-
Name
Glycine hydrochloride
- EINECS 227-841-8
- CAS No. 6000-43-7
- Article Data26
- CAS DataBase
- Density 1.27g/cm3
- Solubility Slightly soluble in water
- Melting Point 176-180 °C(lit.)
- Formula C2H6ClNO2
- Boiling Point 240.9 °C at 760 mmHg
- Molecular Weight 111.528
- Flash Point 99.5 °C
- Transport Information UN 1789 8/PG 3
- Appearance White crystalline powder
- Safety 24/25
- Risk Codes 22
-
Molecular Structure
- Hazard Symbols
- Synonyms Glycine,hydrochloride (8CI,9CI);Aminoacetic acid hydrochloride;Glycocollhydrochloride;
- PSA 63.32000
- LogP 0.53200
Synthetic route
-
-
70288-73-2
methoxycarbonylamino acetic acid methyl ester

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; formic acid In water at 125℃; for 24h; | 98% |
-
-
74743-61-6
(3R)-3-<(1S,5R)-3-benzyl-7-oxo-4-oxa-2,6-diazabicyclo<3.2.0>hept-2-en-6-yl>-3,4,4-trimethylthietan-2-one

-
A
-
103-82-2
phenylacetic acid

-
B
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
C
-
88168-76-7
(2-RS)-2-amino-3-mercapto-2,3-dimethylbutanoic acid hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 15h; Heating; | A 96% B n/a C n/a |

-
-
116963-47-4
Ethyl N-(trichloroacetyl)-2-aminoacetate

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol Ambient temperature; | 95% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 60℃; for 8h; | 91% |
| With hydrogenchloride at 50℃; under 11400.9 Torr; Equilibrium constant; Thermodynamic data; Kinetics; various temperatures, protonation; | |
| With hydrogenchloride In water |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(ll) chloride In water at 20 - 25℃; for 16h; | 61% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
15166-50-4
N-(Trichloroacetyl)glycine

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride Heating; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; potassium hydroxide 1.) methanol, 2 h, r. t.; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water |
-
-
1172-18-5, 36105-20-1
flurazepam dihydrochloride

-
A
-
6000-43-7
2-aminoethanoic acid hydrochloride

| Conditions | Yield |
|---|---|
| Product distribution; |
-
-
7647-01-0
hydrogenchloride

-
-
540-61-4
2-aminoacetonitrile

-
B
-
6000-43-7
2-aminoethanoic acid hydrochloride


-
-
6000-43-7
2-aminoethanoic acid hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| at 100℃; im geschlossenen Rohr; |

-
-
1186048-83-8
desmocyclopeptide

-
A
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
B
-
17585-69-2, 27172-85-6, 28069-46-7, 74496-49-4, 79541-99-4
L-phenylalanine hydrochloride

-
C
-
6003-05-0, 16428-74-3, 25616-13-1, 26570-06-9
L-alanine hydrochloride

-
D
-
17694-98-3
L-isoleucine hydrochloride

-
E
-
7776-34-3
L-proline hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 110℃; for 24h; |

-
-
1334715-68-2
versicoloritide B

-
A
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
B
-
17585-69-2, 27172-85-6, 28069-46-7, 74496-49-4, 79541-99-4
L-phenylalanine hydrochloride

-
C
-
7776-34-3
L-proline hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water at 110℃; for 20h; |

-
-
71-23-8
propan-1-ol

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
13049-01-9
glycine 1-propyl ester hydrochloride

| Conditions | Yield |
|---|---|
| With thionyl chloride at 20 - 70℃; for 15h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd; 2-aminoethanoic acid hydrochloride With trimethyl phosphite at 30 - 40℃; for 1h; Stage #2: With phosphorus trichloride at 10℃; for 24h; | 96% |
-
-
112-53-8
1-dodecyl alcohol

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
16194-11-9
dodecyloxycarbonylmethylammonium chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 110 - 120℃; for 8h; | 95% |
| With hydrogenchloride at 110 - 120℃; for 8h; | 91% |
-
-
14283-99-9, 36186-03-5
bis(N-salicylaldimino)nickel(II)

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
A
-
75214-75-4
bis(N-salicylideneglycinato)Ni(II)

| Conditions | Yield |
|---|---|
| In methanol stirring the soln. for 30 min between room temp. and 60°C, pptn.; filtration, washing with water, recrystn. from acetone; | A 90% B n/a |


| Conditions | Yield |
|---|---|
| With thionyl chloride at -20℃; for 2h; Inert atmosphere; Reflux; | 90% |
-
-
64-17-5
ethanol

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
623-33-6
glycine ethyl ester hydrochloride

| Conditions | Yield |
|---|---|
| With thionyl chloride at -20℃; for 2h; Reflux; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Hydroxy-4-methoxybenzaldehyde; 2-aminoethanoic acid hydrochloride With sodium hydroxide In water at 20℃; for 3h; pH=9; Stage #2: With sodium tetrahydroborate; sodium hydroxide In water for 1h; Cooling with ice; | 90% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetone for 10h; Reagent/catalyst; Solvent; Reflux; | 90% |
-
-
112-30-1
1-Decanol

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
39540-28-8
decyloxycarbonylmethylammonium chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 120 - 130℃; | 89% |
| With hydrogenchloride at 110 - 120℃; for 8h; | 89% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 110 - 120℃; for 8h; | 86% |

| Conditions | Yield |
|---|---|
| With phosphotungstic acid 44-hydrate; alloxan monohydrate In N,N-dimethyl-formamide at 110℃; | 86% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 110 - 120℃; for 8h; | 85% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 37℃; for 72h; pH=10.5; Polyethylene glycol; | 85% |
-
-
474-25-9
chenodeoxycholic acid

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
70779-05-4
ethyl glycochenodeoxycholate

| Conditions | Yield |
|---|---|
| With triethylamine In acetone for 10h; Reflux; | 85% |
-
-
50-00-0
formaldehyd

-
-
3011-82-3
oxyde de dioctyl phosphine

-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
1401662-39-2
N,N-bis(methylenedioctylphosphoryl)aminoethanoic acid

| Conditions | Yield |
|---|---|
| In acetonitrile for 3h; Reflux; | 84% |


| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; for 12h; | 80% |
-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
646517-54-6
(+/-)-(5-methyl-5-hexyl-2-oxothiophen-4-yloxy)acetic acid

-
-
646517-57-9
(+/-)-(5-methyl-5-hexyl-2-oxothiophen-4-yloxy)acetyl methyl glycinate

| Conditions | Yield |
|---|---|
| With tris(1,3-dihydro-2-oxobenzoxazolin-3-yl) phosphine oxide; triethylamine In acetonitrile at 20℃; for 0.333333h; | 80% |
-
-
6000-43-7
2-aminoethanoic acid hydrochloride

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 10 - 15℃; for 1h; Stage #2: 2-aminoethanoic acid hydrochloride at 80℃; for 4h; Stage #3: With hexafluorophosphoric acid In water at 0℃; | 78% |
Glycine hydrochloride Chemical Properties
Molecular Structure of 2-Aminoacetic acid hydrochloride (CAS NO.6000-43-7):
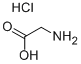
IUPAC Name: 2-Aminoacetic acid hydrochloride
CAS: 6000-43-7
Formula: C2H6ClNO2
Molecular Weight: 111.53
EINECS: 227-841-8
H bond acceptors: 3
H bond donors: 3
Freely Rotating Bonds: 2
Polar Surface Area: 29.54 Å2
Flash Point: 99.5 °C
Storage temp: 2-8°C
Water Solubility: Slightly soluble
Sensitive: Hygroscopic
Merck: 14,4491
BRN: 3909617
Melting Point: 176-180 °C(lit.)
Enthalpy of Vaporization: 52.63 kJ/mol
Boiling Point: 240.9 °C at 760 mmHg
Vapour Pressure: 0.0123 mmHg at 25°C
InChI
InChI=1/C2H5NO2.ClH/c3-1-2(4)5;/h1,3H2,(H,4,5);1H
Smiles
C(CN)(O)=O.Cl
Product Categories: alpha-amino acids; amino acids; biochemistry; omega-aminocarboxylic acids; omega-functional alkanols, carboxylic acids, amines & halides; alphabetic; amino acids, peptides and proteins; amino acidsanalytical standards; g;life sciences standards
Glycine hydrochloride Toxicity Data With Reference
| 1. | orl-rat LD50:3340 mg/kg | JPMSAE Journal of Pharmaceutical Sciences. 62 (1973),49. |
Glycine hydrochloride Consensus Reports
Reported in EPA TSCA Inventory.
Glycine hydrochloride Safety Profile
Moderately toxic by ingestion. When heated to decomposition it emits very toxic fumes of Cl− and NOx.
Safety Statements about 2-Aminoacetic acid hydrochloride (CAS NO.6000-43-7):
S24/25: Avoid contact with skin and eyes.
F10: Keep under argon.
RIDADR: UN 1789 8/PG 3
WGK Germany: 2
RTECS: MC0560000
Glycine hydrochloride Specification
The chemical synonyms of 2-Aminoacetic acid hydrochloride (CAS NO.6000-43-7) are Aminoethanoic acid hydrochloride ; Aminoacetic acid hydrochloride ; Glycocoll hydrochloride ; Glycine hcl ; Glycine-hcl buffer ; Glycine hydrochloride ; Glycine hydrochloride solution ; Aminoaceticacid.hcl=h-gly-oh.hcl . It shoud avoid breathing dust, vapor, mist, or gas and should be stored in a cool, dry place. Ingestion of large amounts may cause gastrointestinal irritation. Low hazard for usual industrial handling. It can be used to improve the bitter taste of beer, but also can be used in behalf of salts, nutritional supplements, flavor enhancer.
Related Products
- Glycine
- Glycine 1-methylethyl ester
- Glycine ethyl ester hydrochloride
- Glycine hydrochloride
- Glycine isopropyl ester hydrochloride
- Glycine lauryl ester hydrochloride
- Glycine Methyl ester Hydrochloride
- GLYCINE NITROGEN MUSTARD
- Glycine sodium salt
- Glycine sodium salt carbonate
- 6000-44-8
- 6000-50-6
- 600-05-5
- 6000-74-4
- 60010-03-9
- 60010-74-4
- 60010-78-8
- 60011-16-7
- 60011-55-4
- 600136-09-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View