-
Name
METHYL FLUORIDE
- EINECS 209-796-6
- CAS No. 593-53-3
- Article Data134
- CAS DataBase
- Density 0,877 g/cm3
- Solubility slight
- Melting Point -115°C
- Formula CH3 F
- Boiling Point -79°C
- Molecular Weight 34.0332
- Flash Point °C
- Transport Information UN 2454
- Appearance colourless gas with an ether-like odour
- Safety Narcotic in high concentrations. Acts as a simple asphyxiant. Burns with evolution of hydrogen fluoride. The flame is about as colorless as that of alcohol. When heated to decomposition it emits toxic fumes of F−.
- Risk Codes R12
-
Molecular Structure
-
Hazard Symbols

- Synonyms F 41;Fluoromethane; Freon 41; HFC 41; Methyl fluoride; Monofluoromethane; R 41; R 41(refrigerant)
- PSA 0.00000
- LogP 0.58570
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium fluoride In water at 100℃; for 5h; Solvent; Reagent/catalyst; Temperature; | 100% |
| With potassium fluoride In water at 100℃; under 1125.11 Torr; for 5h; Reagent/catalyst; Solvent; Temperature; | 100% |
| With potassium fluoride In sulfolane at 100℃; under 750.075 Torr; for 0.166667h; Solvent; Temperature; Autoclave; | 92% |
| With potassium fluoride | |
| With sodium fluoride In sulfolane |
-
-
382-26-3
1,1,1,3,3-pentafluoro-3-methoxy-2-trifluoromethylpropane

-
A
-
593-53-3
Methyl fluoride

-
B
-
382-22-9
3,3,3-trifluoro-2-trifluoromethyl-propionyl fluoride

| Conditions | Yield |
|---|---|
| With titanium(IV) oxide at 150℃; Reagent/catalyst; Temperature; Gas phase; Flow reactor; | A 99% B 100% |
| With alumina at 200℃; Catalytic behavior; Reagent/catalyst; Gas phase; Flow reactor; | A 99% B 91% |
| With alumina at 200℃; Catalytic behavior; Time; Gas phase; Flow reactor; | A 99% B 99% |
-
-
96887-05-7
C3CsF7O2S

-
-
77-78-1
dimethyl sulfate

-
A
-
593-53-3
Methyl fluoride

-
B
-
96025-71-7
1,2,2-trifluoro-1-(trifluoromethyl)ethanesulfonyl fluoride

| Conditions | Yield |
|---|---|
| In diethylene glycol dimethyl ether at 40 - 45℃; | A n/a B 12.2% C 95% |

-
-
84047-25-6
(Z)-4,4,5,5,5-Pentafluoro-3-methoxy-2-trifluoromethyl-pent-2-enoyl fluoride

-
A
-
593-53-3
Methyl fluoride

-
B
-
53352-88-8
pentafluoropropionyl(trifluoromethyl)ketene

| Conditions | Yield |
|---|---|
| titanium(IV) fluoride In neat (no solvent) Heating; | A n/a B 90.8% |

| Conditions | Yield |
|---|---|
| With hydrogen In neat (no solvent) formation on heating to 250°C;; | 89% |
-
-
425-88-7
1,1,2,2-tetrafluoro-1-methoxyethane

-
A
-
593-53-3
Methyl fluoride

-
B
-
2925-22-6
2,2-difluoroacetyl fluoride

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) | A n/a B 88% |
| aluminum phosphate treatment with HF at 300C for 72 h at 210℃; Product distribution / selectivity; Inert atmosphere; | |
| With monoaluminum phosphate; hydrogen fluoride at 200℃; Inert atmosphere; Autoclave; |
-
-
54376-60-2
2-(Trifluoromethyl)-3-methoxy-1,1,1,3,4,4,5,5,5-nonafluoropentane

-
A
-
593-53-3
Methyl fluoride

-
B
-
61637-91-0
2-(Trifluoromethyl)-1,1,1,4,4,5,5,5-octafluoropentan-3-one

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) | A n/a B 88% |
-
-
77946-89-5
1,3-Dimethoxy-2-(trifluoromethyl)-1,3,4,4,5,5,5-heptafluoro-1-pentene

-
-
84047-24-5
1,3-Dimethoxy-2-(trifluoromethyl)-1,1,4,4,5,5,5-heptafluoro-2-pentene

-
A
-
593-53-3
Methyl fluoride

-
B
-
84047-25-6
(Z)-4,4,5,5,5-Pentafluoro-3-methoxy-2-trifluoromethyl-pent-2-enoyl fluoride

| Conditions | Yield |
|---|---|
| titanium(IV) fluoride In neat (no solvent) Heating; | A n/a B 86.5% |

-
-
758-62-3
1,1,2,3,3-pentafluoro-1,3-dimethoxy-propane

-
A
-
593-53-3
Methyl fluoride

-
B
-
77946-94-2
2-Fluoro-propanedioyl difluoride

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) | A n/a B 84% |

-
A
-
593-53-3
Methyl fluoride

-
B
-
76944-21-3
perfluoro-2-pentene-4-one

| Conditions | Yield |
|---|---|
| With antimony pentafluoride cooling, then room t.; | A n/a B 84% |
-
-
382-34-3
1,1,1,2,3,3-hexafluoro-3-methoxypropane

-
A
-
593-53-3
Methyl fluoride

-
B
-
6065-84-5
2,3,3,3-tetrafluoropropionylfluoride

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) | A n/a B 82% |

| Conditions | Yield |
|---|---|
| hydrogen fluoride; boron trifluoride at 300℃; under 114000 Torr; for 6h; | A 1% B 9.5% C 80.7% |


-
A
-
593-53-3
Methyl fluoride

-
B
-
76944-22-4
3-hydroperfluoropentane-2-one

| Conditions | Yield |
|---|---|
| With antimony pentafluoride cooling, then room t.; | A n/a B 79% |
-
-
115-10-6
Dimethyl ether

-
A
-
34557-54-5
methane

-
B
-
593-53-3
Methyl fluoride

-
C
-
79-20-9
acetic acid methyl ester

-
D
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| With carbon monoxide; hydrogen fluoride; boron trifluoride at 190℃; under 121600 Torr; for 24h; Product distribution; variation of molar ratios, temperature, time; other catalysts; | A 0.1% B 3% C 76.1% D 20.1% |

-
-
115-10-6
Dimethyl ether

-
A
-
34557-54-5
methane

-
B
-
593-53-3
Methyl fluoride

-
C
-
79-20-9
acetic acid methyl ester

-
D
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| hydrogen fluoride; boron trifluoride at 190℃; under 121600 Torr; for 24h; | A 0.1% B 3% C 76.1% D 20.1% |

-
-
76-38-0
methoxyflurane

-
A
-
74-87-3
methylene chloride

-
B
-
593-53-3
Methyl fluoride

-
C
-
359-31-9
dichloroacetyl fluoride

-
D
-
661-75-6
2,2,2-Trichloro-1,1-difluoroethyl methyl ether

| Conditions | Yield |
|---|---|
| Stage #1: methoxyflurane With potassium hydroxide Dehydrofluorination; Heating; Stage #2: With chlorine at -40℃; Chlorination; Stage #3: With antimonypentachloride at 40℃; for 0.833333h; Fluorination; Decomposition; | A n/a B n/a C n/a D 74% |

-
-
77946-89-5
1,3-Dimethoxy-2-(trifluoromethyl)-1,3,4,4,5,5,5-heptafluoro-1-pentene

-
-
84047-24-5
1,3-Dimethoxy-2-(trifluoromethyl)-1,1,4,4,5,5,5-heptafluoro-2-pentene

-
A
-
593-53-3
Methyl fluoride

-
B
-
53352-88-8
pentafluoropropionyl(trifluoromethyl)ketene

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) | A n/a B 67% |

-
-
84047-27-8
1,1,1,2,2-Pentafluoro-2-(1,2,2-trifluoro-2-methoxy-ethoxy)-ethane

-
A
-
593-53-3
Methyl fluoride

-
B
-
53997-64-1
Difluoromethyl 1,1,2,2,2-pentafluoroethyl ether

-
C
-
77946-91-9
Fluoro-pentafluoroethyloxy-acetyl fluoride

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) Ambient temperature; | A n/a B 27% C 60% |

-
-
420-37-1
trimethoxonium tetrafluoroborate

-
A
-
34557-54-5
methane

-
B
-
74-84-0
ethane

-
C
-
593-53-3
Methyl fluoride

-
D
-
74-85-1
ethene

-
E
-
115-10-6
Dimethyl ether

| Conditions | Yield |
|---|---|
| With sodium hydride In Dimethyl ether for 8h; Product distribution; Mechanism; Ambient temperature; autoclave; other temperatures; also without solvent; | A 58.4% B 2.1% C 4.6% D 2.5% E 32.4% |

-
-
89909-24-0
Dimethyl((trimethylsilyl)methyl)oxonium Tetrafluoroborate

-
A
-
593-53-3
Methyl fluoride

-
B
-
115-10-6
Dimethyl ether

-
C
-
420-56-4
trimethylsilyl fluoride

-
D
-
10132-71-5
dimethylethylfluorosilane

-
E
-
14704-14-4
(methoxymethyl)trimethylsilane

-
F
-
28871-61-6
Fluormethyltrimethylsilan

| Conditions | Yield |
|---|---|
| With cesium fluoride Product distribution; Mechanism; 1.)-78 deg C, 2.)carefull heating; | A 12.1% B 55.9% C 3.4% D 4.1% E 12.1% F 9.2% G n/a |

-
-
75-09-2
dichloromethane

-
-
7664-41-7
ammonia

-
A
-
74-90-8
hydrogen cyanide

-
B
-
593-53-3
Methyl fluoride

-
C
-
75-10-5
Difluoromethane

-
D
-
75-45-6
Chlorodifluoromethane

-
E
-
124-38-9
carbon dioxide

| Conditions | Yield |
|---|---|
| With catalyst: Pt/C In neat (no solvent) steady-state flow reaction over Pt/C catalyst at 769 K; | A 53.4% B 0.3% C 0.5% D 7.6% E 0.5% |

-
-
666-92-2
Ethyl pentafluoro-1-propenyl ether

-
A
-
593-53-3
Methyl fluoride

-
B
-
667-49-2
2,3,3-trifluoroacryloyl fluoride

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) | A n/a B 47% |
-
-
93636-91-0
2-(N-perfluoro-tert-butyl-N-fluorocarbonylamino)tetrafluoropropenyltrimethoxyphosphorane

-
A
-
593-53-3
Methyl fluoride

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 0.5h; | A n/a B 45% |
-
-
754-28-9
1,2-dichloro-1,1,2-trifluoro-1-methoxyethane

-
A
-
74-87-3
methylene chloride

-
B
-
593-53-3
Methyl fluoride

-
C
-
354-27-8
chlorodifluoroacetyl fluoride

-
D
-
1514-87-0
metyhyl chlorodifluoroacetate

| Conditions | Yield |
|---|---|
| With aluminum(III) fluoride at 200℃; for 1h; Product distribution; Further Variations:; Reagents; | A n/a B n/a C n/a D 43% |

-
-
67-56-1
methanol

-
-
1481-55-6
N-ethyl-N-(trifluoromethyl)ethanamine

-
A
-
593-53-3
Methyl fluoride

-
B
-
115-10-6
Dimethyl ether

-
C
-
4652-44-2
N-(methoxycarbonyl)diethylamine

-
D
-
616-38-6
carbonic acid dimethyl ester

| Conditions | Yield |
|---|---|
| for 0.25h; Heating; | A 40% B 12% C 10% D 37% |

-
-
67-56-1
methanol

-
A
-
593-53-3
Methyl fluoride

-
B
-
115-10-6
Dimethyl ether

-
C
-
4652-44-2
N-(methoxycarbonyl)diethylamine

-
D
-
616-38-6
carbonic acid dimethyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N-(trifluoromethyl)ethanamine for 0.25h; Heating; | A 40% B 12% C 10% D 37% |

-
-
360-53-2
1,3,3,3-tetrafluoro-1-methoxy-2-trifluoromethyl-propene

-
A
-
593-53-3
Methyl fluoride

-
B
-
684-36-6
perfluoromethacryloyl fluoride

-
C
-
684-22-0
bis(trifluoromethyl)ketene

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) Ambient temperature; | A n/a B 37% C 35% |

-
-
59736-15-1
1,3,4,5,5,5-hexafluoro-1,3,4-trimethoxy-2-trifluoromethyl-pent-1-ene

-
A
-
593-53-3
Methyl fluoride

-
B
-
77946-93-1
(Z)-1,1,1,5-Tetrafluoro-5-methoxy-4-trifluoromethyl-pent-4-ene-2,3-dione

| Conditions | Yield |
|---|---|
| antimony pentafluoride In neat (no solvent) Ambient temperature; | A n/a B 37% |

| Conditions | Yield |
|---|---|
| With potassium fluoride |
-
-
593-53-3
Methyl fluoride

-
-
2991-77-7
Trimethylamin-phosphor(V)-fluorid

-
-
558-32-7
tetramethylammonium hexafluorophosphate

| Conditions | Yield |
|---|---|
| at 200℃; under 23560 Torr; for 48h; | 98.2% |

| Conditions | Yield |
|---|---|
| With silicon tetrafluoride at 100℃; for 48h; | 96.4% |

| Conditions | Yield |
|---|---|
| With air; chlorine at 21.9℃; under 700 Torr; Mechanism; Rate constant; Irradiation; | A 87% B 11% |

| Conditions | Yield |
|---|---|
| under 23560 Torr; 1.) 152 deg C, 70 h, 2.) 171 deg C, 62 h; | 84.1% |

| Conditions | Yield |
|---|---|
| With sulfur dioxide; antimony pentafluoride for 0.25h; Ambient temperature; NMR study of products; | 82% |

| Conditions | Yield |
|---|---|
| at 280℃; for 96h; | 76% |

| Conditions | Yield |
|---|---|
| With sulfur dioxide at -196 - -78℃; for 1h; | 76% |

-
-
593-53-3
Methyl fluoride

-
-
106-38-7
para-bromotoluene

-
A
-
583-70-0
1-bromo-2,4-dimethylbenzene

-
B
-
583-71-1
4-bromo-o-xylene

| Conditions | Yield |
|---|---|
| With oxygen at 40℃; under 720 Torr; Mechanism; Irradiation; | A 72% B 28% |


| Conditions | Yield |
|---|---|
| With antimony pentafluoride at 25 - 50℃; for 16h; | 67% |
-
-
593-53-3
Methyl fluoride

-
-
432-04-2
tris(trifluoromethyl)phosphine

-
-
7783-70-2
antimony pentafluoride

-
-
1165950-49-1
[tris(trifluoromethyl)methyl phosphonium][Sb2F11]

| Conditions | Yield |
|---|---|
| In hydrogen fluoride HF (liquid); High Pressure; (N2); addn. of HF, phosphine deriv. and CF4 to antimony compd., warming to 40°C; concg. at -78°C, warming to 40°C, crystn. by cooling to 25°C, NMR; | 66% |

FLUOROMETHANE Chemical Properties
IUPAC Name: Fluoromethane
The MF of Fluoromethane (CAS NO.593-53-3) is CH3F.
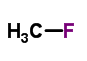
The MW of Fluoromethane (CAS NO.593-53-3) is 34.03.
Synonyms of Fluoromethane (CAS NO.593-53-3): Fluoromethane ; Methane, fluoro- ; Monofluoromethane
Product Categories: refrigerants
Appearance: colourless gas with an ether-like odour
Stability: Stable. Extremely flammable. Incompatible with strong oxidizing agents
Index of Refraction: 1.212
EINECS: 209-796-6
Density: 0.673 g/ml
Melting Point: -115 °C
Boiling Point: -79 °C
FLUOROMETHANE Uses
Fluoromethane (CAS NO.593-53-3) is used as chemical reagents, organic intermediates, fine chemicals, pharmaceutical research and development.
FLUOROMETHANE Production
Halogenated aliphatic compounds, such as METHYL FLUORIDE, are moderately or very reactive. Reactivity generally decreases with increased degree of substitution of halogen for hydrogen atoms. Low molecular weight haloalkanes are highly flammable and can react with some metals to form dangerous products. Materials in this group are incompatible with strong oxidizing and reducing agents. Also, they are incompatible with many amines, nitrides, azo/diazo compounds, alkali metals, and epoxides. The prolonged mixing of halogenated solvents with metallic or other azides may cause the slow formation of explosive azides, for example methylene chloride and sodium azide.
FLUOROMETHANE Consensus Reports
Reported in EPA TSCA Inventory.
FLUOROMETHANE Safety Profile
Narcotic in high concentrations. Acts as a simple asphyxiant. Burns with evolution of hydrogen fluoride. The flame is about as colorless as that of alcohol. When heated to decomposition it emits toxic fumes of F−.Safety information of Fluoromethane (CAS NO.593-53-3):
Hazard Codes  F,F+
F,F+
Risk Statements
12 Extremely Flammable
Safety Statements
16 Keep away from sources of ignition - No smoking
33 Take precautionary measures against static discharges
RIDADR 2454
Hazard Note Flammable
HazardClass 2.1
FLUOROMETHANE Standards and Recommendations
OSHA PEL: TWA 2.5 mg(F)/m3
ACGIH TLV: TWA 2.5 mg(F)/m3; BEI: 3 mg/g creatinine of fluorides in urine prior to shift; 10 mg/g creatinine of fluorides in urine at end of shift.
NIOSH REL: (Fluorides, Inorganic) TWA 2.5 mg(F)/m3
FLUOROMETHANE Specification
It is a colorless flammable gas which is heavier than air. METHYL FLUORIDE has an agreeable ether-like odor. METHYL FLUORIDE is narcotic in high concentrations. METHYL FLUORIDE burns with evolution of hydrogen fluoride. The flame is colorless, similar to alcohol. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View