-
Name
N,N-Diethylformamide
- EINECS 210-533-2
- CAS No. 617-84-5
- Article Data144
- CAS DataBase
- Density 0.875 g/cm3
- Solubility miscible with ethanol, acetone and benzene. Soluble in alcohol and ether
- Melting Point 176-177 °C
- Formula C5H11NO
- Boiling Point 177.499 °C at 760 mmHg
- Molecular Weight 101.148
- Flash Point 69.109 °C
- Transport Information UN 1993 3/PG 3
- Appearance clear colorless to pale yellow liquid
- Safety 26-36/37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Diethylformamide;N,N-Diethylformamide;N-Formyldiethylamine;NSC 6242;
- PSA 20.31000
- LogP 1.12050
Synthetic route

| Conditions | Yield |
|---|---|
| With C21H24N2; phenylsilane In tetrahydrofuran at 20℃; under 750.075 - 2250.23 Torr; for 1.5h; Solvent; Concentration; Reagent/catalyst; Temperature; Time; Inert atmosphere; Glovebox; | 100% |
| With phenylsilane at 25℃; under 37503.8 Torr; for 12h; Pressure; Temperature; Autoclave; | 100% |
| With Zn(salen); phenylsilane; tetrabutylammomium bromide at 25℃; under 3750.38 Torr; for 15h; Reagent/catalyst; Autoclave; | 99% |

| Conditions | Yield |
|---|---|
| With potassium tetrachloroaurate(III); potassium carbonate In water; acetonitrile at 40℃; for 12h; | 98% |
| With sodium hydroxide In water at 25℃; for 6h; | 91% |
| With potassium iodide In water Ambient temperature; electrolysis; | 85% |
-
-
10545-36-5
N-<(trimethylsilyl)methyl>-N,N-diethylamine

-
-
617-84-5
N-formyldiethylamine

| Conditions | Yield |
|---|---|
| With copper(I) bromide In acetonitrile at 25℃; for 4h; chemoselective reaction; | 94% |

| Conditions | Yield |
|---|---|
| With Thiamine hydrochloride at 80℃; for 0.5h; | 90% |
| With sulfated polyborate In neat (no solvent) at 70℃; for 0.333333h; | 86% |
| With silica gel for 0.0236111h; microwave irradiation; | 80% |

-
-
617-84-5
N-formyldiethylamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; diethyl ether In diethyl ether at -78℃; for 0.5h; | 85% |

| Conditions | Yield |
|---|---|
| With Cu/Al2O3; dihydrogen peroxide In water at 25℃; for 24h; Green chemistry; | 83% |
-
-
27959-42-8
4-tert-butyl-2-(2-nitrophenylazo)phenol

-
A
-
615-15-6
2-Methyl-1H-benzimidazole

-
B
-
617-84-5
N-formyldiethylamine

-
C
-
3147-76-0
2-(5-tert-butyl-2-hydroxyphenyl)benzotriazole

-
D
-
88-74-4
2-nitro-aniline

| Conditions | Yield |
|---|---|
| With carbon monoxide; triethylamine In 1,2-dichloro-benzene at 200℃; under 60800 Torr; for 8h; Further byproducts given; | A n/a B n/a C 63% D 82% |

-
-
14873-98-4
bis(diethylamin)nickeldibromid

-
-
109-89-7
diethylamine

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
1187-03-7
N,N,N',N'-tetraethylurea

-
C
-
14288-05-2
N,N,N',N'-tetraethyloxamide

-
D
-
6274-12-0
diethylamine hydrobromide

-
E
-
13463-39-3, 71564-36-8
tetracarbonyl nickel

| Conditions | Yield |
|---|---|
| With carbon monoxide In tetrahydrofuran; diethyl ether reaction in THF/Et2O = 2/1 soln., room temp., under CO (1 bar); | A 7% B 0.5% C 80% D n/a E n/a |


| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; oxygen In chlorobenzene at 100℃; under 4500.45 Torr; for 4h; Autoclave; Green chemistry; | 80% |
| With Eosin Y In ethanol at 23℃; Irradiation; | 16% |
| With pyridine; copper(II) bis(trifluoromethanesulfonate) In acetonitrile at 130℃; under 7500.75 - 30003 Torr; Catalytic behavior; Reagent/catalyst; Pressure; Temperature; Autoclave; |

| Conditions | Yield |
|---|---|
| Ambient temperature; | 78% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.15h; | 78% |
-
-
18243-21-5
trimethylsilyl formate

-
-
6022-10-2
(diethylamino)triethylsilane

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
107-46-0
Hexamethyldisiloxane

-
C
-
2652-41-7
1,1,1-triethyl-3,3,3-trimethyl-disiloxane

-
D
-
994-49-0
hexaethyl disiloxane

| Conditions | Yield |
|---|---|
| With N-Methylformamide at 20℃; for 2h; | A 72.5% B 13.6% C n/a D 23.8% |
| With N-Methylformamide at 20℃; for 2h; Product distribution; Mechanism; various amides as catalysts, other temperature, other time; | A 72.5% B 13.6% C n/a D 23.8% |

-
-
18243-21-5
trimethylsilyl formate

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
107-46-0
Hexamethyldisiloxane

-
C
-
2652-41-7
1,1,1-triethyl-3,3,3-trimethyl-disiloxane

-
D
-
994-49-0
hexaethyl disiloxane

| Conditions | Yield |
|---|---|
| With N-Methylformamide; (diethylamino)triethylsilane at 20℃; for 2h; | A 72.5% B 13.6% C n/a D 23.8% |

-
-
6022-10-2
(diethylamino)triethylsilane

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
107-46-0
Hexamethyldisiloxane

-
C
-
2652-41-7
1,1,1-triethyl-3,3,3-trimethyl-disiloxane

-
D
-
994-49-0
hexaethyl disiloxane

| Conditions | Yield |
|---|---|
| With N-Methylformamide; trimethylsilyl formate at 20℃; for 2h; | A 72.5% B 13.6% C n/a D 23.8% |


| Conditions | Yield |
|---|---|
| With dioxo[bis(sulfato-κO)]molybdenum In ethanol for 5h; Reflux; Green chemistry; | 70% |
-
-
109-89-7
diethylamine

-
-
160663-31-0
2,2-Dichloro-3-oxo-butyraldehyde

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
513-88-2
1,1-Dichloroacetone

| Conditions | Yield |
|---|---|
| In tetrachloromethane for 12h; Ambient temperature; | A 68% B 60% |

-
-
201230-82-2
carbon monoxide

-
-
109-89-7
diethylamine

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
1187-03-7
N,N,N',N'-tetraethylurea

-
C
-
14288-05-2
N,N,N',N'-tetraethyloxamide

| Conditions | Yield |
|---|---|
| With (Et2NH)2CuBr; (Et2NH)2NiBr In tetrahydrofuran under 15001.2 Torr; for 16h; Ambient temperature; | A n/a B n/a C 66% |

-
-
201230-82-2
carbon monoxide

-
-
109-89-7
diethylamine

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
14288-05-2
N,N,N',N'-tetraethyloxamide

| Conditions | Yield |
|---|---|
| With (Et2NH)2CuBr; (Et2NH)2NiBr In tetrahydrofuran under 15001.2 Torr; for 16h; Ambient temperature; | A n/a B 66% |


| Conditions | Yield |
|---|---|
| With copper(l) iodide; N,N,N,N,-tetramethylethylenediamine; oxygen In dimethyl sulfoxide at 100℃; for 24h; Sealed tube; | A n/a B 66% |


| Conditions | Yield |
|---|---|
| With sodium ethanolate at 50℃; Riemer-Tiemann reaction; Inert atmosphere; | 62% |
| With potassium tert-butylate; water; benzene | |
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| In ethyl acetate for 1h; Ambient temperature; | 62% |
-
-
598-30-1
sec.-butyllithium

-
-
88-10-8
N,N-diethylcarbamyl chloride

-
A
-
617-84-5
N-formyldiethylamine

-
B
-
59002-07-2
2-methyl-butyric acid diethylamide

| Conditions | Yield |
|---|---|
| Stage #1: sec.-butyllithium With CuCN In tetrahydrofuran at -30℃; for 0.5h; Stage #2: N,N-diethylcarbamyl chloride In tetrahydrofuran at -30 - 0℃; for 2h; | A 11 % Chromat. B 62% |

-
-
13839-14-0
N,N-diethylthioformamide

-
-
617-84-5
N-formyldiethylamine

| Conditions | Yield |
|---|---|
| With triphenyltin(IV) hydroxide In benzene-d6 at 90℃; for 8h; | 60% |

| Conditions | Yield |
|---|---|
| With tetracarbonyl nickel In toluene at 180℃; for 24h; | 55% |
| With 1,3-bis-(2,6-diisopropylphenyl)-imidazol-2-ylidene In methanol at 0 - 130℃; under 22502.3 Torr; for 36h; Autoclave; | 12.1% |
| In tetrahydrofuran under 760 Torr; for 24h; Product distribution; Mechanism; Ambient temperature; other conditions: other catalyst, other times, addition of complexing agents, addition of LiBr; |
-
-
109-89-7
diethylamine


-
A
-
617-84-5
N-formyldiethylamine

-
B
-
953-21-9
(1E,2E)-N,3-diphenylprop-2-en-1-imine

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at 0℃; for 3h; | A 50% B n/a |

-
-
201230-82-2
carbon monoxide

-
-
74-86-2
acetylene

-
A
-
96-48-0
4-butanolide

-
B
-
617-84-5
N-formyldiethylamine

-
C
-
25883-23-2
tetraethyl-butynediamide

-
D
-
51590-64-8
N,N-diethylpropiolamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether under 750.06 Torr; Ambient temperature; | A 8.9 % Chromat. B 2.4 % Chromat. C 45% D 11% |


| Conditions | Yield |
|---|---|
| 12% |

| Conditions | Yield |
|---|---|
| durch folgendes Erwaermen; |

-
-
617-84-5
N-formyldiethylamine

-
-
40416-60-2
1,1-dichloromethyl-N,N-diethylamine

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane at 20℃; for 2h; Inert atmosphere; | 100% |
| With (pentachloroethyl)phosphorimidic trichloride In benzene |
-
-
617-84-5
N-formyldiethylamine

-
-
132868-67-8
(tert-butyldimethylsilyl)(phenyl)methanone

-
-
119701-95-0
N-(p-methoxybenzylidene)diphenylphosphinamide

| Conditions | Yield |
|---|---|
| Stage #1: N-formyldiethylamine; (tert-butyldimethylsilyl)(phenyl)methanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; Stage #2: N-(p-methoxybenzylidene)diphenylphosphinamide In tetrahydrofuran at -78℃; for 0.5h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 100% |

-
-
617-84-5
N-formyldiethylamine

-
-
119701-95-0
N-(p-methoxybenzylidene)diphenylphosphinamide

| Conditions | Yield |
|---|---|
| Stage #1: N-formyldiethylamine; tert-butyldimethylsilyl 4-fluorophenyl ketone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; Stage #2: N-(p-methoxybenzylidene)diphenylphosphinamide In tetrahydrofuran at -78℃; for 0.5h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 100% |

-
-
617-84-5
N-formyldiethylamine

-
-
119701-95-0
N-(p-methoxybenzylidene)diphenylphosphinamide

| Conditions | Yield |
|---|---|
| Stage #1: N-formyldiethylamine; C17H28OSi With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; Stage #2: N-(p-methoxybenzylidene)diphenylphosphinamide In tetrahydrofuran at -78℃; for 0.5h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 100% |

-
-
617-84-5
N-formyldiethylamine

-
-
119701-95-0
N-(p-methoxybenzylidene)diphenylphosphinamide

| Conditions | Yield |
|---|---|
| Stage #1: N-formyldiethylamine; C15H24OSi With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; Stage #2: N-(p-methoxybenzylidene)diphenylphosphinamide In tetrahydrofuran at -78℃; for 0.5h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 100% |

-
-
617-84-5
N-formyldiethylamine

-
-
141-97-9
ethyl acetoacetate

-
-
1416425-91-6
3-diethylcarbamoyloxybut-2-enoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(l) chloride In water at 20 - 70℃; for 0.75h; Green chemistry; stereoselective reaction; | 99% |
| With tert.-butylhydroperoxide; copper nanoparticles on black carbon In water; N,N-dimethyl-formamide at 80℃; for 5h; Inert atmosphere; | 80% |
| With tert.-butylhydroperoxide In water at 80℃; for 4h; Inert atmosphere; stereoselective reaction; | 70% |
| With tert.-butylhydroperoxide; copper(ll) bromide In water at 80℃; for 3h; stereoselective reaction; | 68% |
| With tert.-butylhydroperoxide; copper(II) oxide In water at 20℃; for 4h; Sonication; |
-
-
617-84-5
N-formyldiethylamine

-
-
118-93-4
o-hydroxyacetophenone

-
-
211449-24-0
diethylcarbamic acid 2-acetylphenyl ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(l) chloride In water at 20 - 70℃; for 0.75h; Green chemistry; | 99% |
| With tert.-butylhydroperoxide at 70℃; for 0.5h; | 95% |
| With tert.-butylhydroperoxide; copper diacetate In water at 80℃; for 3h; | 84% |
-
-
617-84-5
N-formyldiethylamine

-
-
94-02-0
ethyl 3-oxo-3-phenylpropionate

-
-
1416425-90-5
3-diethylcarbamoyloxy-3-phenylacrylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(l) chloride In water at 20 - 70℃; for 0.75h; Green chemistry; stereoselective reaction; | 99% |
| With tert.-butylhydroperoxide; copper(ll) bromide In water at 80℃; for 3h; stereoselective reaction; | 80% |
-
-
617-84-5
N-formyldiethylamine

-
-
105-45-3
acetoacetic acid methyl ester

-
-
1416425-92-7
3-diethylcarbamoyloxybut-2-enoic acid methyl ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(l) chloride In water at 20 - 70℃; for 0.5h; Green chemistry; stereoselective reaction; | 99% |
| With tert.-butylhydroperoxide; copper nanoparticles on black carbon In water; N,N-dimethyl-formamide at 80℃; for 5h; Inert atmosphere; | 84% |
| With tert.-butylhydroperoxide; copper(ll) bromide In water at 80℃; for 3h; stereoselective reaction; | 66% |
| With tert.-butylhydroperoxide; copper(II) oxide In water at 20℃; for 4h; Sonication; |
-
-
617-84-5
N-formyldiethylamine

-
-
86-52-2
1-Chloromethylnaphthalene

-
-
35693-45-9
1-<(N,N-diethylamino)methyl>naphthalene

| Conditions | Yield |
|---|---|
| With NHC-Pd(II)-Im; sodium hydroxide In water at 50℃; for 3h; Inert atmosphere; Schlenk technique; | 99% |
| With potassium hydroxide In water at 50℃; for 3h; Green chemistry; | 85% |
-
-
617-84-5
N-formyldiethylamine

-
-
98837-46-8, 67764-52-7
P,P-diphenyl-N-(phenylmethylene)phosphinic amide

-
-
132868-67-8
(tert-butyldimethylsilyl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| Stage #1: N-formyldiethylamine; (tert-butyldimethylsilyl)(phenyl)methanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; Stage #2: P,P-diphenyl-N-(phenylmethylene)phosphinic amide In tetrahydrofuran at -78℃; for 0.5h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 99% |

-
-
617-84-5
N-formyldiethylamine

-
-
70-55-3
toluene-4-sulfonamide

-
-
29397-20-4
(E)-N'-((4-methylphenyl)sulfonyl)-N,N-diethylformimidamide

| Conditions | Yield |
|---|---|
| With diazoacetic acid ethyl ester; zinc trifluoromethanesulfonate In cyclohexane for 12h; Reflux; stereoselective reaction; | 99% |
| With thionyl chloride In chloroform at 60℃; for 3h; | 96% |
-
-
617-84-5
N-formyldiethylamine

-
-
19952-47-7
4-chlorobenzo[d]thiazol-2-amine

-
-
112446-70-5
N,N-diethyl-N',N''-bis(4-chloro-2-benzothiazolyl)triaminomethane

| Conditions | Yield |
|---|---|
| With benzenesulfonyl chloride In pyridine | 98% |
-
-
617-84-5
N-formyldiethylamine

-
-
824-98-6
m-methoxybenzyl chloride

-
-
27958-95-8
N,N-diethyl-3-(methoxyl)benzenemethanamine

| Conditions | Yield |
|---|---|
| With NHC-Pd(II)-Im; sodium hydroxide In water at 50℃; for 3h; Inert atmosphere; Schlenk technique; | 98% |
| With potassium hydroxide In water at 50℃; for 3h; Green chemistry; | 92% |

| Conditions | Yield |
|---|---|
| With silver trifluoromethanesulfonate at 130℃; for 2h; Inert atmosphere; Schlenk technique; | 98% |
| With silver hexafluoroantimonate In N,N-dimethyl-formamide at 80℃; for 20h; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With ammonium iodide at 90℃; for 0.25h; Reagent/catalyst; Temperature; Time; Microwave irradiation; | 97% |
| With iodine In neat (no solvent) at 80℃; for 2h; Schlenk technique; Green chemistry; | 80% |

-
-
617-84-5
N-formyldiethylamine

| Conditions | Yield |
|---|---|
| With water In dichloromethane at 60℃; for 24h; | 97% |
-
-
617-84-5
N-formyldiethylamine

-
-
80716-36-5
2-(2-methylbenzyl)benzoic acid

-
-
80716-39-8
N,N-diethyl-2-(2-methylbenzyl)benzamide

| Conditions | Yield |
|---|---|
| With thionyl chloride for 20h; | 96% |

-
-
1036248-62-0
4,4'-benzene-1,4-diylbis(1H-pyrazole)

-
-
617-84-5
N-formyldiethylamine

| Conditions | Yield |
|---|---|
| In further solvent(s) mixt. Co(OTf)2, 1,4-benzenedipyridyl and HCONEt2 in sealed evacuated tube was heated at 150°C for 6 days; ppt. was filtered, washed with HCONEt2 and dried in vacuo for 30 min; elem. anal.; | 95% |
| In further solvent(s) react. in N,N-diethylformamide at 130°C; |

-
-
617-84-5
N-formyldiethylamine

-
-
554-95-0
benzene-1,3,5-tricarboxylic acid

-
-
71-91-0
tetraethylammonium bromide

| Conditions | Yield |
|---|---|
| In further solvent(s) C6H3(COOH)3, (C2H5)4NBr, In(NO3)3*xH2O in N,N-diethylformamide was heated at 120°C for 4 days, cooled to room temp.; washed with ethanol; elem. anal.; | 95% |

-
-
617-84-5
N-formyldiethylamine

-
-
766-77-8
Dimethylphenylsilane

-
-
1366432-35-0
phenyldimethylsilyloxymethyldiethylamine

| Conditions | Yield |
|---|---|
| With CpMn(CO)3 for 16h; Irradiation; | 95% |
-
-
617-84-5
N-formyldiethylamine

-
-
119701-95-0
N-(p-methoxybenzylidene)diphenylphosphinamide

| Conditions | Yield |
|---|---|
| Stage #1: N-formyldiethylamine; (tert-butyldimethylsilyl)(naphthalen-2-yl)methanone With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.333333h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; Stage #2: N-(p-methoxybenzylidene)diphenylphosphinamide In tetrahydrofuran at -78℃; for 0.5h; Brook Silaketone Rearrangement; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 95% |


| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran at 25℃; for 0.0166667h; | 95% |
-
-
95-16-9
1,3-Benzothiazole

-
-
617-84-5
N-formyldiethylamine

-
-
24255-48-9
N,N-diethylbenzo[d]thiazol-2-amine

| Conditions | Yield |
|---|---|
| With silver(I) acetate; sodium t-butanolate In neat (no solvent) at 120℃; for 5h; Inert atmosphere; | 95% |
-
-
617-84-5
N-formyldiethylamine

-
-
501-52-0
3-Phenylpropionic acid

-
-
18859-19-3
N,N-diethyl-3-phenylpropanamide

| Conditions | Yield |
|---|---|
| With sulfur trioxide pyridine complex at 160℃; Schlenk technique; Inert atmosphere; | 95% |
-
-
617-84-5
N-formyldiethylamine

-
-
15959-53-2
N-(1-phenyl)sulfamide

| Conditions | Yield |
|---|---|
| With trichlorophosphate In dichloromethane at 40℃; for 9h; | 95% |
-
-
855766-92-6
4,4'-bis-(1H-imidazol-1-yl)biphenyl

-
-
617-84-5
N-formyldiethylamine

-
-
1394899-62-7
2′,3′,5′,6′-tetramethyl(1,1′:4′,1″-terphenyl)-4,4″-dicarboxylic acid

| Conditions | Yield |
|---|---|
| at 120℃; for 24h; Sealed tube; | 95% |

N,N-Diethylformamide Consensus Reports
Reported in EPA TSCA Inventory.
N,N-Diethylformamide Specification
The N,N-Diethylformamide, with the CAS registry number 617-84-5, is also known as Diethylformamide. It belongs to the product categories of Amides; Carbonyl Compounds; Organic Building Blocks. Its EINECS number is 210-533-2. This chemical's molecular formula is C5H11NO and molecular weight is 101.15. What's more, its systematic name is N,N-Diethylformamide. This chemical is stable at common pressure and temperature, and it should be sealed and stored in a cool, ventilated and dry place. This chemical is an industrial solvent claimed to have HIV protease inhibitory action. It is used as solvent, corrosion remover and vermifuge.
Physical properties of N,N-Diethylformamide are: (1)ACD/LogP: 0.19; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.19; (4)ACD/LogD (pH 7.4): 0.19; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 30.21; (8)ACD/KOC (pH 7.4): 30.21; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 20.31 Å2; (13)Index of Refraction: 1.418; (14)Molar Refractivity: 29.121 cm3; (15)Molar Volume: 115.641 cm3; (16)Polarizability: 11.545×10-24cm3; (17)Surface Tension: 27.8 dyne/cm; (18)Density: 0.875 g/cm3; (19)Flash Point: 69.109 °C; (20)Enthalpy of Vaporization: 41.38 kJ/mol; (21)Boiling Point: 177.499 °C at 760 mmHg; (22)Vapour Pressure: 1.0 mmHg at 25°C.
Preparation: this chemical can be prepared by diethylamine and formaldehyde at the ambient temperature. This reaction will need reagent KI and solvent H2O. The yield is about 85%.
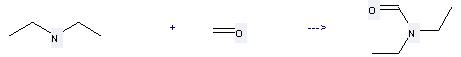
Uses of N,N-Diethylformamide: it can be used to produce Diethyltrifluoromethylamine at the temperature of 150 °C. It will need reagents sulphur tetrafluoride, potassium fluoride with the reaction time of 48 hours. The yield is about 89.5%.

When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: O=CN(CC)CC
(2)Std. InChI: InChI=1S/C5H11NO/c1-3-6(4-2)5-7/h5H,3-4H2,1-2H3
(3)Std. InChIKey: SUAKHGWARZSWIH-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LDLo | intraperitoneal | 3200mg/kg (3200mg/kg) | Therapie. Vol. 26, Pg. 409, 1971. | |
| rat | LD50 | intraperitoneal | 1740mg/kg (1740mg/kg) | Biochemical Journal. Vol. 85, Pg. 72, 1962. | |
| rat | LD50 | unreported | 1740mg/kg (1740mg/kg) | "Prehled Prumyslove Toxikologie; Organicke Latky," Marhold, J., Prague, Czechoslovakia, Avicenum, 1986Vol. -, Pg. 330, 1986. |
Related Products
- N-[(10-Oxido-9,10-dihydro-9-oxa-10-phosphaphenanthrene)methyl]-1,3,5-triazine-2,4,6-triamine
- N10-(Trifluoroacetyl)pteroic acid
- N-[1,1'-Biphenyl]-4-yl-9,9-dimethyl-9H-fluoren-2-amine
- N-[1,1'-Biphenyl]-4-yl-9,9-dimethyl-9H-fluoren-3-amine
- N-[1,1'-Biphenyl]-4-yl-N-(4-bromophenyl)-9,9-dimethyl-9H-fluoren-2-amine
- N-[1,1-Bis[(acetyloxy)methyl]-3-(4-octylphenyl)propyl]acetamide
- N'-[(1,1-Dimethylethoxy)carbonyl]-N-[(9H-fluoren-9-ylmethoxy)carbonyl]-N'-methyl-L-lysine
- N1,1-Diphenyl-1,2-ethanediamine
- N-(1,2-Dimethylpropyl)-2-pyridinamine
- N<sup xmlns="">1</sup>-(3,4-DIMETHYL-5-ISOXAZOLYL)SULFANIL-AMIDE LITHIUM SALT
- 61785-35-1
- 61786-07-0
- 61786-58-1
- 617-86-7
- 617880-45-2
- 61788-32-7
- 61788-33-8
- 61788-40-7
- 61788-44-1
- 61788-45-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View