-
Name
(2-Methoxyethyl)benzene
- EINECS 222-619-7
- CAS No. 3558-60-9
- Article Data57
- CAS DataBase
- Density 0.938 g/cm3
- Solubility 682mg/L at 25℃
- Melting Point
- Formula C9H12O
- Boiling Point 172.3 °C at 760 mmHg
- Molecular Weight 136.194
- Flash Point 52.5 °C
- Transport Information
- Appearance colourless liquid
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Ether,methyl phenethyl (6CI,7CI,8CI);1-Methoxy-2-phenylethane;2-Phenylethyl methylether;Kewda ether;Methyl 2-phenethyl ether;Methyl 2-phenylethyl ether;NSC 81229;Pandanol;b-Phenylethyl methyl ether;
- PSA 9.23000
- LogP 1.87550
Synthetic route

| Conditions | Yield |
|---|---|
| With triethanolamine; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; [Ni(2,2′:6′,2''-terpyridine)(pyridine)(CH3CN)2](PF6)2 In acetonitrile at 23 - 28℃; for 12h; Inert atmosphere; Sealed tube; Irradiation; | 96% |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid at 25℃; for 1h; | 95% |
-
-
60-12-8
2-phenylethanol

-
-
77891-02-2
methyl trimethylamine phosphate

-
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With potassium chloride; sodium sulfite at 85℃; for 4h; Temperature; | 95% |

| Conditions | Yield |
|---|---|
| With silver tetrafluoroborate; 1-(2-bromo-1,1-dimethoxyethyl)benzene at 25℃; for 4h; | 90% |
| (i) HgO, aq. HClO4, (ii) /BRN= 507487/; Multistep reaction; | |
| With tetrafluoroboric acid; HgBF4 | |
| at 138 - 145℃; under 8250.7 Torr; for 1.8h; Irradiation; | 38 % Turnov. |
| at 140℃; under 7500.6 Torr; for 2h; Irradiation; Yield given; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-phenylethanol With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 1h; Stage #2: methyl iodide In tetrahydrofuran; mineral oil at 0 - 20℃; for 72h; | 86% |
| With aluminum oxide; potassium fluoride In acetonitrile for 43h; | 66% |
| With potassium hydroxide In dimethyl sulfoxide at 20℃; for 2h; | 65% |

| Conditions | Yield |
|---|---|
| With ammonia; calcium In tetrahydrofuran at -33℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| With (PPh3)(CO)4MnC(O)CH3; phenylsilane In benzene-d6 for 2h; | 83% |
| With triethylsilane; trimethylsilyl trifluoromethanesulfonate; titanium tetrachloride In dichloromethane at 20℃; for 20h; | 40% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; zirconium (benzyldiethylammoniomethylphosphonate chloride) phosphate In ethanol; water; Petroleum ether at 42℃; for 8h; | 81.3% |
| With sodium hydride In diethyl ether for 20h; Heating; | 60% |
| With sodium hydroxide; tetra-(n-butyl)ammonium iodide |
-
-
60-12-8
2-phenylethanol

-
-
616-38-6
carbonic acid dimethyl ester

-
A
-
3558-60-9
1-methoxy-2-phenylethane

-
B
-
1796-66-3
methyl (2-phenyl)ethyl carbonate

| Conditions | Yield |
|---|---|
| With Amberlyst 15 for 12h; Reflux; | A 14% B 79% |

-
-
67-56-1
methanol

-
-
103-63-9
1-phenyl-2-bromoethane

-
A
-
100-42-5
styrene

-
B
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| In methanol at 95℃; under 5475.4 Torr; for 0.0283333h; Irradiation; | A 75% B 9% |


| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 95℃; under 5475.4 Torr; for 0.0283333h; Irradiation; | A 75% B 9% |

| Conditions | Yield |
|---|---|
| With (triphenylphosphine)gold(I) chloride; 1-(trifluoromethanesulfonyloxy)-1,2-benziodoxol-3(1H)-one In acetonitrile at 50℃; under 750.075 Torr; for 18h; Reagent/catalyst; Temperature; Solvent; Sealed tube; | 72% |

-
-
67-56-1
methanol

-
-
81609-30-5
2-phenylethyl phenyl telluride

-
A
-
100-42-5
styrene

-
B
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid at 20 - 25℃; for 1h; | A 14% B 67% |
| With 3-chloro-benzenecarboperoxoic acid at 20 - 25℃; for 1h; | A 14 % Chromat. B 67% |


| Conditions | Yield |
|---|---|
| With 5%-palladium/activated carbon; hydrogen; toluene-4-sulfonic acid In methanol at 130℃; under 22502.3 Torr; for 6h; Autoclave; Inert atmosphere; | 66.5% |

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide | 63% |

| Conditions | Yield |
|---|---|
| With terephthalonitrile In acetonitrile for 2h; UV-irradiation; | 35% |
| With N,N′-bis(2-ethylhexyl)-1,7-dicyanoperylene-3,4:9,10-bis(dicarboximide); thiophenol In dichloromethane at 30℃; for 42h; Irradiation; Inert atmosphere; regioselective reaction; | 32% |

| Conditions | Yield |
|---|---|
| at 150℃; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dimethyl sulfate | |
| With diazomethane; diethyl ether; boron trifluoride diethyl etherate |

-
-
107-30-2
chloromethyl methyl ether

-
-
6921-34-2
benzylmagnesium chloride

-
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
60-29-7
diethyl ether

-
-
591-51-5
phenyllithium

-
-
6482-24-2
2-Bromoethyl methyl ether

-
A
-
108-86-1
bromobenzene

-
B
-
3558-60-9
1-methoxy-2-phenylethane

-
C
-
71-43-2
benzene


| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
13057-17-5
bromethyl methyl ether

-
-
6921-34-2
benzylmagnesium chloride

-
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With hydrogen; nickel at 180℃; | |
| With carbon monoxide; hydrogen; dicobalt octacarbonyl at 180℃; under 124116 Torr; |
-
-
100-42-5
styrene

-
-
67-56-1
methanol

-
A
-
3558-60-9
1-methoxy-2-phenylethane

-
B
-
1083-56-3
1,4-diphenylbutane

-
C
-
4013-34-7
3-phenyl-2-oxabutane

-
D
-
32345-80-5, 4013-37-0
1,2-dimethoxyethylbenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide; borane-THF 1) THF, 2) THF, Pt-anode, 30 mF; Yield given. Multistep reaction. Further byproducts given. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
67-56-1
methanol

-
-
60-12-8
2-phenylethanol

-
A
-
100-42-5
styrene

-
B
-
3558-60-9
1-methoxy-2-phenylethane

-
C
-
4013-34-7
3-phenyl-2-oxabutane

-
D
-
100-52-7
benzaldehyde

| Conditions | Yield |
|---|---|
| With methane; oxygen at 37.5℃; Product distribution; Irradiation; different partial pressures of components; |

-
-
67-56-1
methanol

-
-
458-87-7
1-fluoro-2-phenylethane

-
A
-
100-42-5
styrene

-
B
-
538-86-3
benzyl methyl ether

-
C
-
3558-60-9
1-methoxy-2-phenylethane

-
D
-
4013-34-7
3-phenyl-2-oxabutane

-
E
-
100-52-7
benzaldehyde

| Conditions | Yield |
|---|---|
| With methane; oxygen at 37.5℃; Product distribution; Irradiation; different bulk gas and methanol partial pressures; |

-
-
60-12-8
2-phenylethanol

-
A
-
100-42-5
styrene

-
B
-
92-52-4
biphenyl

-
C
-
3558-60-9
1-methoxy-2-phenylethane

-
D
-
100-41-4
ethylbenzene

-
E
-
103-29-7
1,1'-(1,2-ethanediyl)bisbenzene

-
F
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| at 446.4 - 493.7℃; Product distribution; Kinetics; thermal elimination; |

-
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With hydrogen; water-d2; palladium on activated charcoal at 20℃; for 72h; | 88% |
-
-
3558-60-9
1-methoxy-2-phenylethane

-
-
29610-84-2
(1‐bromo‐2‐methoxyethyl)benzene

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) In tetrachloromethane for 2h; Heating / reflux; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-methoxy-2-phenylethane With thianthrene-5-oxide; trifluoromethylsulfonic anhydride In acetonitrile at -20 - 20℃; for 0.666667h; Inert atmosphere; Schlenk technique; Stage #2: acetophenone With palladium diacetate; cesium fluoride; XPhos In N,N-dimethyl-formamide; acetonitrile at 80℃; for 11h; Inert atmosphere; Schlenk technique; | 80% |
-
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With hydrogen; water-d2; palladium on activated charcoal at 110℃; under 760.051 Torr; for 24h; | 79% |

| Conditions | Yield |
|---|---|
| With phosphazene base-P4-tert-butyl In 1,4-dioxane; hexane at 130℃; for 24h; Sealed tube; Inert atmosphere; Molecular sieve; | 79% |
-
-
3558-60-9
1-methoxy-2-phenylethane

-
A
-
622-24-2
2-phenylethyl chloride

-
B
-
103-45-7
acetic acid phenethyl ester

| Conditions | Yield |
|---|---|
| With acetyl chloride; zinc In Petroleum ether at 28℃; for 4h; | A 78% B 20% |
-
-
3558-60-9
1-methoxy-2-phenylethane

-
-
75-36-5
acetyl chloride

-
A
-
622-24-2
2-phenylethyl chloride

-
B
-
103-45-7
acetic acid phenethyl ester

| Conditions | Yield |
|---|---|
| With zinc In Petroleum ether at 28℃; for 4h; | A 78% B 20% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; trimethylsilylazide; copper dichloride at 80℃; for 12h; | 68% |

| Conditions | Yield |
|---|---|
| With hydrogen; AIOTfbpy-Pd In 1,2-dichloro-ethane at 130℃; under 750.075 Torr; for 24h; Glovebox; Inert atmosphere; | 61% |
| With hydrogen; Al(OH)(2,2'-bipyridine-5,5'-dicarboxylic acid)0.81(PdCl2)0.48(OTf)0.38 In 1,2-dichloro-ethane at 130℃; under 750.075 Torr; for 24h; | 55% |
-
-
3558-60-9
1-methoxy-2-phenylethane

-
-
140-88-5
ethyl acrylate

-
A
-
1450816-66-6
(2E,2'E)-diethyl 3,3'-(2-(2-methoxyethyl)-1,3-phenylene)diacrylate

-
B
-
1450816-65-5
(E)-ethyl 3-(2-(2-methoxyethyl)phenyl)acrylate

| Conditions | Yield |
|---|---|
| With silver (II) carbonate; palladium diacetate; N-acetylglycine at 80℃; for 48h; Sealed tube; | A 25% B 60% |

-
-
3558-60-9
1-methoxy-2-phenylethane

-
-
62-53-3
aniline

-
A
-
31230-89-4
2-benzyl-2,3-dihydrobenzo[d]thiazole

-
B
-
6265-94-7
2-benzylbenzothiazole

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sulfur; potassium iodide In water; dimethyl sulfoxide at 130℃; for 6h; | A 15% B 52% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; trimethylsilylazide; copper(l) chloride at 80℃; for 16h; Sealed tube; regioselective reaction; | 51% |
-
-
3558-60-9
1-methoxy-2-phenylethane

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; water In dichloromethane at 45℃; for 6h; Inert atmosphere; | A 23% B 39% |

Methyl phenethyl ether Consensus Reports
Methyl phenethyl ether Specification
The 2-Phenylethyl methyl ether, with the CAS registry number 3558-60-9 and EINECS registry number 222-619-7, has the systematic name of (2-methoxyethyl)benzene. It is a kind of colourless liquid. And the molecular formula of this chemical is C9H12O.
The physical properties of 2-Phenylethyl methyl ether are as following: (1)ACD/LogP: 2.14; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.14; (4)ACD/LogD (pH 7.4): 2.14; (5)ACD/BCF (pH 5.5): 24.97; (6)ACD/BCF (pH 7.4): 24.97; (7)ACD/KOC (pH 5.5): 348.3; (8)ACD/KOC (pH 7.4): 348.3; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 9.23 Å2; (13)Index of Refraction: 1.493; (14)Molar Refractivity: 42.18 cm3; (15)Molar Volume: 145.1 cm3; (16)Polarizability: 16.72×10-24cm3; (17)Surface Tension: 31.3 dyne/cm; (18)Density: 0.938 g/cm3; (19)Flash Point: 52.5 °C; (20)Enthalpy of Vaporization: 39.19 kJ/mol; (21)Boiling Point: 172.3 °C at 760 mmHg; (22)Vapour Pressure: 1.79 mmHg at 25°C.
Preparation of 2-Phenylethyl methyl ether: This chemical can be prepared by iodomethane and 2-phenyl-ethanol. The reaction will need reagent Na. The reaction time is 2 hours with heating, and the yield is about 42%.
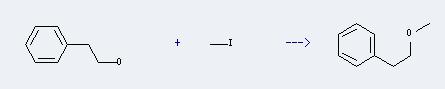
You should be cautious while dealing with this chemical. It is a kind of flammable chemcial, and it also irritates eyes, respiratory system and skin. In addition, if contact with water, it will liberate extremely flammable gases. Therefore, you had better take the following instructions: Wear suitable protective clothing, gloves and eye/face protection, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O(CCc1ccccc1)C
(2)InChI: InChI=1/C9H12O/c1-10-8-7-9-5-3-2-4-6-9/h2-6H,7-8H2,1H3
(3)InChIKey: CQLYXIUHVFRXLT-UHFFFAOYAS
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LD50 | skin | 3970mg/kg (3970mg/kg) | Food and Chemical Toxicology. Vol. 20, Pg. 807, 1982. | |
| rat | LD50 | oral | 4100mg/kg (4100mg/kg) | Food and Chemical Toxicology. Vol. 20, Pg. 807, 1982. |
Related Products
- Methyl (((methoxymethylphosphinothioyl)thio)acetyl)methylcarbamate
- Methyl (2E)-3-(4-methylphenyl)propenoate
- Methyl (2E)-3-cyclohexylprop-2-enoate
- Methyl (2R)-2-[(tert-butoxycarbonyl)amino]-3-iodopropanoate
- Methyl (2R)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2R)-2-amino-2-cyclohexylethanoate hydrochloride
- Methyl (2R)-2-bromo-2-(2-chlorophenyl)acetate
- Methyl (2R,3S)-3-(4-methoxyphenyl)-2-oxiranecarboxylate
- Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate
- Methyl (2Z)-2-methyl-3-phenylprop-2-enoate
- 3558-61-0
- 355-86-2
- 3558-69-8
- 35588-36-4
- 35588-53-5
- 35589-32-3
- 35590-37-5
- 35591-41-4
- 355-93-1
- 35597-43-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View