-
Name
Itaconic acid
- EINECS 202-599-6
- CAS No. 97-65-4
- Article Data84
- CAS DataBase
- Density 1.366 g/cm3
- Solubility
- Melting Point 165-168 °C(lit.)
- Formula C5H6O4
- Boiling Point 381.4 °C at 760 mmHg
- Molecular Weight 130.1
- Flash Point 198.7 °C
- Transport Information
- Appearance white crystalline powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Succinic acid, methylene-;disodium 2-methylidenebutanedioate;Disodium methylenesuccinate;methylenebutanedioic acid;Butanedioic acid, methylene-;Succinic acid, methylene- (8CI);1/C5H6O4/c1-3(5(8)9)2-4(6)7/h1-2H2,(H,6,7)(H,8,9;butanedioic acid, 2-methylene-;sodium 2-methylidenebutanedioate;Methylenebutanedioic acid, disodium salt;2-Propene-1,2-dicarboxylic acid;Butanedioic acid,methylene-;
- PSA 74.60000
- LogP 0.10190
Synthetic route
-
-
77-92-9
citric acid

-
A
-
79-41-4
poly(methacrylic acid)

-
B
-
498-23-7
citraconic acid

-
C
-
498-24-8
Mesaconic acid

-
D
-
97-65-4
2-methylenesuccinic acid

| Conditions | Yield |
|---|---|
| With 4-methoxy-phenol; triphenylphosphine In water at 225℃; under 20686.5 Torr; for 1.5h; Temperature; Glovebox; Inert atmosphere; | A 24.1% B n/a C n/a D n/a |
| With sodium hydroxide at 250℃; for 0.25h; Reagent/catalyst; |

-
-
50-00-0
formaldehyd

-
-
922-84-9
ethane-1,1,2-tricarboxylic acid

-
A
-
97-65-4
2-methylenesuccinic acid

-
B
-
498-89-5
paraconic acid

| Conditions | Yield |
|---|---|
| With acetic anhydride; acetic acid |


| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With water at 120 - 160℃; | |
| With dipotassium peroxodisulfate; water; mercury dichloride | |
| With sodium hydroxide In water at 190℃; under 112511 Torr; for 0.133333h; Temperature; | 19.22 %Spectr. |

| Conditions | Yield |
|---|---|
| With aspergillus terreus | |
| With aspergillus terreus |
-
-
50-99-7, 59-23-4, 921-60-8, 1949-88-8, 1990-29-0, 2152-76-3, 2595-97-3, 2595-98-4, 3458-28-4, 4205-23-6, 5934-56-5, 5978-95-0, 5987-68-8, 6027-89-0, 6038-51-3, 7635-11-2, 10030-80-5, 15572-79-9, 19163-87-2, 23567-25-1, 26566-61-0, 30077-17-9, 31103-86-3, 39665-52-6, 40866-07-7, 58367-01-4, 58407-05-9, 58407-06-0, 83198-69-0, 83198-70-3, 83198-71-4, 93780-23-5, 145920-48-5
glucose

-
-
97-65-4
2-methylenesuccinic acid

| Conditions | Yield |
|---|---|
| With aspergillus terreus Bei der oxydativen Vergaerung; | |
| With aspergillus itaconicus Bei der oxydativen Vergaerung; |

| Conditions | Yield |
|---|---|
| bei der Destillation; | |
| bei der Destillation ensteht zunaechst in Form ihres Anhydrids; | |
| With water at 180℃; |
-
-
499-12-7
1 propene 1,2,3 tricarboxylic acid

-
A
-
97-65-4
2-methylenesuccinic acid

-
B
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

| Conditions | Yield |
|---|---|
| Erhitzen ueber die Schmelzpunkt; |

| Conditions | Yield |
|---|---|
| With aspergillus terreus | |
| With immune responsive gene 1 In aq. buffer at 30℃; for 1h; pH=7.1; |

| Conditions | Yield |
|---|---|
| With water; calcium carbonate at 140℃; |
-
-
854654-03-8
3-methoxy-propane-1,2,2-tricarboxylic acid triethyl ester

-
-
97-65-4
2-methylenesuccinic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With water at 120 - 160℃; | |
| With water at 150℃; |

| Conditions | Yield |
|---|---|
| With water Kinetik der Bildung; |
-
-
60-29-7
diethyl ether

-
-
727413-03-8
(3-ethoxycarbonyl-4,5-dioxo-tetrahydro-[3]furyl)-acetic acid ethyl ester

-
-
298-14-6
potassium hydrogencarbonate

-
A
-
97-65-4
2-methylenesuccinic acid

-
B
-
144-62-7
oxalic acid

-
-
50-00-0
formaldehyd

-
-
922-84-9
ethane-1,1,2-tricarboxylic acid

-
-
108-24-7
acetic anhydride

-
-
64-19-7
acetic acid

-
A
-
97-65-4
2-methylenesuccinic acid

-
B
-
498-89-5
paraconic acid


| Conditions | Yield |
|---|---|
| With acetic acid |

| Conditions | Yield |
|---|---|
| at 170 - 280℃; unter vermindertem Druck; | |
| With water at 280 - 300℃; under 30 - 40 Torr; |

| Conditions | Yield |
|---|---|
| bei der Destillation; | |
| bei der Destillation ensteht zunaechst in Form ihres Anhydrids; | |
| With water at 160℃; | |
| With sulfuric acid at 160℃; | |
| durch rasche trockne Destillation und unter Vermeidung von Ueberhitzung und Kochen des entstandenen Itaconsaeureanhydrids mit Wasser; |
-
-
151-50-8
potassium cyanide

-
-
6127-92-0
E-Ethyl β-chlorocrotonate

-
A
-
97-65-4
2-methylenesuccinic acid

-
B
-
99-14-9
tricarallylic acid

| Conditions | Yield |
|---|---|
| und nachfolgender Verseifung mit Kali; |


| Conditions | Yield |
|---|---|
| With aspergillus Bei der oxydativen Vergaerung; | |
| With aspergillus terreus Bei der oxydativen Vergaerung; | |
| With aspergillus terreus | |
| With aspergillus terreus |
-
-
34105-39-0
N-phenylitaconimide

-
-
97-65-4
2-methylenesuccinic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
77-92-9
citric acid

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
498-24-8
Mesaconic acid

-
C
-
499-12-7
1 propene 1,2,3 tricarboxylic acid

-
D
-
97-65-4
2-methylenesuccinic acid

-
E
-
616-02-4
citraconic acid anhydride

-
F
-
2170-03-8
itaconic acid anhydride

| Conditions | Yield |
|---|---|
| With Sn/Pb solder at 150 - 210℃; |


| Conditions | Yield |
|---|---|
| at 150℃; beim Erhitzen im geschlossenen Rohr; |
-
-
39629-86-2
5-oxo-1-phenyl-3-pyrrolidinecarboxylic acid

-
A
-
3120-04-5
3-methyl-1-phenylpyrrole-2,5-dione

-
B
-
498-23-7
citraconic acid

-
C
-
97-65-4
2-methylenesuccinic acid

| Conditions | Yield |
|---|---|
| at 260℃; |

-
-
97-65-4
2-methylenesuccinic acid

-
-
498-21-5, 636-60-2
2-methylbutanedioic acid

| Conditions | Yield |
|---|---|
| With hydrogen; (-)-(η5-2-menthyl-4,7-dimethylindenyl)Rh(dppe) In methanol at 60 - 70℃; under 10500.8 Torr; for 4h; Product distribution; Further Variations:; Catalysts; | 100% |
| With formic acid; triethylamine; chloro(1,5-cyclooctadiene)rhodium(I) dimer; (R,R)-methyldiopium iodide In tetrahydrofuran at 40℃; for 9h; | 100% |
| With sulfuric acid; palladium In water at 20℃; under 760.051 Torr; for 1h; Temperature; Pressure; Reagent/catalyst; Electrochemical reaction; | 98% |

| Conditions | Yield |
|---|---|
| With trimethylsilylethoxyacetylene In dichloromethane at 40℃; for 5h; | 100% |
| With acetic anhydride at 80℃; for 4h; | 92% |
| With hydrogenchloride; acetic anhydride at 40℃; for 1h; | 92% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
100-46-9
benzylamine

-
-
5733-86-8
1-benzyl-5-oxopyrrolidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| at 130℃; Inert atmosphere; | 100% |
| 86% | |
| at 130℃; for 2.5h; Inert atmosphere; | 82.9% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
63995-70-0
trisodium tris(3-sulfophenyl)phosphine

-
-
115524-89-5
C23H18O13PS3(3-)*3Na(1+)

| Conditions | Yield |
|---|---|
| In water | 100% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
63995-70-0
trisodium tris(3-sulfophenyl)phosphine

-
-
115524-90-8, 122865-77-4
C23H17(2)HO13PS3(3-)*3Na(1+)

| Conditions | Yield |
|---|---|
| With water-d2 | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen; [(R)-BINAP]RuBr2 at 50℃; under 2250.2 Torr; | 100% |
| With hydrogen; [(R)-(+)-binap](1,5-cyclooctadiene)rhodium(I) perchlorate In tetrahydrofuran; methanol at 20℃; under 13680 Torr; for 2h; | 95% |
| With hydrogen; In ethanol at 20℃; under 1 Torr; Yield given; |

| Conditions | Yield |
|---|---|
| With hydrogen; triethylamine; Ru2Cl4((R)-(+)-2,2'-bis(diphenylphosphino)-1,1'-binaphthyl)2 In tetrahydrofuran; toluene at 35℃; under 2206.5 Torr; for 24h; op = 88percent, var. cat.; RuHCl*<(+)-BINAP>2; Title compound not separated from byproducts; | A n/a B 100% |
| With hydrogen; triethylamine; chloro(1,5-cyclooctadiene)rhodium(I) dimer In ethanol; benzene for 24h; Ambient temperature; Title compound not separated from byproducts; | A n/a B 100% |
| With formic acid; triethylamine; chloro(1,5-cyclooctadiene)rhodium(I) dimer In dimethyl sulfoxide at 27℃; for 20h; Title compound not separated from byproducts; | A 89% B 11% |

| Conditions | Yield |
|---|---|
| With BPPM-Rh; hydrogen; triethylamine In methanol at 20℃; under 15200 Torr; for 20h; | 100% |
| With [(R)-BINAP]RuBr2; hydrogen In tetrahydrofuran at 50℃; under 2280 Torr; for 24h; | 100% |
| With BPPM-Rh; hydrogen; triethylamine In methanol at 20℃; under 15200 Torr; for 20h; Mechanism; Product distribution; other β-methylene acid, other reaction time and in the presence of triethylamine in some cases; | 100% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
6298-96-0, 22038-86-4, 35600-82-9, 41851-59-6
(R)-1-(4-methoxyphenyl)ethylamine

-
-
1319738-32-3
(1'R,3R/S)-1-(1'-(4-methoxyphenyl)ethyl)-5-oxo-3-pyrrolidine carboxylic acid

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 80 - 120℃; for 5h; | 100% |
| In 1-methyl-pyrrolidin-2-one at 80 - 120℃; for 5h; | 100% |
| In 1-methyl-pyrrolidin-2-one at 80 - 120℃; for 5h; | 100% |
| In 1-methyl-pyrrolidin-2-one at 80 - 120℃; for 5h; | 100% |
| In 1-methyl-pyrrolidin-2-one at 80 - 120℃; for 5h; | 195 g |
-
-
97-65-4
2-methylenesuccinic acid

-
-
6298-96-0, 22038-86-4, 35600-82-9, 41851-59-6
(S)-1-(4-methoxyphenyl)ethylamine

-
-
1319738-33-4
(1'S,3R/S)-1-(1'-(4-methoxyphenyl)ethyl)-5-oxo-3-pyrrolidine carboxylic acid

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 80 - 120℃; for 5h; | 100% |
| With 1-methyl-pyrrolidin-2-one at 130℃; for 4h; | 89% |
| at 130℃; for 1h; | |
| at 130℃; for 1h; | 12.7 g |

| Conditions | Yield |
|---|---|
| With calcium In 5,5-dimethyl-1,3-cyclohexadiene at 120℃; under 750.075 Torr; for 1.5h; Reagent/catalyst; | 99.3% |
| Stage #1: 2-methylenesuccinic acid With acetic anhydride at 80℃; Stage #2: at 230℃; under 262.526 Torr; for 4h; | 94% |
| 4Cs(1+)*SiW12O40(4-) = Cs4{SiW12O40} at 170 - 200℃; under 50 - 100 Torr; for 5.5h; Product distribution / selectivity; | 92.5% |
-
-
67-56-1
methanol

-
-
97-65-4
2-methylenesuccinic acid

-
-
7338-27-4
4-methoxy-2-methylene-4-oxobutanoic acid

| Conditions | Yield |
|---|---|
| toluene-4-sulfonamide at 40℃; for 48h; | 99% |
| With toluene-4-sulfonic acid at 20℃; for 3h; Esterification; | 98% |
| With Amberlyst 15 H+ resin at 20℃; for 72h; | 97% |

| Conditions | Yield |
|---|---|
| in neutral medium, boiling; | 99% |
| in neutral medium, boiling; | 99% |
| in acidic medium, boiling, 1 h; | 83% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
10038-98-9
germaniumtetrachloride

-
-
114686-96-3
diethyl [(trichlorogermyl)methyl]succinate

| Conditions | Yield |
|---|---|
| With thionyl chloride; ethanol; hypophosphorous acid In ethanol mixt. of GeCl4 and H3PO2 (from NaH2PO2*7H2O and concd. HCl), boiled (14h, N2); addn. of itaconic acid at 0°C, extn. (room temp., CH2Cl2), ext. evapd. , residue dried (azeotropic method), iolated polymer boiled with SOCl2 (1 h), distn. with EtOH; excess SOCl2 distd. with ethanol (1 h); vac. distn.; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| With dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; silver carbonate at 80℃; for 8h; | 99% |
| With dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; silver carbonate In N,N-dimethyl-formamide at 120℃; Inert atmosphere; | 40% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
574743-24-1
C5H5(2)H3O4

| Conditions | Yield |
|---|---|
| With formic acid; C20H25N2O3Rh(1+)*O4S(2-); water-d2 at 50℃; for 24h; Inert atmosphere; sealed tube; | 99% |

| Conditions | Yield |
|---|---|
| With dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; silver carbonate at 80℃; for 8h; | 99% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 20℃; for 3h; Esterification; | 98% |
| With hydrogenchloride | |
| With acetyl chloride | |
| With acetyl chloride |

| Conditions | Yield |
|---|---|
| With potassium dioxotetrahydroxoosmate(VI); sodium hydrogencarbonate In water for 6h; | 98% |
| 96% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
67-63-0
isopropyl alcohol

-
-
53720-10-8
diisopropyl 2-methylidenebutanedioate

| Conditions | Yield |
|---|---|
| With sulfuric acid In benzene at 70℃; for 48h; | 98% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
107534-96-3
tebuconazole

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With hydrogenated D001 type strong acid cation exchange resin at 115℃; for 4h; Temperature; | 97.8% |
| With La3+SO42-/TiO2-SiO2 for 3.5h; Reagent/catalyst; Time; Concentration; | 95.02% |
| With sulfuric acid; benzene unter Entfernen des gebildeten Wassers; | |
| With sulfuric acid; toluene unter Entfernen des gebildeten Wassers; | |
| With sulfuric acid In cyclohexane at 115 - 130℃; Dean-Stark; |
-
-
97-65-4
2-methylenesuccinic acid

-
-
108-42-9
3-chloro-aniline

-
-
92847-41-1
1-(3-chlorophenyl)-5-oxopyrrolidine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| Heating; | 97.7% |
| at 100℃; | 95% |
| at 120℃; | 92% |

| Conditions | Yield |
|---|---|
| With La3+SO42-/TiO2-SiO2 for 1h; Reagent/catalyst; Time; Concentration; | 97.61% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
104-94-9
4-methoxy-aniline

-
-
56617-47-1
1-(4-methoxyphenyl)-2-oxopyrrolidine-4-carboxylic acid

| Conditions | Yield |
|---|---|
| In water for 3h; Heating; | 97% |
| Heating; | 94.6% |
| In melt at 130 - 140℃; for 0.5h; | 85% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
62-53-3
aniline

-
-
39629-86-2
5-oxo-1-phenyl-3-pyrrolidinecarboxylic acid

| Conditions | Yield |
|---|---|
| In water at 110℃; for 30h; Sealed tube; | 97% |
| Heating; | 93.3% |
| 90% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
591-27-5
m-Hydroxyaniline

-
-
91891-24-6
1-(3-hydroxyphenyl)-5-oxo-3-pyrrolidinecarboxylic acid

| Conditions | Yield |
|---|---|
| at 120 - 130℃; for 0.0833333h; | 97% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
3886-69-9
(R)-1-phenyl-ethyl-amine

-
-
915302-94-2
(3RS)-1-<(R)-1-phenylethyl>-5-oxo-3-pyrrolidinecarboxylic acid

| Conditions | Yield |
|---|---|
| at 160℃; for 4h; | 97% |
-
-
97-65-4
2-methylenesuccinic acid

-
-
13423-15-9
3-methyltetrahydrofuran

| Conditions | Yield |
|---|---|
| With ammonium hexafluorophosphate; Λ(+)-tris(pentane-2,5-dionato)ruthenium; hydrogen; toluene-4-sulfonic acid; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In 1,4-dioxane at 195℃; under 75007.5 Torr; for 18h; Inert atmosphere; | 97% |
| With indium(III) bromide; 1,1,3,3-Tetramethyldisiloxane In toluene at 60℃; for 17h; Inert atmosphere; |
Itaconic acid Chemical Properties
Structure of Itaconic acid (CAS NO.97-65-4):
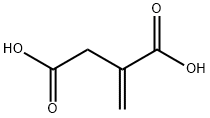
IUPAC Name: 2-methylidenebutanedioic acid
Empirical Formula: C5H6O4
Molecular Weight: 130.0987
EINECS: 202-599-6
Index of Refraction: 1.498
Molar Refractivity: 27.91 cm3
Molar Volume: 95.2 cm3
Polarizability: 11.06×10-24cm3
Surface Tension: 55.4 dyne/cm
Density: 1.366 g/cm3
Flash Point: 198.7 °C
Enthalpy of Vaporization: 69.14 kJ/mol
Melting Point: 165-168 °C(lit.)
Boiling Point: 381.4 °C at 760 mmHg
Vapour Pressure: 7.11E-07 mmHg at 25°C
Sensitive: Hygroscopic
Product Categories: Organic Chemicals
Canonical SMILES: C=C(CC(=O)O)C(=O)O
InChI: InChI=1S/C5H6O4/c1-3(5(8)9)2-4(6)7/h1-2H2,(H,6,7)(H,8,9)
InChIKey: LVHBHZANLOWSRM-UHFFFAOYSA-N
Itaconic acid Uses
Itaconic acid (CAS NO.97-65-4) is primarily used as a co-monomer in the production of styrene-butadiene-acrylonitrile and acrylate latexes with applications in the paper and architectural coating industry.
Itaconic acid Production
In nature, itaconic acid is being produced by Aspergillus terreus. Scientists are developing plants to produce itaconic acid. This may allow replacement of petrochemical products by plant based, thus more sustainable, raw material.
Itaconic acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
WGK Germany: 1
HS Code: 29171990
Itaconic acid Specification
Itaconic acid , its cas register number is 97-65-4. It also can be called 2-Propene-1,2-dicarboxylic acid ; AI3-16901 ;
Butanedioic acid, methylene- ; Methylenebutanedioic acid ; NSC 3357 ; Propylenedicarboxylic acid ; Succinic acid, methylene- . Itaconic acid (CAS NO.97-65-4) is a white crystalline powder that is soluble in water, and partially soluble in methanol, ethanol or acetone.
Related Products
- Itaconic acid
- Itaconic anhydride
- 976-56-7
- 97657-92-6
- 97659-27-3
- 97659-35-3
- 97659-36-4
- 97659-47-7
- 97659-67-1
- 97660-12-3
- 97660-18-9
- 97660-61-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View