,2-a]pyrimidin-4-one

| Conditions | Yield |
|---|---|
| With bismuth(III) chloride at 100℃; for 3h; Green chemistry; | 100% |
| With Al3PW12O40 In ethanol at 80℃; for 0.2h; Reagent/catalyst; Green chemistry; | 93% |
| In ethylene glycol at 100℃; for 4h; Solvent; Temperature; Green chemistry; | 89% |
-
-
14371-10-9
(E)-3-phenylpropenal

-
-
141-97-9
ethyl acetoacetate

-
-
137153-74-3
2-oxo-6-phenyl-cyclohex-3-enecarboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| In ethanol | 100% |
| With ethanol; sodium ethanolate at -10℃; |
-
-
75-77-4
chloro-trimethyl-silane

-
-
141-97-9
ethyl acetoacetate

-
-
13257-83-5
ethyl 3-trimethylsiloxy-2-butenoate

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran; hexane at 21 - 45℃; for 3h; | 100% |
| With triethylamine In tetrahydrofuran Ambient temperature; | 89% |
| Stage #1: chloro-trimethyl-silane; ethyl acetoacetate In tetrahydrofuran; hexane at 0 - 5℃; for 0.5h; Inert atmosphere; Stage #2: With triethylamine In tetrahydrofuran; hexane at 0 - 20℃; Inert atmosphere; | 87% |
-
-
2896-60-8
4-ethyl-1,3-benzenediol

-
-
141-97-9
ethyl acetoacetate

-
-
1484-73-7
6-Ethyl-7-hydroxy-4-methylcumarin

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 100℃; for 0.5h; Pechmann condensation; Microwave irradiation; regioselective reaction; | 100% |
| With boron trifluoride diethyl etherate at 105 - 108℃; for 0.333333h; | 95.2% |
| With boron trifluoride diethyl etherate at 105 - 108℃; for 0.333333h; Mechanism; other 4-alkylresorcinols; also ethyl benzoylacetate; | 95.2% |
-
-
95-71-6
2-methylbenzene-1,4-diol

-
-
141-97-9
ethyl acetoacetate

-
-
5248-20-4
6-hydroxy-4,7-dimethyl-2H-chromen-2-one

| Conditions | Yield |
|---|---|
| With sulfuric acid at 0℃; for 1h; Pechmann Condensation; Milling; | 100% |
| With sulfuric acid Pechmann Condensation; Inert atmosphere; | 77% |
| With sulfuric acid | |
| With sulfuric acid at 20℃; for 24h; Pechmann condensation; Inert atmosphere; |
-
-
102-96-5
(2-nitroethenyl)benzene

-
-
141-97-9
ethyl acetoacetate

-
-
72709-62-7
ethyl 2-acetyl-4-nitro-3-phenylbutyrate

| Conditions | Yield |
|---|---|
| With nickel polymer catalyst In chloroform for 48h; Reflux; | 100% |
| With C23H37N3O9S In dichloromethane at 20℃; for 16h; asymmetric Michael addition; enantioselective reaction; | 99% |
| With C31H36N6OS3 In toluene at 25℃; for 48h; Michael Addition; enantioselective reaction; | 99% |
-
-
141-97-9
ethyl acetoacetate

-
-
555-16-8
4-nitrobenzaldehdye

-
-
57-13-6
urea

-
-
123371-45-9
ethyl 6-methyl-4-(4-nitrophenyl)-2-oxo-1,2,3,4-tetrahydro-5-pyrimidinecarboxylate

| Conditions | Yield |
|---|---|
| With 1-methylimidazole based ionic liquid terminated dendritic moiety prepared from 2,4,6-trichloro-1,3,5-triazine and 1,3-diaminopropane immobilized on 3-aminopropyltriethoxysilane functionalized bentonite In ethanol; water at 50℃; for 3h; Biginelli Pyrimidone Synthesis; Green chemistry; | 100% |
| With composite of cross-linked chitosan beads and a cyclodextrin nanosponge In water for 0.25h; Biginelli Pyrimidone Synthesis; Sonication; Green chemistry; | 100% |
| With guanidine In neat (no solvent) at 80℃; for 2h; Biginelli Pyrimidone Synthesis; | 99% |

-
-
141-97-9
ethyl acetoacetate

-
-
100-52-7
benzaldehyde

-
-
57-13-6
urea

-
-
123237-03-6, 5395-36-8
ethyl 6-methyl-4-phenyl-3,4-dihydropyrimidin-2(1H)-one-5-carboxylate

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate) In ethanol at 100℃; for 1h; Biginelli reaction; Microwave irradiation; Inert atmosphere; | 100% |
| With composite of cross-linked chitosan beads and a cyclodextrin nanosponge In water for 0.25h; Biginelli Pyrimidone Synthesis; Sonication; Green chemistry; | 100% |
| With Cl7Fe2(1-)*C6H9N2O2(1+); C8H15N2(1+)*C4H12B(1-) at 80℃; for 2h; Reagent/catalyst; Biginelli Pyrimidone Synthesis; | 99% |

-
-
141-97-9
ethyl acetoacetate

-
-
100-52-7
benzaldehyde

-
-
1165-06-6
Diethyl 2,6-dimethyl-4-phenyl-1,4-dihydropyridine-3,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With ammonia for 3h; Heating; | 100% |
| With C23H3BF16N2O; ammonium acetate In toluene at 100℃; for 10h; Hantzsch Dihydropyridine Synthesis; | 100% |
| With ammonium carbonate In water at 55 - 60℃; for 3.5h; Hantzsch pyridine synthesis; | 99% |

| Conditions | Yield |
|---|---|
| With piperidine at 20℃; | 100% |
| Stage #1: ethyl acetoacetate; salicylaldehyde With piperidine In ethanol for 24h; Stage #2: at 80℃; for 0.0166667h; Microwave irradiation; | 97% |
| With piperidine In ethanol at 80℃; for 0.0166667h; Solvent; Reagent/catalyst; Knoevenagel Condensation; Microwave irradiation; Green chemistry; | 97% |

| Conditions | Yield |
|---|---|
| In acetic acid Heating; | 100% |
| for 0.166667h; Irradiation; | 100% |
| at 0 - 90℃; for 1.5h; | 100% |

| Conditions | Yield |
|---|---|
| With zirconium(IV) phosphate at 110℃; for 0.166667h; Pechmann condensation; Microwave irradiation; chemoselective reaction; | 100% |
| With silica gel supported zirconyl chloride octahydrate at 90℃; for 0.583333h; Pechmann condensation reaction; | 98% |
| With tetrakis(actonitrile)copper(I) hexafluorophosphate at 25℃; for 0.166667h; Pechmann condensation; neat (no solvent); | 98% |
-
-
141-97-9
ethyl acetoacetate

-
-
1761-61-1
5-bromosalicyclaldehyde

-
-
2199-93-1
3-Acetyl-6-bromo-chromen-2-one

| Conditions | Yield |
|---|---|
| With piperidine In ethanol at 80℃; for 0.0166667h; Knoevenagel Condensation; Microwave irradiation; Green chemistry; | 100% |
| With zinc oxide nanoparticle at 120℃; under 7200.72 Torr; for 0.1h; Knoevenagel condensation; Microwave irradiation; Neat (no solvent); | 95% |
| With piperidine In acetonitrile at 20℃; for 4h; Knoevenagel Condensation; | 94.1% |
-
-
141-97-9
ethyl acetoacetate

-
-
563-47-3
3-Chloro-2-methylpropene

-
-
20962-70-3
ethyl 2-acetyl-4-methylpent-4-enoate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 40 - 75℃; for 6h; | 100% |
| With tetrabutylammomium bromide; potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 40 - 75℃; for 6h; | 100% |
| With tetrabutylammomium bromide; potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 40 - 75℃; for 6h; | 100% |

| Conditions | Yield |
|---|---|
| [Cl(C6F13C2H4)2SnOSn(C2H4C6F13)2Cl]2 In toluene for 6h; Heating; | 100% |
| With rhizopus niveus lipase; Pseudomonas sp. lipoprotein lipase; Candida antarctica lipase B immobilized on acrylic resin; Carica papaya protease In toluene at 40℃; for 48h; Enzymatic reaction; | 98% |
| 1-chloro-3-hydroxy-1,1,3,3-tetrabutyldistannoxane In toluene for 16h; Heating; | 96% |
-
-
141-97-9
ethyl acetoacetate

-
-
108-46-3
recorcinol

-
-
90-33-5, 79566-13-5
7-hydroxy-4-methyl-chromen-2-one

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 100℃; for 0.5h; Pechmann condensation; Microwave irradiation; regioselective reaction; | 100% |
| With silica-supported methanesulfonic acid catalyst In octadecane at 160℃; for 2h; Pechmann reaction; | 99% |
| With iron(III) chloride In neat (no solvent) at 70℃; for 12h; Pechmann Condensation; Green chemistry; | 99% |
-
-
141-97-9
ethyl acetoacetate

-
-
84911-18-2, 609-13-2
ethyl 2-bromoacetoacetate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; dihydrogen peroxide; potassium bromide In toluene at 20℃; for 1.5h; | 100% |
| With bromine; sodium hydride In tetrahydrofuran; dichloromethane | 100% |
| With hydrogenchloride; dihydrogen peroxide; potassium bromide In water; toluene at 20℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With trichloroisocyanuric acid In water; acetone at 20℃; for 15h; | 100% |
| With sodium hypochlorite In acetic acid; acetone at 0℃; for 1h; | 98% |
| Stage #1: ethyl acetoacetate With ammonium chloride In acetonitrile for 0.0833333h; Stage #2: With 1,3-dichloro-5,5-dimethylhydantoin In acetonitrile at 35℃; for 12h; | 98% |

| Conditions | Yield |
|---|---|
| With thionyl chloride In tetrachloromethane at 20℃; for 1h; | 100% |
| With sulfuryl dichloride In dichloromethane at 0 - 20℃; | 98.4% |
| With sulfuryl dichloride In tetrachloromethane at 20℃; for 1h; | 97% |
-
-
141-97-9
ethyl acetoacetate

-
-
70-11-1
α-bromoacetophenone

-
-
52313-46-9
ethyl 2-acetyl-4-oxo-4-phenylbutanoate

| Conditions | Yield |
|---|---|
| Stage #1: α-bromoacetophenone With sodium hydride In tetrahydrofuran; mineral oil at 20℃; for 0.5h; Inert atmosphere; Stage #2: ethyl acetoacetate In tetrahydrofuran at 20℃; for 4h; | 100% |
| Stage #1: ethyl acetoacetate With sodium hydride In tetrahydrofuran; mineral oil at 0℃; Inert atmosphere; Stage #2: α-bromoacetophenone In tetrahydrofuran; mineral oil at 20℃; Inert atmosphere; | 98% |
| Stage #1: ethyl acetoacetate With sodium hydride In tetrahydrofuran; mineral oil at 0℃; Inert atmosphere; Stage #2: α-bromoacetophenone In tetrahydrofuran; mineral oil at 0 - 20℃; Inert atmosphere; | 98% |
-
-
141-97-9
ethyl acetoacetate

-
-
149-73-5
trimethyl orthoformate

-
-
3510-99-4
3-methoxy-but-2-enoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol | 100% |
| With hydrogenchloride In methanol | 100% |
| Stage #1: ethyl acetoacetate; trimethyl orthoformate With sulfuric acid at 20℃; for 18h; Stage #2: With potassium carbonate for 0.5h; | 38% |
| Stage #1: ethyl acetoacetate; trimethyl orthoformate With sulfuric acid at 20℃; for 18h; Stage #2: With potassium carbonate for 0.5h; | 38% |
-
-
141-97-9
ethyl acetoacetate

-
-
56816-01-4
ethyl (S)-3-hydroxybutyrate

| Conditions | Yield |
|---|---|
| With [NEt2H2]-[{RuCl(S)-BINAP}2(μ-Cl)3]; hydrogen In methanol; ethanol at 50℃; under 22801.5 Torr; for 12h; Inert atmosphere; Autoclave; optical yield given as %ee; | 100% |
| With hydrogen In ethanol at 80℃; under 15001.5 Torr; for 10h; enantioselective reaction; | 99.5% |
| With C32H40F6O6P2Pd; hydrogen In acetone at 0℃; under 23272.3 Torr; for 12h; Autoclave; enantioselective reaction; | 99% |
-
-
123-75-1
pyrrolidine

-
-
141-97-9
ethyl acetoacetate

-
-
2723-42-4, 54716-02-8, 70526-06-6
ethyl 3-pyrrolidinobut-2-enoate

| Conditions | Yield |
|---|---|
| at 20℃; for 16h; Inert atmosphere; Schlenk technique; | 100% |
| In toluene for 1h; Dean-Stark; Reflux; Inert atmosphere; | 94.8% |
| With dichloro( 1,5-cyclooctadiene)platinum(ll); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In N,N-dimethyl-formamide at 60℃; for 5h; Inert atmosphere; Schlenk technique; Microwave irradiation; regioselective reaction; | 82% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; trimethyl orthoformate | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl acetoacetate With lithium diisopropyl amide In tetrahydrofuran; hexane at 0℃; for 1h; Stage #2: 1-bromo-butane In tetrahydrofuran; hexane at -78 - 20℃; for 14h; Further stages.; | 100% |
| Stage #1: ethyl acetoacetate With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.25h; Inert atmosphere; Stage #2: With n-butyllithium In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.25h; Inert atmosphere; Stage #3: 1-bromo-butane In tetrahydrofuran; mineral oil at 0 - 20℃; Inert atmosphere; | 73% |
| 70% | |
| With n-butyllithium; sodium hydride In tetrahydrofuran; pentane at 0 - 20℃; for 14h; | 38% |
| Stage #1: ethyl acetoacetate With n-butyllithium; sodium hydride In tetrahydrofuran at 0℃; Inert atmosphere; Stage #2: 1-bromo-butane at 0 - 25℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In 1,4-dioxane | 100% |
| With potassium hydroxide In N,N-dimethyl-formamide at 25℃; for 24h; | |
| Stage #1: ethyl acetoacetate With potassium carbonate In N,N-dimethyl-formamide for 1h; Stage #2: carbon disulfide In N,N-dimethyl-formamide for 2h; |
-
-
930-30-3
cyclopent-2-enone

-
-
141-97-9
ethyl acetoacetate

-
-
66077-94-9
ethyl 3-oxo-2-(3-oxocyclopentyl)butanoate

| Conditions | Yield |
|---|---|
| With hydrotalcite at 80℃; for 6h; Michael addition; | 100% |
| With 1,3,4,6,7,8-hexahydro-2H-pyrimido[1,2-a]pyrimidine In toluene at 25℃; for 10h; Michael reaction; | 99% |
| With zinc trifluoromethanesulfonate at 20℃; for 6h; | 95% |
-
-
820-71-3
methallyl acetate

-
-
141-97-9
ethyl acetoacetate

-
-
20962-70-3
ethyl 2-acetyl-4-methylpent-4-enoate

| Conditions | Yield |
|---|---|
| With isopropylmagnesium chloride; sodium hydride; 1,2-bis(diphenylphosphino)ethane nickel(II) chloride In tetrahydrofuran at 160℃; for 72h; Product distribution; other temp./react. time; | 100% |
| With isopropylmagnesium chloride; sodium hydride; 1,2-bis(diphenylphosphino)ethane nickel(II) chloride In tetrahydrofuran at 160℃; for 72h; | 100% |
| With potassium carbonate In tetrahydrofuran at 70℃; for 50h; Tsuji-Trost Allylation; | 63% |
Ethyl acetoacetate Consensus Reports
Reported in EPA TSCA Inventory.
Ethyl acetoacetate Specification
The Ethyl acetoacetate, with the CAS registry number 141-97-9, is also known as Acetoacetic ester; Ethyl 3-oxobutanoate; Ethyl beta-ketobutyrate; EAA. It belongs to the product categories of Carbonyl Compounds;Chemical Synthesis;E-H;Esters;Organic Building Blocks.Its EINECS number is 205-516-1.This chemical's molecular formula is C6H10O3 and molecular weight is 130.14. What's more,Its systematic name is Ethyl acetoacetate.It is a colorless liquid with a fruity odor.And it is stable,Incompatible with acids,bases,oxidizing agents,reducing agents,alkali metals,Combustible.The Ethyl acetoacetate may cause adverse health effects if ingested or inhaled, and it is irritate to skin, eyes and mucous membranes.When you use it ,you should avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.It should Store in a tightly closed container,in a cool,dry place.Keep away from sources of ignition.
Physical properties about Ethyl acetoacetate are:
(1)ACD/LogP: 0.716; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.72; (4)ACD/LogD (pH 7.4): 0.72; (5)ACD/BCF (pH 5.5): 2.06; (6)ACD/BCF (pH 7.4): 2.06; (7)ACD/KOC (pH 5.5): 58.42; (8)ACD/KOC (pH 7.4): 58.39; (9)#H bond acceptors: 3; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 3; (12)Index of Refraction: 1.408; (13)Molar Refractivity: 31.651 cm3; (14)Molar Volume: 128.178 cm3; (15)Surface Tension: 30.2719993591309 dyne/cm; (16)Density: 1.015 g/cm3; (17)Flash Point: 67.019 °C; (18)Enthalpy of Vaporization: 41.683 kJ/mol; (19)Boiling Point: 180.567 °C at 760 mmHg; (20)Vapour Pressure: 0.889999985694885 mmHg at 25°C.
Preparation of Ethyl acetoacetate:
Ethyl acetoacetate is produced industrially by treatment of diketene with ethanol which is a classic laboratory procedure. It is prepared via the Claisen condensation of ethyl acetate. Two moles of ethyl acetate condense to form one mole each of ethyl acetoacetate and ethanol.
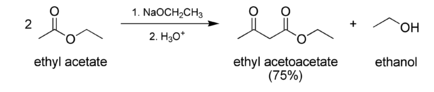
Uses of Ethyl acetoacetate:
Ethyl acetoacetate is used as starting material for the syntheses of alpha-substituted acetoacetic esters and cyclic compounds, e.g. pyrazole, pyrimidine and coumarin derivatives as well as intermediate for vitamins and pharmaceuticals.
You can still convert the following datas into molecular structure:
(1)SMILES:CCOC(=O)CC(=O)C;
(2)Std. InChI:InChI=1S/C6H10O3/c1-3-9-6(8)4-5(2)7/h3-4H2,1-2H3;
(3)Std. InChIKey:XYIBRDXRRQCHLP-UHFFFAOYSA-N.
The toxicity data of Ethyl acetoacetate as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 5105mg/kg (5105mg/kg) | Journal of Pharmaceutical Sciences. Vol. 60, Pg. 1810, 1971. | |
| rabbit | LD | skin | > 20mL/kg (20mL/kg) | Union Carbide Data Sheet. Vol. 3/12/1969, | |
| rat | LD50 | oral | 3980mg/kg (3980mg/kg) | Journal of Industrial Hygiene and Toxicology. Vol. 31, Pg. 60, 1949. |
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- ethyl (1R,2R)-1-phenyl-2-(trideuteriomethylamino)cyclohex-3-ene-1-carboxylate,hydrochloride
- Ethyl (1S,2R)-2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate hydrochloride
- Ethyl (2,4,6-trimethylbenzoyl) phenylphosphinate
- Ethyl (2-amino-4-hydroxy-6-methyl-5-pyrimidinyl)acetate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- ETHYL (2E,4Z)-DECADIENOATE
- Ethyl (2-hydroxyethyl)dimethyl-ammonium benzilate chloride
- Ethyl (2-mercaptoethyl) carbamate S-ester with O,O-dimethyl phosphorodithioate
- 141-98-0
- 141981-64-8
- 14199-15-6
- 141992-44-1
- 141998-77-8
- 141998-90-5
- 142002-57-1
- 1420-04-8
- 1420-07-1
- 142009-99-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View